Abstract
Introduction: Primary refractory DLBCL is an extremely difficult condition to treat and represent an unmet medical need. The clarification of the molecular pathogenesis of this disease can contribute the development of new therapeutic possibilities. PVT1 is located adjacent to MYC at 8q24 and a non-protein coding gene. PVT1 produces a variety of long non-coding RNAs (lncRNAs) and has now emerged as a potential regulator in the pathogenesis of a lot of cancers. However, the precise biological and clinical significance of PVT1 remains largely unknown. In this study we established a novel human DLBCL cell line with hsr (homogeneous staining region) of 8q24 inducing high expression of PVT1 lncRNAs, named AMU-ML2. The cell line was established from a patient with primary refractory DLBCL before initiation of chemotherapy. We analyzed the genetic characteristics and investigated the drug sensitivity of the cell line in comparison with other B-cell lymphoma cell lines which had 8q24 abnormalities.
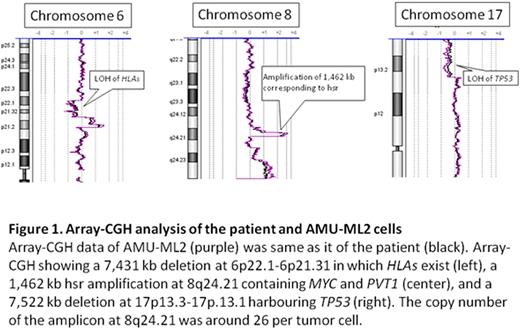
Case: A 64-year-old man was diagnosed as DLBCL involved with bone marrow and pleural effusion. He was refractory to initial R-CHOP and subsequent R-hyper-CVAD/MA therapy and died of Trichosporon asahiisepsis 6 months after diagnosis. His lymphoma cells at diagnosis were positive for CD19, CD20, BCL6 and HLA-DR, and negative for CD3, CD5, CD10, cyclinD1, BCL2, MUM1, TP53 and EBER by flow cytometry and/or immunohistochemical staining, indicative of germinal center B-cell-like (GCB) DLBCL. The representative karyotype of cells was 46,XY, including add(6)(p11), add(8)(q24), hsr 8q24, add(9)(p13) and add(17)(p11.2).
Establishment of the cell line: After 2 weeks of culture, the cells in pleural effusion collected before chemotherapy started to grow in suspension. The cell line was designated as AMU-ML2 after confirmation that the cells started growing again after the conventional freeze-thaw procedure.
Materials and methods: B-cell lymphoma cell lines; AMU-ML2, SU-DHL-10, Raji, P3HR-1 and VAL were used in the present study. These cell lines had t(8;14)(q24;q32) or 8q24 amplification. The genomic aberration of AMU-ML2 was analyzed by cytogenetics including G-banding and FISH (fluorescence in situ hybridization) combined with array-CGH (SurePrint G3, Agilent). The TaqMan real time RT-PCR was used for measurement of expression levels of MYC and PVT1. MTT assay was used for the cell proliferation to analyze the drug sensitivities of cyclophosphamide, doxorubicin, vincristine (VCR) and prednisolone.
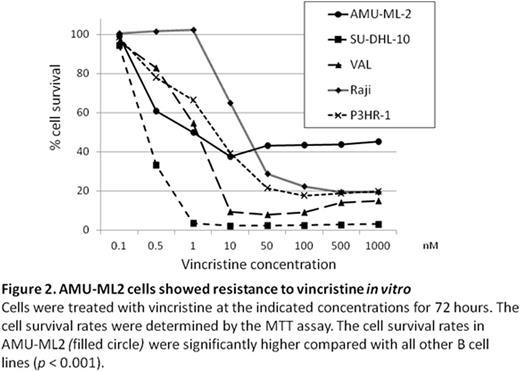
Results: AMU-ML2 cells showed same immunophenotypic feature and karyotype as the primary sample from the patient. FISH using a MYC/IGH probe set showed no fusion signal for IGH and MYC; however, the MYC copy number was extremely increased, corresponding to hsr on chromosome 8q24. Array-CGH revealed that a 1,462 kb region, containing both the entire MYC and PVT1 genes at 8q24.21, was amplified, with greater than 20 copies present in cells (Figure 1). In addition to amplification of the MYC/PVT1 region at 8q24, 14 additional copy number alterations were detected. These included segment losses on 6p22.1-6p21.31 and 17p13 led to the LOH of human leukocyte antigen (HLA) loci and TP53, respectively (Figure 1). Using the real time RT-PCR, the expression level of the PVT1 lncRNAs were highest in AMU-ML2 among other B-cell lymphoma cell lines that we used, while the expression level of MYC in AMU-ML2 was relatively low. The proliferation rate of AMU-ML2 was significantly higher after 72-h exposure to VCR (100, 500 and 1,000 nM) compared with other cell lines (Figure 2).
Discussion: AMU-ML2 was established in 2-week culture from a refractory patient before starting the chemotherapy, therefore this cell line seems to reflect the real nature of primary refractory DLBCL, not related to chemotherapy and/or cell culture. MYC amplification and the LOH of TP53 and HLA may contribute to the development of lymphoma in our case. High expression of PVT1 lncRNAs was likely to be more related with drug resistance to VCR than MYC expression, although AMU-ML2 had co-amplification of MYC and PVT1. Thus, AMU-ML2 can provide insight into the genetic and biological features and the therapeutic approaches in primary refractory DLBCL.
Ueda:Mundipharma KK: Consultancy; Kyowa Hakko Kirin: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal