Abstract
Background: Polycythemia vera (PV) is a myeloproliferative neoplasm driven by JAK/STAT activation and is associated with erythrocytosis and a broad symptom burden that may negatively impact patient quality of life (QoL). Hydroxyurea (HU) is often used as first-line therapy for high-risk patients but may not effectively control or reduce symptom burden. RESPONSE is a phase III trial comparing ruxolitinib (RUX) with best available therapy (BAT) in patients with PV who were intolerant of or resistant to HU according to modified European LeukemiaNet (ELN) criteria. The primary study endpoint (a composite of hematocrit control and ≥35% spleen volume reduction at Week 32) was achieved by 21% of patients in the RUX arm vs 1% in the BAT arm (P<0.0001); 77% of patients in the RUX arm achieved at least one component of the primary endpoint. The current analysis was conducted to evaluate the effect of RUX on PV-related symptoms and QoL measures in the RESPONSE trial.
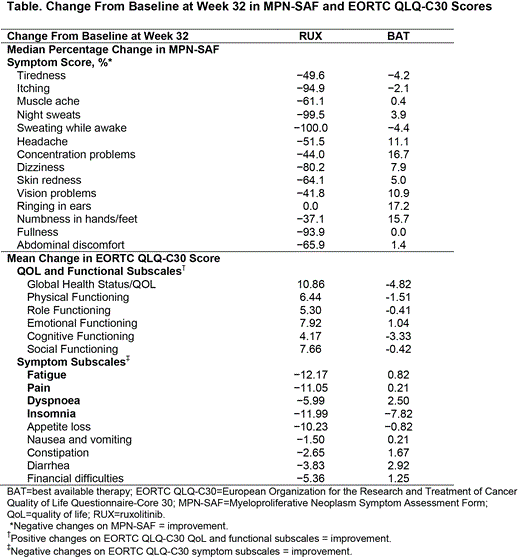
Methods: Patients with PV aged ≥18 years, resistant to or intolerant of HU (modified ELN criteria) with splenomegaly, and who required phlebotomy for hematocrit control were randomized 1:1 to receive open-label RUX 10 mg twice daily (BID) or BAT (administered based on investigator judgment). Dose adjustments were permitted (RUX, 5-mg BID increments [25 mg BID max]; BAT was adjusted per investigator judgment). Objectives of this analysis included assessment of improvement in symptom burden as assessed by patient-reported outcomes using the 14-item modified Myeloproliferative Neoplasm Symptom Assessment Form (MPN-SAF), the Pruritus Symptom Impact Scale (PSIS), the European Organization for the Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 (EORTC QLQ-C30), and the Patient Global Impression of Change. The 14-item MPN-SAF (graded from 0 [absent] to 10 [worst imaginable]) comprised symptoms related to cytokines (tiredness, itching, muscle ache, night sweats, and sweats while awake), hyperviscosity (vision problems, dizziness, concentration problems, headache, numbness/tingling, ringing in ears, and skin redness), and splenomegaly (abdominal discomfort and early satiety). Changes in total symptom score (TSS; maximum score = 140) and individual symptom scores from baseline to Week 32 were summarized by treatment group. For the EORTC QLQ-C30 Global Health Status/QOL score, the percentage of patients with a minimally important difference (MID) from baseline (10-point change) at Week 32 was summarized.
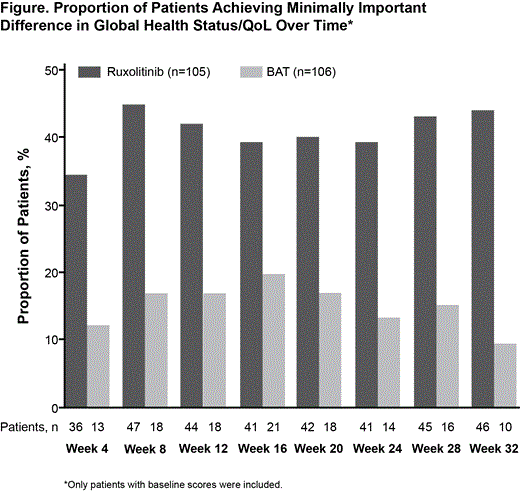
Results: Overall, 222 patients were randomized (RUX, 110; BAT, 112). Median age (range) was similar between arms (RUX, 62.0 [34.0–90.0]; BAT, 60.0 [33.0–84.0]); the RUX and BAT arms were 60% and 71% male, respectively. At Week 32, a higher proportion of patients in the RUX vs the BAT arms had a ≥50% improvement in MPN-SAF TSS (49% vs 5%, respectively) and MPN-SAF symptom cluster scores (cytokine, 64% vs 11%; hyperviscosity, 37% vs 13%; splenomegaly, 62% vs 17%). Median percentage changes in individual symptom scores are presented in the Table. Mean changes from baseline at Week 32 on the 5 items of the PSIS indicated that the severity of pruritus and its interference on daily life improved with RUX (range, −1.5 to −2.2) and was unchanged/worsened with BAT (range, −0.1 to 0.3). Treatment with RUX vs BAT was associated with improved mean changes from baseline at Week 32 on EORTC QLQ-C30 symptom subscales, functional subscales, and Global Health Status/QOL (Table); 46% of RUX patients versus 10% of BAT patients achieved an MID in Global Health Status/QOL (Figure). At Week 32, RUX patients were more likely to rate their global impression of symptom changes as “very much improved” or “much improved” (67%) vs BAT patients (13%).
Conclusion: In patients with PV who were resistant to or intolerant of HU, treatment with RUX was associated with greater and clinically meaningful improvements in PV-related symptom burden and QoL measures compared with BAT.
Mesa:Incyte Corporation: Research Funding; CTI: Research Funding; Gilead: Research Funding; Genentech: Research Funding; Eli Lilly: Research Funding; Promedior: Research Funding; NS Pharma: Research Funding; Sanofi: Research Funding; Celgene: Research Funding. Off Label Use: Ruxolitinib is a JAK1/JAK2 inhibitor approved for the treatment of patients with intermediate or high-risk myelofibrosis, including primary myelofibrosis, post polycythemia vera myelofibrosis, and post-essential thrombocythemia myelofibrosis. Verstovsek:Incyte Corporation: Research Funding. Kiladjian:Novartis Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Masszi:Novartis Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Durrant:Novartis Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Harrison:Novartis Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. He:Incyte Corporation: Employment, Equity Ownership. Jones:Incyte Corporation: Employment, Equity Ownership. Parasuraman:Incyte Corporation: Employment, Equity Ownership. Li:Novartis Pharmaceuticals: Employment, Equity Ownership. Côté:Novartis Pharmaceuticals: Employment, Equity Ownership. Habr:Novartis Pharmaceuticals: Employment, Equity Ownership. Vannucchi:Novartis Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal