Abstract
Transplant-related mortality (TRM) is the largest limiting factor to successful URD-BMT as curative therapy. Identification of non-HLA genetic factors in either recipients or donors could improve BMT outcomes through better matching at these loci. We performed a genome-wide association study (GWAS), named DISCOVeRY-BMT (Determining the Influence of Susceptibility COnveying Variants Related to one-Year mortality after BMT) of 1-year TRM in 3,532 patients treated for AML, ALL or MDS (recipients) reported to CIBMTR from 2000-2011 and their HLA-matched URD (donors); donor DNA was available for 98% of HCT recipients. Cohort 1 includes 2,240 donor-recipient pairs; patients received a 10/10 HLA URD-BMT from 2000-08. Cohort 2 includes 823 donor-recipient pairs; patients received either a 10/10 HLA URD-BMT from 2009-11 or 8/8 (but <10/10) HLA URD-BMT. Genotyping of recipient and donor DNA was performed using the Illumina HumanOmniExpress-24 BeadChip containing 729,293 single nucleotide polymorphisms (SNPs) at University of Southern California. Due to the small number of non-European individuals, we report analyses of European American recipients only. After quality control, Cohort 1 includes 2,052 donor-recipient pairs and Cohort 2 includes 763 donor-recipient pairs typed at 637,655 SNPs. For each SNP, a shared genotype variable was created to capture donor-recipient allele sharing. The shared genotype was assigned a value of 0 if the recipient and donor have the same genotype at a given SNP and a value of 1 if they differ by 1 or 2 alleles. This idea, that a "matched" genotype can impact risk of TRM, is similar to the idea behind HLA matching. For all survival analyses we accommodated the competing risk of death due to disease; covariates including age, body mass index, and graft type (blood or marrow) were included in all analyses. Analyses of the shared genotype variable with TRM were run for all diseases together and excluding ALL (AML+MDS). P-values for each cohort were combined using METAL software with weights proportional to the square root of the number of cases. We report on results for combined P-value (Pmeta) <5 x 10-8.
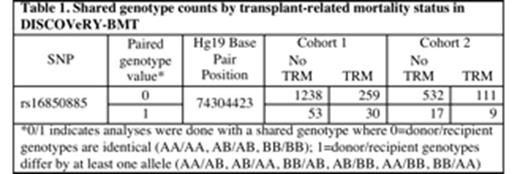
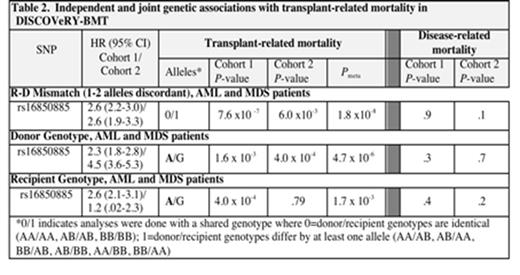
In analyses of AML+MDS donor-recipient pairs four typed SNPs, rs9884653, rs16850885, rs1246576 and rs10014791, spanning 865,040 base pairs in and near MTHFD2L and EPGN on chromosome 4q13.3 were significant at Pmeta <5 x 10-8; several SNPs in this region approach genome wide significance level. The most significant SNP, rs16850885 (P=7.6 x 10-7 in Cohort 1, P=6 x10-3 in Cohort 2, Pmeta =1.8 x 10-8) at 74,304,423 bp, places recipients who differ from their donors by at least one allele (5.2% of patients in Cohort 1 and 3.9% in Cohort 2) at a 2.6 fold increased risk of TRM (Tables 1 and 2). It is in perfect linkage disequilibrium (r2=1) with rs9884653 at 74,280,426 and rs10014791 at 75,145,466; rs1246576 is in strong linkage disequilibrium with rs16850885 (r2 =.91). All four SNPs shows hazard ratios (HRs) of similar magnitude across both cohorts for patients who differ from their donors by at least 1 allele. These SNPs are not associated with death due to disease in either cohort. The difference was not allele specific; of those patients who died of TRM with at least one allele difference approximately 50% of the donors provided the less common A allele (donor A/G or A/A with recipient G/G) and 50% of the recipients had the A allele (recipient A/G with a G/G donor). Analyses of the donors and recipients separately each show some evidence of association with the minor allele A at rs16850885 (Table 2), but it is patients who differ from their donors at this locus who have greatest risk of TRM.
A previous GWAS showed that individuals with at least one copy of the A allele at rs16850885 have lower levels of secreted IL-1β following small pox vaccination compared with those who are homozygous GG (P=7.3 x 10-9). IL-1β, a pro-inflammatory cytokine secreted early in the inflammatory response, may be contributing to the risk of TRM through the combination of initiation and maintenance of host tissue inßammation and donor cell inßammatory response. Our study, DISCOVeRY-BMT, is the largest GWAS of TRM HLA-matched URD-BMT. Further confirmation of these findings in a third cohort may aid in donor selection.
Sucheston-Campbell:NIH/NHLBI: Research Funding. McCarthy:The Binding Site: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Onyx: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees. Hahn:NIH/NHLBI: Research Funding; Novartis: Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal