Abstract
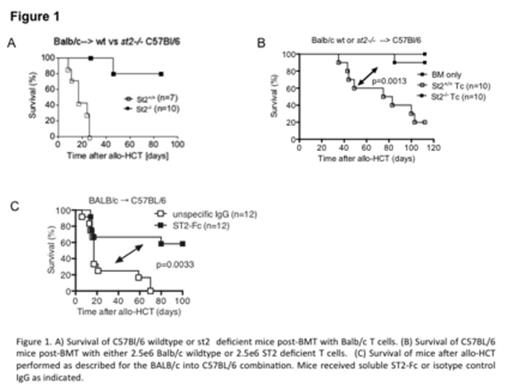
The IL-1 superfamily member IL-33 is produced in barrier tissues. IL-33 binds to the receptor suppression of tumorigenicity 2 (ST2), expressed on stromal cells, regulatory T cells (Tregs), myeloid derived suppressor cells (MDSCs), and macrophages. IL-33 has both anti-inflammatory and pro-inflammatory properties. It is not known if IL-33 plays a role in acute GvHD, and if so what properties it exerts. By immunohistochemistry staining of gut tissues, IL-33 production by non-hematopoietic cells was increased in mice post-conditioning and in patients during GvHD. To determine whether IL-33 could augment GvHD via a host signaling mechanism, we compared st2-/-to wildtype (wt) hosts and observed decreased GvHD lethality (Figure 1A). Additionally, IL-33-/- versus wt hosts had a marked decrease in GvHD lethality and reduced TNFα production. Conversely, IL-33 administration during the peak inflammatory response worsened GvHD. Previous studies have shown increased levels of the soluble form of ST2 (sST2) are a biomarker for steroid-refractory GvHD (Vander Lugt, NEJM, 2013). We hypothesized that sST2 acted not only as an indicator of tissue injury and biomarker of GvHD but also as an immune modulator during GvHD. In rodents, we found that ST2 was upregulated on alloreactive T cells and sST2 increased as GvHD progressed. St2-/-versus wt donor T cells had a marked reduction in GvHD lethality (Figure 1B) without compromise of graft-vs-leukemia responses. Comparable data was seen in 2 different strain combinations. Alloantigen-induced IL-18 receptor upregulation was significantly lower in the absence of ST2, which was linked to significantly reduced IFNγ production by st2-/- vs wt CD4 and CD8 T cells during GvHD. Similarly, sST2 transgenic hosts and wt recipients given exogenous sST2-Fc fusion protein infusions (Figure 1C) to block ST2/IL-33 interaction each had significantly reduced GVHD lethality, establishing the functional role of ST2 as a decoy receptor modulating GVHD. During the peak of the GvHD inflammatory response, IL-33 signalling of either donor or host cells promoted activation of donor T cells, while the use of exogenous sST2-Fc protein to prevent IL33/ST2 engagement ameliorates disease. Together, these studies point to targeting of the IL-33/ST2 axis as a novel and potent target for GvHD therapy.
Warncke:Novartis Pharma AG: Employment. Junt:Novartis Pharma AG: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal