Abstract
Background:
Aberrant DNA methylation is an epigenetic hallmark of oncogenesis implicated in the pathogenesis of myelodysplastic syndromes (MDS). TET2 is one of the most frequently mutated genes in MDS, and is associated with net global hypermethylation in animal models. However, patients with MDS rarely present with a single somatic mutation, and thus methylation must be interpreted in the context of several mutated genes. We postulate that TET2 mutant MDS patients are not uniform and that epigenetic subgroups associated with clinical outcomes may exist. Methylation profiles at key regulatory regions could therefore serve as biomarkers in conjunction with somatic mutations. The aim of this study was to characterize the epigenetic heterogeneity of TET2 mutant MDS, and how specific methylation patterns relate to the molecular and clinical features of patients.
Methods:
To study methylation patterns in 96 patients with MDS, we utilized bisulfite padlock probes (BSPP), a novel enrichment technique (Deng et al. Nat Biotechnol 2009 and Diep et al. Nat Methods 2012) that targets ~500,000 unique CpGs in selected regions known to contain differentially methylated sites that have regulatory relevance, including transcription repressor CTCF binding sites, DNase I accessibility clusters, all microRNA genes, and all RefSeq gene promoters. BSPP was performed on genomic DNA purified from bone marrow mononuclear cells from untreated patients diagnosed with MDS that were genetically characterized in previous studies (Bejar et al. N Engl J Med 2011 and J Clin Oncol 2012). BSPP products were PCR amplified with primers containing individual barcodes as well as adaptors compatible with Illumina sequencers, allowing for massively parallel high throughput sequencing and digital quantification of CpG methylation.
Results:
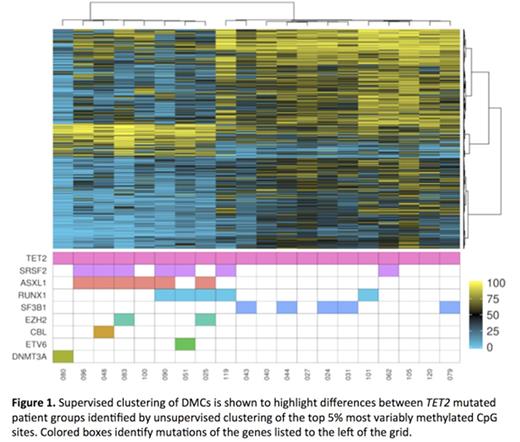
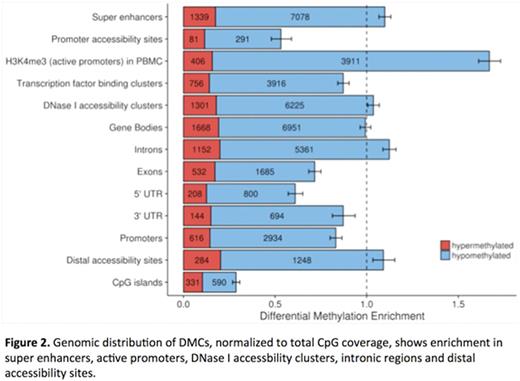
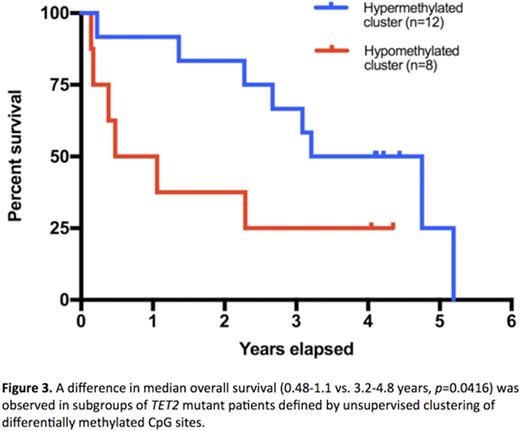
Unsupervised clustering of the top 5% most variably methylated CpGs among 96 patients did not identify a correlation between gene mutations and methylation patterns. However, unsupervised analysis on the 26 patients with TET2 mutations (mean VAF 56.1%, not determined in 6 patients) sorted 20 patients into one of two discrete methylation clusters. Supervised analysis comparing these two clusters is shown in figure 1, highlighting the 9712 differentially methylated cytosines (DMCs) with >25% methylation difference and FDR-corrected p <0.05. The cluster with net hypermethylation of DMCs included 12 patients who had a lower mutational burden compared with the 8 patients in the relatively hypomethylated cluster (mean 1.8 vs. 3.4 mutations/patient, respectively). Patients in the hypomethylated cluster more frequently harbored mutations associated with poor prognosis (ASXL1 in 6, SRSF2 in 5, EZH2 in 2). In this cluster, 7775 DMCs were hypomethylated and 1937 were hypermethylated. DMCs were enriched in super enhancers, active promoters, DNase I accessibility clusters and intronic regions and underrepresented in CpG islands (Figure 2). Assessment of genes overlapping with DMCs compared to genes at all tested CpG sites were enriched for pathways involved in embryonic development and mesenchymal differentiation. Clinically, the hypomethylated patients were significantly more thrombocytopenic with a mean platelet count (1000/mm3) of 54.9±11.2 compared to a mean platelet count of 181.8±43.8 for the hypermethylated patients (p=0.045). The median follow up was 2.9 years, and the median overall survival was 0.48-1.1 years for the hypomethylated group versus 3.2-4.8 years for the hypermethylated group (Figure 3; HR 5.2, 95% CI 1.8-15.0, p=0.042).
Discussion:
We used BSPP to identify an epigenetic signature associated with survival among TET2 mutated patients with MDS. Unsupervised clustering identified methylation subgroups within TET2 mutants that are distinguished by differences in mutational burden, platelet count, and median overall survival. Between these two subgroups, DMCs are distributed in distinct patterns that identify regulatory regions that could serve as clinical biomarkers. Larger methylation profiling studies of MDS patients with various mutations and known treatment outcomes are ongoing. These will increase our ability to incorporate both epigenetic and genetic biomarkers into clinical practice.
Bejar:Genoptix: Consultancy, Honoraria, Patents & Royalties: No royalties; Celgene: Consultancy, Honoraria; Foundation Medicine: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal