Abstract
Background: The 2016 revision to the World Health Organization (WHO) classification of myeloid neoplasms has specifically categorized germline mutations that are associated with myeloid clonal evolution (Arber et al. Blood 2016). This group consists of myeloid neoplasms with an isolated germline predisposition (CEBPA, DDX41), myeloid neoplasms associated with congenital thrombocytopenia (ETV6, RUNX1, ANKRD26) and germline myeloid neoplasms with multi-organ dysfunction (GATA2, chromosomal breakage disorders, telomere biology disorders etc). We carried out this study to describe the clinical spectrum of germline disorders with predisposition to myeloid neoplasms as categorized by the 2016 WHO classification revision.
Methods: After Institutional Review Board (IRB) approval, the adult and pediatric bone marrow failure syndrome database (1990-2016) and the electronic medical record were queried for germline disorders involving GATA2, CEBPA, DDX41, ETV6, RUNX1, ANKRD26, Down syndrome and Noonan syndrome. Chromosomal breakage assays (Diepoxybutane/Mitomycin-C), flow-fluorescent in-situ hybridization (FISH) for telomere length assessment, Fanconi anemia complementation assays and Sanger/Next Generation sequencing (NGS) for the aforementioned germline disorders with myeloid predisposition were carried out in Clinical Laboratory Improvement Amendments (CLIA)-certified laboratories. These disorders were then classified based on the 2016 WHO classification revision.
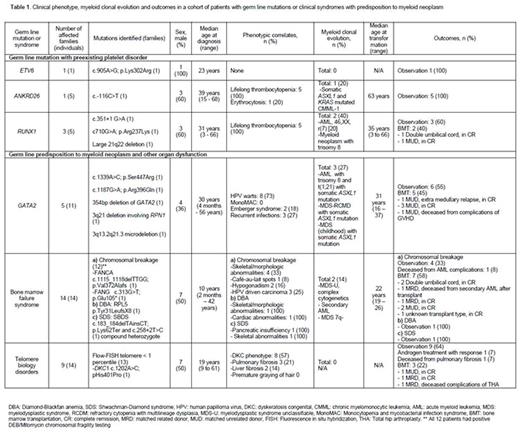
Results: 54 individuals (37 families) were included in the study. Eleven (20%) patients belonging to 5 families were identified as having germline mutations with a preexisting platelet disorder: ETV6 (n=1), ANKRD26 (n=5), RUNX1 (n=5). Forty-three (79%) patients (32 families) had inherited syndromes with multi-organ dysfunction: GATA2 (n=11, 26%), bone marrow failure syndromes (n=14, 33%) and telomere biology disorder (n=14, 33%). There was one patient with neurofibromatosis with a germline PTPN11 mutation who developed juvenile myelomonocytic leukemia, while there were three patients with Down syndrome; 2 with transient abnormal myelopoiesis and one who developed acute megakaryocytic leukemia. The clinical phenotype, prevalence and characteristics of myeloid clonal evolution and outcomes are presented in Table 1. No patients with germline CEBPA or DDX41 mutations were identified.
Patients with germline platelet disorders did not have any prominent non-hematological manifestations. Erythrocytosis (20%) with long-standing thrombocytopenia (100%) was a unique feature associated with ANKRD26 mutations. Non-hematologic clues such as human papillomavirus (HPV)-driven warts, primary lymphedema (Emberger syndrome) and frequent atypical infections with monocytopenia were seen in patients with germline GATA2 mutations, and preceded myeloid clonal evolution (morphologic, cytogenetic and molecular). Notably, the age at presentation and penetrance of myeloid transformation was variable, with individuals from the same family developing symptoms during the first decade of life and others remaining asymptomatic to date (fifth decade). Somatic ASXL1 mutations were detected in all 3 (100%) patients with GATA2 mutations and in one patient with ANKRD26 mutation that developed myeloid clonal evolution. In our study myeloid clonal evolution was seen in 40% with RUNX1 mutations, 27% with GATA2 mutations, and 20% with ANKRD26 mutations. We could not calculate the same for bone marrow failure syndromes as the total number of cases seen are still being assessed. Outcomes with allogeneic stem cell transplantation were favorable in appropriately selected patients (Table 1).
Conclusion: The 2016 WHO revision to the classification of myeloid neoplasms highlights the importance of recognition and molecular characterization of germline mutations (syndromic and non-syndromic) with risk for myeloid clonal evolution. While some of these disorders (GATA2, Fanconi anemia, telomere biology disorders) may have important non-hematological clues, many present with isolated thrombocytopenia (RUNX1, ETV6). The age and frequency of myeloid evolution is highly variable. Acquisition of somatic ASXL1 mutations at the time of clonal myeloid transformation highlights the role of epigenetic dysregulation in disease evolution.
Kenderian:Novartis: Patents & Royalties, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal