Abstract
Introduction: Klotho is an anti-aging gene with an extracellular domain. Mice with Klotho knockout exhibited obvious impairment in B-cell development. Evolving evidence indicates that Klotho modulates the proliferation and survival via targeting insulin-like growth factor-1 receptor (IGF-1R) in several cancers. However, the expression and biological role of Klotho in B-cell non-Hodgkin lymphoma (B-NHL) has not been elucidated to date. We hypothesized that Klotho could modulate the tumor growth and predicts prognosis in diffuse large B-cell lymphoma (DLBCL) through inhibiting IGF-1R activation. The aim of this study is to characterize the functional significance of Klotho and the therapeutic potential of its secreted form in DLBCL.
Methods: Lymph nodes samples from 50 de novo DLBCL and 20 reactive hyperplasia cases were collected with informed consents. Klotho expression were assessed by Immunohistochemistry. CD19+ B-cells and peripheral blood mononuclear cells were isolated with informed consents from healthy donors. Expression levels of Klotho mRNA and protein in DLBCL cells were determined by quantitative RT-PCR and western blotting. Lentivirus vectors either encoding Klotho (LV-KL) or empty lentiviral vector (LV-Con) were stably transfected into DLBCL cells. Cell viability and apoptosis were analyzed by cell counting kit-8 and Annexin V-PE/7AAD staining. Animal experiments were performed in accordance with the principles of the Institutional Animal Care. SCID-Beige mice were subcutaneously injected with DLBCL cells to establish xenograft model.
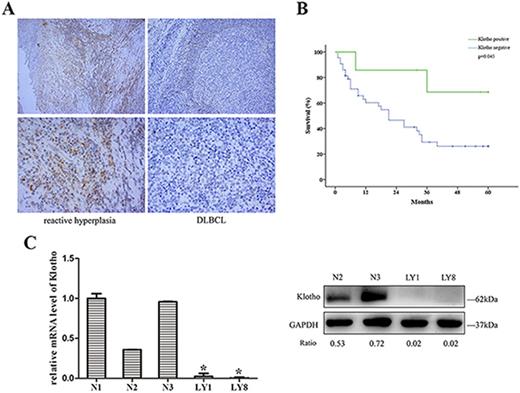
Results: We observed markedly decreased level of Klotho protein in DLBCL lymph nodes (Fig. 1A). Expression of Klotho protein exhibited significantly negative correlation with Ann Arbor stage of DLBCL patients (p=0.002). Level of Klotho protein was negatively correlates with the media overall survival (OS), suggesting lower Klotho expression is associated with poor OS in DLBCL ((Fig. 1B, p=0.045). Reduction of Klotho was also confirmed in DLBCL cell lines at mRNA and protein level (Fig. 1C).
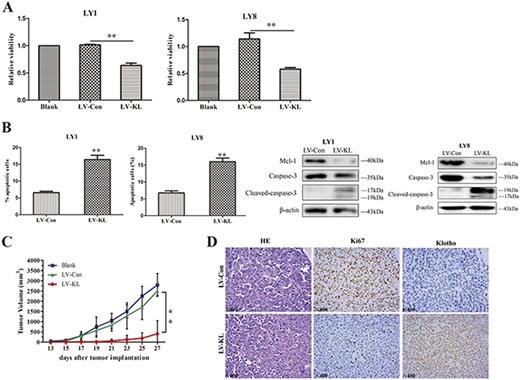
We next functionally interrogated the role of Klotho in DLBCL cell lines and xenograft models. Stably expression of LV-KL in DLBCL cell lines resulted in dramatically decreased cell proliferation and incremental apoptotic rates when compared to LV-Con (Fig. 2A and B). We validated the changed expression of critical targets known to govern apoptosis in DLBCL cells transfected with LV-KL. Xenograft models with Klotho overexpression revealed significantly abrogated tumor growth compared to control group (Fig. 2C). Interestingly, lower levels of Ki67 were observed in mice treated with LV-KL (Fig. 2D). These results highlighted the proliferation-inhibitory and apoptosis-inductive activities of Klotho in DLBCL cells.
The underlying mechanism driving the tumor suppressive potential of Klotho was investigated. Surprisingly, we observed that the Klotho-induced inhibition of cell viability was only fewer restored by IGF-1 in DLBCL cells transfected with LV-KL (Fig. 3A). Reductive phosphorylation of IGF-1R and its downstream targets (AKT and ERK1/2) were observed in DLBCL cells with Klotho overexpression (Fig. 3B). In addition, we evaluated the regulation of Klotho on IGF-1R signaling in vivo. Decreased phosphrolation of IGF-1R as well as its downstream targets were observed in mice treated with LV-KL compared to the control group (Fig. 3C).
Lastly, we explored the activity of secreted Klotho protein (rhKL). The rhKL was found to be active in vitro and significantly reduced the viabilities of DLBCL cells (Fig. 3D). Moreover, combination with rhKL increased the sensitivity of DLBCL cells to adriamycin. The in vivo activity of rhKL in DLBCL xenograft model was also detected. Significantly decreased tumor volumes were noted in mice treated with rhKL compared with those treated with vehicle control (Fig. 3E). Moreover, reductive expression level of Ki67 was observed in rhKL-treated group (Fig. 3F).
Conclusions: Our observations identified for the first time that loss of Klotho expression contributed to the development and poor prognosis via activating IGF-1R in DLBCL. Given the in vivo tumor suppressive activity of secreted Klotho protein, it may serve as a potential strategy for the development of novel therapeutic interventions for DLBCL.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal