Abstract
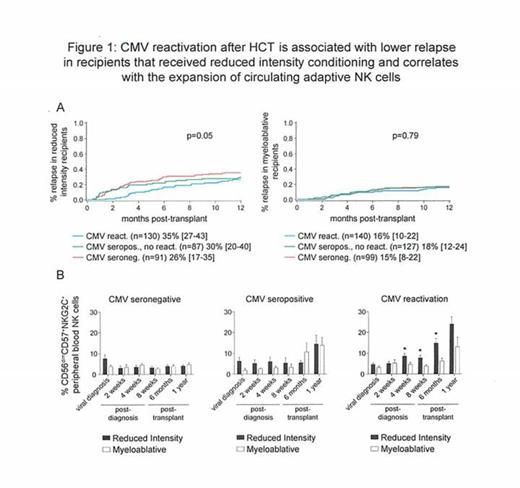
We have previously shown that while NK cells are the first lymphocyte reconstituting after allogeneic transplantation, these NK cells are immature and functionally hyporesponsive when exposed to tumor targets. Remarkably, this can be reversed by reactivation of human cytomegalovirus (CMV). A distinct population of natural killer (NK) cells with attributes of immunological memory arises specifically in response to acute CMV infection in healthy individuals or reactivation of latent CMV in hematopoietic cell transplantation (HCT) recipients. These adaptive NK cells are defined by expression of the activating receptor NKG2C and maturation marker CD57 and display a heightened capacity for inflammatory cytokine production and cytolysis in response to target cell recognition. Since the differentiation and maturation of NK cells is dependent upon interactions with cells of the myeloid lineage, including antigen-presenting cells, we hypothesized that the expansion of adaptive NK cells could influence relapse risk, particularly after a reduced intensity conditioning (RIC) regimen where residual host myeloid cells may intensify donor adaptive NK cell expansion in response to CMV reactivation after HCT. Thus, we analyzed 1-year relapse rates in 674 allogeneic HCT recipients at the University of Minnesota (470 umbilical cord blood (UCB) and 204 matched sibling donor (MSD)) between 2001-2013 with AML (n=313), ALL (n=187), MDS (n=85), CML (n=28), NHL (n=42), Hodgkins (n=14) and Multiple Myeloma (n=5) segregated by recipient CMV serostatus (270 seronegative and 404 seropositive). Within the CMV seropositive group, 190 individuals experienced CMV reactivation. We observed no association between CMV reactivation and 1-year relapse rates in recipients of myeloablative conditioning (n=366; 16% [10%-22%] for CMV seronegative vs. 18% [12-24%] for CMV seropositive without CMV reactivation vs. 15% [8%-22%] for CMV seropositive recipients with CMV reactivation). In marked contrast, CMV reactivation was associated with lower 1-year relapse rates following RIC (n=308; 35% [27%-43%] for CMV seronegative vs. 30% [20-40%] for CMV seropositive without CMV reactivation vs. 26% [17%-35%] for CMV seropositive with CMV reactivation [p for trend=0.05, Figure 1A]). In multivariate analyses adjusting for recipient CMV status, conditioning regimen, donor type (MSD vs. UCB), diagnosis (AML [shown to be sensitive to NK cell effects] vs. other), gender, disease risk (standard vs. high) and prior autologous transplant, CMV reactivation was independently associated with lower relapse risk (RR=0.6 [0.4-1.0], p=0.06) and increased DFS (RR=0.7 [0.5-1.0], p=0.05) following RIC, but not myeloablative HCT. Although CMV reactivation led to superior DFS within the entire cohort (RR=0.7 [0.6-1.0], p=0.04), CMV reactivation was not independently associated with lower relapse risk (RR=1.0 [0.5-1.8], p=0.88) with myeloablative conditioning. To determine whether the preparative regimen also influenced adaptive NK cell differentiation post-transplant, we analyzed the percentage of CD56dimCD57+NKG2C+ NK cells. Compared to myeloablative recipients, after RIC, the percentage of circulating CD56dimCD57+NKG2C+ NK cells was significantly higher at 4 weeks post-CMV reactivation (8.58% vs. 4.64%, p=0.02), 8 weeks post-CMV reactivation (7.77% vs. 3.84%, p=0.02), and 6 months post-transplant (14.80% vs. 6.29%, p=0.01) (Figure 1B). Our results demonstrate a relationship between CMV reactivation and enhanced graft-versus-leukemia (GVL) in patients treated with RIC. The association between lower relapse risk and in vivo expansion and survival of adaptive NK cells suggests that these cells may be critical to reducing the risk of relapse. Strategies to isolate and expand adaptive NK cells in order to enhance the GVL effect without clinical CMV reactivation are being actively explored.
Miller:Coronado: Speakers Bureau; BioSciences: Membership on an entity's Board of Directors or advisory committees; SAB: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal