Abstract
Background: SCD is characterized by chronic vaso-occlusive crises and multiorgan failure resulting in poor quality of life and early mortality (Bhatia/Cairo et al, BMT 2014). There is presently no curative therapy for patients with high risk SCD other than HLA-identical sibling AlloSCT. (Freed/Cairo et al BMT 2012). However, less than 15% of eligible SCD patients have an unaffected MSD with a 10-15% increase of graft failure and TRM (Talano/Cairo et al, EJH, 2015). Similarly, most patients lack a matched related donor and UCB is an inferior source in SCD recipients (Radhakrishman/Cairo et al, BBMT 2013). Haploidentical familial donors with SCD trait offers an opportunity for a new donor source for children with high risk SCD. To overcome HLA barriers, Geyer/Cairo et al (BJH, 2012) demonstrated that T cell depletion using CD34 enriched HPC products with PB MNC addback transplanted in pediatric recipients utilizing MUD was associated with sustained engraftment, low risk of aGVHD but limited by delayed immune reconstitution. Efforts to use FHI donors and T replete AlloSCT in patients with SCD were associated with high rates of graft failure (Bolanes-Meade J et al Blood 2012; Ruggieri et al BBMT 2011). We previously reported FHI CD34 enriched/PB MNC addback AlloSCT is feasible and well tolerated in patients with high risk SCD (Abikoff/Cairo, ASBMT 2015).
Objective: To characterize immunological reconstitution following FHI AlloSCT with CD34 enriched grafts with PB MNC addback in children and adolescents with high risk SCD.
Methods: 15 patients were evaluatedpretransplant at D+30, 60, 100 and 180 following FHI AlloSCT. GCSF mobilized HPC were collected by apheresis (Spectra OPTIA, Terumo BCT) and products underwent CD34 enrichment using the CliniMACS cell separation system (materials generously supplied by Miltenyi Biotec, Cambridge , MA) with a PB MNC addback dose of 2x10*5 CD3/kg. Immune cell and subset reconstitution was assessed by flow cytometry. NK function was determined by cytotoxic activity against K562 tumor targets at 10:1 E:T ratio by europium release assay and intracellular LAMP-1 (CD107a) and granzyme B expression by flow cytometry. Whole blood, T cell and RBC chimerism (CD71) determined by flow cytometry and by STR.
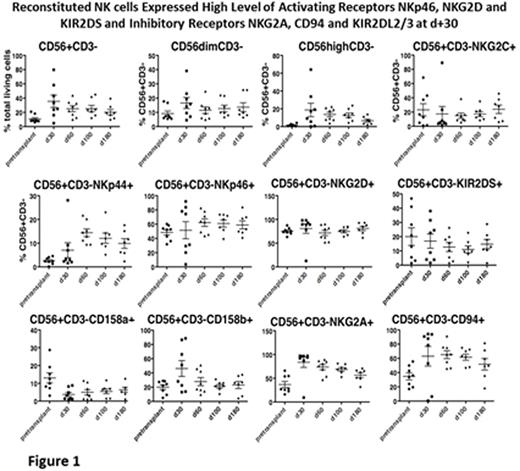
Results: Patients achieved neutrophil and platelet engraftment in a median time of 10 and 16 days, respectively. By D+30, median whole blood donor chimerism was ≥93% and ≥95% at most recent followup (D+30-730). Median donor chimerism in the erythroid lineage was 95% by D+60, with 7 of 13 patients ≥99% at D+30. This was maintained at most recent followup (D+30-730). Median T cell chimerism was 90% (D+60-550) and median NK cell chimerism was 90% by D+30 and maintained at ≥95% through D+730. NK (CD3-/56+) and NKT (CD3+/56+) cell reconstitution following FHI AlloSCT was rapid and peaked at D+30 (35.5±8.6%, 271x10*3/ul; 14.2±4%, 179x10*3/ul, respectively). Moreover, there was robust NK cell receptor expression reconstitution with high levels of activating receptors, NKp46, NKG2D and KIR2DS and inhibitory receptors NKG2A, CD94 and KIR2DL2/3 at D+30 [Fig 1]. NK cytotoxicity against K562 at E;T 10:1 peaked at D+30 (26±3%) and D+180 (28±3%) compared to pretransplant (16±2%, p<0.01). NK activation marker, CD107a, peaked at D+30 (37±9%) and D+180 (41±6%) and there was robust granzyme B degranulation at D+30. CD3+, CD4+, CD8+ and CD19+ immune reconstitution occurred between D+180 and D+270. One year absolute (mean±SEM) cells/ul of CD3+, CD4+, CD8+, CD19+ and CD56+ was 795±168, 408±102, 375±90, 815±352 and 204±37, respectively. [Fig 2]
Conclusion: Immune reconstitution and donor chimerism was relatively rapid after FHI AlloSCT with CD34 enriched grafts with PB MNC addback in high risk SCD patients. The donor MNC addback after CD34 selection may in part contribute to rapid engraftment and immune reconstitution along with sustained donor chimerism.
This research was supported by FDA grant 5R01FD004090.
Cairo:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal