Abstract
Introduction: Cyclic thrombocytopenia (CT) is a rare disease characterized by periodic fluctuations in platelet counts. The etiology of this disease has not been fully elucidated. Here, we present molecular characterization of CT in a 53-year-old male patient who has platelet counts ranging between 1 x 109/L to >400 x 109/L during a cycle period of 40 days which has occurred over several years.
Methods: Blood transcriptome profiles and plasma cytokine levels were investigated in 24 samples that were sequentially collected every 3-4 days to cover two complete cycles. Total RNA was extracted after lysing red blood cells. 3SEQ (3′-end RNA sequencing for expression quantification) was performed for transcriptome profiling. SAMSeq quantitative analyses were conducted to identify differentially expressed genes. Three screening criteria were applied to this data set to identify a panel of exclusive platelet-specific genes. Plasma TPO (thrombopoietin)/testosterone/ estradiol/CD41a/CD42b levels were detected by ELISA. A total of 62 cytokines were screened using a luminex immunoassay. MPL (TPO receptor) gene was Sanger-sequenced using blood DNA and mRNA templates. DNA was extracted from the patient's hair follicles to exam the presence of the identified MPL mutation, and in-silico predictions were conducted to evaluate the MPL mutation. The wild-type (WT) and c.1210G>A mutant MPL expression constructs were generated and transfected into Ba/F3 cells for stable MPL expression. TPO-stimulated, IL-3 independent growth of MPL-expressing Ba/F3 cells were assessed to determine the function of MPL proteins.
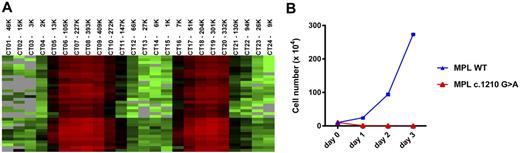
Results: Plasma TPO levels cycled between 6 to 2745 pg/mL during the sampling period; TPO levels inversely mirrored the fluctuation of platelet counts and preceded the latter by 3-4 days. Additional plasma cytokines demonstrated either an inverse correlation (e.g. FasL, GM-CSF, and TNF-β, etc.), or well synchronized correlation (e.g. BDNF, PDGF-BB, and RANTES etc.), with platelet counts. The patient was negative for platelet autoantibodies. Transcriptome analysis revealed 977 genes with expression that positively correlated with platelet count changes. Unsupervised clustering stratified these genes into subgroups, including: i) platelet-specific genes, which precede platelet count changes by 3-4 days; ii) platelet-modulated genes, which follow platelet count changes and are regulated by coagulation factors (e.g. F2) and platelet-contained growth factors (e.g. PDGF-BB and EGF); and iii) neutrophil-specific genes, which precede platelet count changes by 7-10 days. Among platelet-specific genes, 34 genes were identified to have >50-fold induction over a cycle, and are present in the platelet transcriptome [Rowley JW, 2011]. A heat map of these exclusive platelet genes is shown in Figure 1A.
A novel MPL c.1210G>A heterozygous mutation, which leads to a p.Gly404Arg substitution, was identified in both blood and hair follicle DNA samples of the patient. This substitution resides in a highly conserved site and is predicted to be deleterious. In addition, the mutation creates a novel splice site, which results in the insertion of a 36 bp intron fragment in approximately 15% of MPL transcripts. In vitro analysis confirmed that MPL c.1210G>A is a loss-of-function (LOF) variant. Ba/F3 cells that express WT MPL exhibited IL-3 independent growth in the presence of TPO; however, the Gly404Arg mutant failed to support TPO-stimulated growth (Figure 1B).
Conclusion: We describe a patient with CT who exhibits fluctuations of multiple cytokines including an inverse correlation between TPO and platelet counts, and profound gene expression changes in neutrophil- and platelet-specific gene expressions which precede platelet count fluctuations. These data suggest that the cyclic megakaryopoiesis and thrombopoiesis underlies pathogenesis of the disease. Moreover, the identified novel LOF MPL allele may contribute to the disease pathogenesis. The biologic and molecular annotation of this case presents a unique opportunity to examine the transcriptional regulation of platelet homeostasis.
(A) Heat map of 34 genes that are exclusively expressed in platelet. (B) Growth of Ba/F3 cells expressing WT or c.1210G>A MPL in IL-3 free medium containing TPO (50 ng/mL). Average viable cell numbers of duplicate experiments were plotted with SD as error bars.
(A) Heat map of 34 genes that are exclusively expressed in platelet. (B) Growth of Ba/F3 cells expressing WT or c.1210G>A MPL in IL-3 free medium containing TPO (50 ng/mL). Average viable cell numbers of duplicate experiments were plotted with SD as error bars.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal