Abstract
BACKGROUND: The CD20 antigen is an attractive immunotherapy target for B cell non-Hodgkin lymphomas, and adoptive transfer of T cells genetically modified to express a chimeric antigen receptor (CAR) targeting CD20 is a promising strategy. A theoretical limitation of this approach is that residual serum rituximab from prior chemoimmunotherapy regimens might block CAR binding to CD20 and prevent T cell mediated anti-lymphoma responses. However, previous data from our group and others have suggested that CD20 CAR+ T cell function is only partially blocked by anti-CD20 antibody (Ab), and T cell function in the setting of anti-CD3 x anti-CD20 bispecific Ab is not blocked by rituximab levels of up to 100 μg/ml. We have further tested the impact of various concentrations of rituximab on CD20-CAR T cell activity in vitro and in vivo.
METHODS: CD3+ T cells (proliferation and cytokine assays) or CD8+ selected T cells (cytotoxicity assays) were isolated from healthy donors, activated with anti-CD3/CD28 beads, and transduced with epHIV7 lentiviral vectors encoding 2nd or 3rd generation anti-CD20 CAR constructs (Leu16-28-ζ, Leu16-28-BB-ζ, or fully human 1-5-3-NQ-28-BB-ζ). Functional assays, performed using target cells pre-incubated for 30 min. with varying concentrations of rituximab, included a CFSE assay to assess CAR T cell proliferation, Luminex assays for cytokine secretion, and a 5-hour standard 51 chromium release assay for cytotoxicity. Target cells included K562 cells transduced to express CD80 with or without CD20 (denoted "K80" and "K80-20"), Raji, Daudi, Granta, Rec-1, and FL-18 lymphoma cells. K80-20 cell lines expressing low, medium, and high CD20 were established by limiting dilution cloning.
For in vivo experiments, NOD/SCID/γ-/- (NSG) mice were inoculated i.v. with rituximab-resistant Raji-ffLuc lymphoma cells. After 5 days, rituximab was administered i.p. at 25 μg/ml or 200 μg/ml, and then at 24 hours after rituximab administration CAR+ central memory T cells expressing the 1-5-3-NQ-28-BB-ζ vector were injected i.v. Tumor growth was measured with bioluminescence imaging twice weekly and mice were followed for survival.
RESULTS: The availability of CD20 binding sites on Ramos lymphoma cells pre-incubated with various concentrations of rituximab was assessed with flow cytometry, and as expected, we found a dose-dependent blockade of CD20, with complete blockade at 50 μg/ml rituximab at 4°C. However, when anti-CD20-PE was incubated at 37°C, low-level CD20 binding could occur even at 200 μg/ml of rituximab. Despite the low number of available CD20 binding sites after rituximab, proliferation of CFSE-labeled CAR+ T cells was largely unimpaired in rituximab concentrations up to 200 μg/ml. In contrast, cytokine secretion was impaired in a dose-dependent manner, although even at 100 μg/ml of rituximab, interferon-γ, interleukin-2, and tumor necrosis factor a were still produced at 34-51%, 70-92%, and 79-108% of baseline levels, respectively. Cytotoxicity also decreased with increasing rituximab concentration but >75% of baseline cytolytic activity was retained at 100 μg/ml. We hypothesized that the level of CD20 expression on target cell lines might impact sensitivity to rituximab blockade. Using K80-20 cells with low, medium, or high CD20 expression we found that cytokine secretion and cytotoxicity (but not proliferation) were highly impaired upon stimulation with CD20low target cells, whereas T cell function remained completely intact when CD20high cells were used as targets.
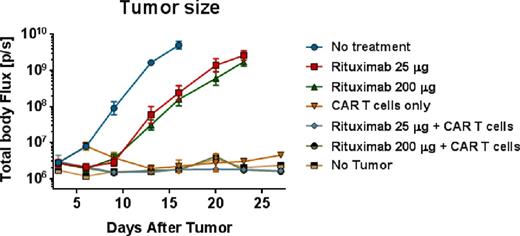
In vivo, mice bearing rituximab-refractory Raji-ffLuc tumors experienced only slight delay of tumor growth when treated with either low or high doses of rituximab, and mice treated with T cells alone had significant clearance of tumor. In mice that received low or high-dose rituximab prior to T cell infusions, tumor rejection and survival prolongation were equivalent to or better than that observed with mice receiving T cells alone (see figure).
CONCLUSION: We have shown that the in vitro and in vivo activity of CD20-targeted CAR T cells is minimally impacted after rituximab, despite a low number of available CD20 binding sites. These data suggest that residual serum rituximab levels will not present a significant impediment to CD20-targeted CAR therapy in patients who have received rituximab-containing chemotherapy regimens.
Jensen:Juno Therapeutics: Equity Ownership, Patents & Royalties, Research Funding. Gopal:Merck: Research Funding; BioMarin: Research Funding; Seattle Genetics: Consultancy, Honoraria; Gilead: Consultancy, Research Funding; Spectrum: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Piramal: Research Funding; Millenium: Honoraria, Research Funding; BMS: Research Funding; Janssen: Consultancy; Emergent/Abbott: Research Funding; Sanofi-Aventis: Honoraria. Riddell:Juno Therapeutics: Equity Ownership, Patents & Royalties, Research Funding; Cell Medica: Membership on an entity's Board of Directors or advisory committees; Adaptive Biotechnologies: Consultancy. Till:Pfizer: Research Funding; Roche/Genentech: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal