Abstract
Introduction: Outcomes of most acute myeloid leukemia (AML) patients (pts) remain poor. Enhanced risk-stratification and individualized treatment approaches are urgently needed. The Hedgehog (Hh) signaling pathway is important in embryonic development & stem cell biology & converges into activation of the transcription factor GLI1. In mice, loss of Gli1 increased the number of quiescent long-term hematopoietic stem cells with enhanced engraftment capacities & impaired myeloid development. Nevertheless, the biological role & prognostic impact of GLI1 expression in human AML remains to be fully elucidated.
Non-myeloablative conditioning regimens (NMA) are increasingly used in AML pts undergoing hematopoietic stem cell transplantation (HCT) ineligible for conventional conditioning. In NMA-HCT the therapeutic approach is mainly based on an immunological graft-versus-leukemia (GvL) effect. Here, we tested whether the expression of GLI1 is associated with outcome in AML pts undergoing NMA-HCT.
Patients & Methods: We analyzed 135 pts with diagnostic bone marrow (BM) available who were treated at our institution between January 2000 & June 2012. Median age at diagnosis was 64 years (y; range 38-75y). We included pts with de novo (n=84; 62.2%), secondary (n=37; 27.4%) or therapy-related (n=14; 10.4%) AML. All pts received NMA-HCT consisting of fludarabine (30mg/m² at days -4 to -2) & 2Gy total body irradiation (day 0). All pts received granulocyte colony stimulating factor (G-CSF)-mobilized peripheral blood stem cells on day 0. Donors were HLA-matched related (n=22; 16.3%) or HLA-matched (n=80; 59.3%) or mismatched (≥1 antigen; n=33; 24.4%) unrelated. Median follow-up was 4.3 y for pts alive.
At diagnosis, cytogenetics were determined using standard techniques for banding & fluorescence in-situ hybridization in BM of all pts. The presence of FLT3 -ITD, FLT3 -TKD & expression status of EVI1, BAALC, ERG, MN1, mir-9 & mir-181a as well as the mutation status of the NPM1, CEBPA, IDH1, IDH2 & DNMT3A genes were determined. All mononuclear cells in pts' BM were assessed for presence of CD7, CD14, CD34, CD38, CD34+/CD38-, CD45, CD56, CD117, CD56 & glycophorin A. GLI1 expression was measured by a Taqman-probe based quantitative reverse transcription polymerase chain reaction & normalized to 18S as internal control. The third quartile of the normalized gene expression was used to define high & low GLI1 expressers.
Results: At diagnosis, pts with high GLI1 expression had increased % of blasts in BM (p <.01) & in peripheral blood by trend (p =.10) & were more likely to have de novo AML by trend (p =.06). Pts with high GLI1 expression were less likely to have a monosomal karyotype by trend (p =.07) & high GLI1 expression associated with higher % of CD14 (p =.05) & lower % of CD45 (p =.01) positive cells. High GLI1 expressers more frequently had IDH2 gene mutations (p =.01) & were lower EVI1 expressers (p =.05). High GLI1 expression was associated with significantly increased MN1 (p <.01), BAALC (p =.02) & ERG (p <.01) expression as well as lower expression of mir-9 (p =.01) & mir-181a (p <.01).
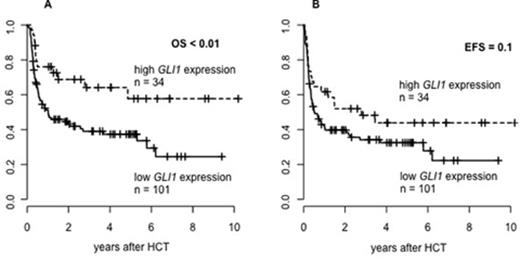
High GLI1 expressers also had a significantly longer overall survival (OS; p <.01, Figure 1A) & longer event-free survival (EFS; p =.10; Figure 1B) by trend. In multivariable analysis a high GLI1 expression associated with longer OS (Hazard Ratio 0.42; 95% Confidence Interval 0.23-0.79; p <.01). None of the analyzed clinical or biological parameters were significantly associated with EFS in multivariable analysis.
Conclusion: High GLI1 expression associated with distinct clinical and biological characteristics. The observation that elevated GLI1 expression was linked to IDH2 mutations suggests that - similar to glioblastoma - IDH mutations may be associated with an activated hedgehog signaling pathway in AML. Moreover, we found that high GLI1 expression was an independent prognostic factor for longer OS in AML pts receiving NMA-HCT. An effect that may be mediated by a decreased number of quiescent long-term AML stem cells in high GLI1 expressers, which may be especially beneficial in NMA-HCT treated AML pts. Further mechanistic studies are needed to explore the observed results biologically & clinical validation studies are necessary to confirm our finding of an improved outcome of high GLI1 expressers in AML receiving NMA-HCT.
Franke:Novartis: Other: Travel Costs; MSD: Other: Travel Costs; BMS: Honoraria. Niederwieser:Novartis: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal