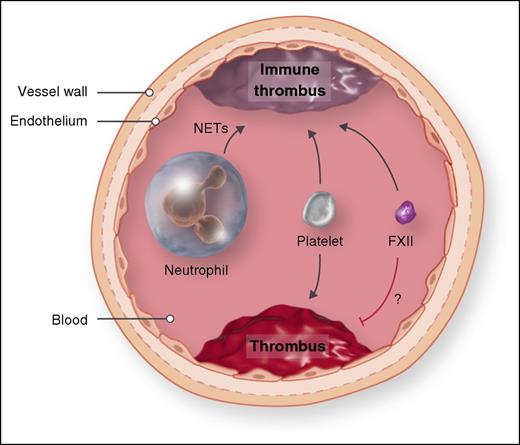
Comparison of two mouse models of venous thrombosis. In the inferior vena cava stenosis model of venous thrombosis, the inferior vena cava is ligated to reduce blood flow by ∼90%. Platelets, FXII, and neutrophils contribute to immune thrombosis. In the spontaneous venous thrombosis model, mice are treated with small interfering RNAs to reduce levels of the anticoagulants antithrombin and protein C. Platelets contribute to thrombosis in this model, whereas there is no role for neutrophils, and decreasing FXII increases thrombosis. NET, neutrophil extracellular trap. Professional illustration by Somersault18:24.
Comparison of two mouse models of venous thrombosis. In the inferior vena cava stenosis model of venous thrombosis, the inferior vena cava is ligated to reduce blood flow by ∼90%. Platelets, FXII, and neutrophils contribute to immune thrombosis. In the spontaneous venous thrombosis model, mice are treated with small interfering RNAs to reduce levels of the anticoagulants antithrombin and protein C. Platelets contribute to thrombosis in this model, whereas there is no role for neutrophils, and decreasing FXII increases thrombosis. NET, neutrophil extracellular trap. Professional illustration by Somersault18:24.
Venous thrombosis is a major medical problem in industrialized countries.2,3 It is multifactorial with both genetic and acquired risk factors. Genetic factors include either deficiencies in the anticoagulants antithrombin, protein C, or protein S, or the presence of FV or prothrombin variants (FV Leiden and prothrombin 20210A).2 Acquired risk factors include older age (older than age 50 years), use of hormonal therapy (birth control or replacement), trauma, major surgery, prolonged immobilization, and certain diseases, such as cancer and antiphospholipid antibody syndrome.3 Vein damage and/or activation of the endothelium, changes in blood flow, and the presence of procoagulant factors in blood appear to be the triggers of venous thrombosis. In patients with pancreatic cancer, tumor-derived, tissue factor–positive extracellular vesicles may induce thrombosis, whereas in patients with artificial heart valves, FXII may drive thrombosis. Most venous thrombi are thought to originate in valve pockets as a result of the low oxygen tension, and they develop over a period of hours to days.
Mouse models are used to provide new insights to human disease. There are a variety of mouse models of venous thrombosis available to researchers that use large (inferior vena cava [IVC]), medium (femoral vein), or small (mesenteric and cremaster venules) vessels.4 The IVC has become the most popular vessel for studying venous thrombosis because it is the largest vein available in the mouse. Some mouse models are better than others. Adding ferric chloride to the IVC and measuring thrombosis at 30 minutes could be considered a good model of trauma-induced thrombosis, but is not a good model of nontrauma-related venous thrombosis in humans. Although complete ligation of the IVC leads to reproducible thrombosis, it is not ideal for studying nontrauma-related venous thrombosis because of the absence of blood flow.
The IVC stenosis model reduces blood flow by ∼90% via partial ligation of the IVC, and thrombi form upstream of the site of stenosis because of activation of the endothelium by the disturbed blood flow and possibly because of some vessel injury induced during the surgery.5,6 Thrombi can be detected as early as 3 hours but are usually measured at 48 hours. Von Bruhl et al6 described roles for several different cell types and clotting factors in this model, including platelets, FXII, and neutrophils (see figure). Indeed, the contribution of neutrophils was a particularly novel finding that led to the concept that the innate immune response is involved in this form of thrombosis, and the term “immunothrombosis” was coined. Neutrophils are proposed to enhance immunothrombosis via several mechanisms, including the generation of neutrophil extracellular traps.
A mouse model of spontaneous venous thrombosis was recently described that involves reducing levels of antithrombin and protein C with small interfering RNAs.7 Mice develop thrombi in veins of the head, leg, and liver with fewer thrombi in vessels in the heart, lung, and brain and no thrombi in the kidney or gastrointestinal tract. This pattern of thrombosis is consistent with the notion of differential expression of anticoagulants that regulate tissue-specific hemostasis. The prothrombotic phenotype in the mice develops within 3 days and is fatal. Anticoagulating the mice with the thrombin inhibitor dabigatran etexilate prevents the coagulopathy.7 A selective reduction of antithrombin in mice produced a milder prothrombotic phenotype, whereas reducing protein C alone did not induce thrombosis.7 One unanswered question with the model and in individuals with deficiencies in anticoagulants is, What is triggering the spontaneous thrombosis?
Heestermans et al examined the roles of platelets, neutrophils, and FXII in this new spontaneous venous thrombosis model (see figure). Depletion of platelets prevented the thrombotic coagulopathy and fibrin deposition in the liver, which is consistent with a role of platelets in the IVC stenosis model.5,6 However, depletion of neutrophils did not reduce thrombosis in mice, in contrast to the results with the IVC stenosis model.6 This indicates that neutrophils do not contribute to all forms of venous thrombosis. FXII has been shown to contribute to thrombosis in a variety of mouse models, including the IVC stenosis model.6,8 Surprisingly, Heestermans et al found that decreasing levels of FXII increased thrombosis. How do we explain this result? Heestermans et al explored the possibility that a deficiency of FXII enhances secondary bleeding changes in fibrinolysis and edema without any definitive conclusions. It is possible that reducing levels of FXII leads to destabilization of clots with increased embolization.
In summary, all mouse models have limitations, but it is always important to remember that we are modeling human disease. Venous thrombosis is a complex disease that may be triggered by different risk factors and combinations of risk factors. Further studies are needed to determine the role of neutrophils and other immune cells in different forms of venous thrombosis in mouse models and humans. FXII is an attractive target for anticoagulant drug therapy8 and therefore we need to understand why it was apparently antithrombotic in the mouse model of spontaneous venous thrombosis. It would be very interesting to determine the effect of reducing FXI levels in this spontaneous thrombosis model, given the close relationship between FXII and FXI and results from a recent study showing reduced thrombosis without hemorrhage in patients undergoing total knee arthroplasty.9 Finally, platelets have been shown to play roles in different mouse models of thrombosis, and clinical studies have shown that aspirin reduces the recurrence of venous thrombosis in patients.10 These results suggest that antiplatelet therapy may reduce the incidence of venous thrombosis in patients.
Conflict-of-interest disclosure: The author declares no competing financial interests.