Abstract
Rationale:
Overcoming proteasome inhibitor (PI) resistance is a challenge in multiple myeloma (MM) therapy since most MM patients ultimately develop PI resistance. Induction of excessive activation of the unfolded protein response (UPR) is the major mechanism of PI-induced cytotoxicity in MM. The UPR is a complex transcriptional response that balances biosynthesis, folding and proteasomal destruction of cellular protein. UPR inactivation results in PI resistance in vitro, and MM cells with low UPR activation accumulate and drive the relapse in PI-resistant MM patients. Pharmacologic activation of the UPR overcomes PI-resistance in preclinical models of MM and provides an option for clinical testing. The HIV protease inhibitor nelfinavir (NFV) has UPR-inducing activity via an unknown mechanism that may involve interference with regulatory proteases in the UPR and/or proteasome activity. NFV has single agent activity in MM and sensitizes MM and AML cells for PI treatment in vitro and in vivo.
Methods:
We performed a multicenter phase I dose escalation study to assess safety and recommended dose for phase II of NFV in combination with bortezomib (BTZ) in patients with advanced hematologic malignancies, and to detect signals for activity. NFV was given d 1-14 twice daily p.o. at the dose levels 1250 mg (DL0), 1875 mg (DL1) and 2500 mg (DL2), BTZ was dosed 1.3 mg/m2 d 1, 4, 8, 11 i.v. in 21 day cycles. The first treatment cycle was preceded by one week of NFV monotherapy for assessment of pharmacokinetic/pharmacodynamic parameters (NFV plasma concentrations, proteasome activity and expression of UPR-related proteins in peripheral blood mononuclear cells (PBMC)). Patients were treated for 3 cycles per protocol with the option to receive up to a total of 7 cycles.
Results:
12 patients were treated in the dose escalation cohort (median age 58 years; 8 patients with MM, 1 each with ALL, AML, DLBCL, MCL) for an average of 2.6 cycles. All MM patients had received prior BTZ. DLT was determined in cycle 1 in which 93 % of planned dose was delivered. One DLT was observed (G4 ALT elevation at DL2 that spontaneously resolved). Toxicity was mostly mild, could be handled symptomatically, and did not lead to study drug discontinuation except for one case of thrombocytopenia. Diarrhoea G1-2 was the most frequent toxicity observed. Ten patients were evaluable for best response while on trial therapy after having received at least one full cycle. Of these, three patients achieved a PR (1 MCL, 2 MM), 4 remained in SD for at least 2 cycles (2 MM, 1 AML, 1 ALL), while 3 progressed (2 MM, 1 DLBCL). Peak NFV plasma concentrations during monotherapy were in the dose range putatively required for UPR activation, tended to be higher in patients treated at DL1, compared to DL2 (means 13.3 vs. 8.9 mM, p=0.08) and were significantly higher during NFV monotherapy than during combination therapy with BTZ (means 9.24 vs. 6.60 mM, p=0.04), suggesting induction of NFV clearance either by autoinduction, concomitant BTZ application, or both. Pharmacodynamic analysis revealed upregulation of proteins related to UPR-induced apoptosis by NFV monotherapy in PBMC (CHOP +56%, p=0.008; PARP +57%, p=0.04, n=10). Activity of the BTZ-insensitive proteasome b2 subunit in PBMC decreased (-16%, p=0.01) during NFV monotherapy, compared to baseline, as did the BTZ-sensitive b1/b5 subunit (-17%, p=0.001).
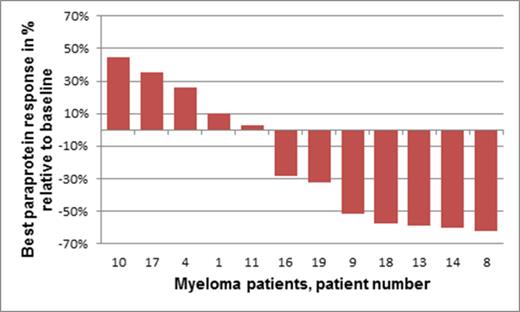
To detect additional signals for activity, an extension cohort of 6 heavily pretreated MM patients that had shown BTZ-resistance during the past 12 months and were in addition lenalidomide-resistant was treated at the recommended dose (DL2). Three of these patients achieved a PR and 2 a MR, while 1 showed PD with a mean of 4.3 cycles administered. Overall, 12 MM patients could be evaluated for best response while on therapy with BTZ + NFV in this study, of which 5 achieved a paraprotein reduction of > 50% compared to baseline (figure 1).
Conclusion:
Nelfinavir 2500 mg p.o. twice daily induces UPR activation and proteasome inhibition. It can safely be combined with bortezomib (1.3 mg/m2 d 1, 4, 8, 11) to potentially increase bortezomib sensitivity of hematologic malignancies. The combination yields promising clinical activity signals in patients with bortezomib-resistant myeloma.
Best paraprotein response, relative to baseline, of evaluable patients with relapsed-refractory myeloma treated with bortezomib + nelfinavir at any dose level for at least one full cycle.
Best paraprotein response, relative to baseline, of evaluable patients with relapsed-refractory myeloma treated with bortezomib + nelfinavir at any dose level for at least one full cycle.
Off Label Use: the presentation will include off label use of nelfinavir as investigational medicinal product (IMP). Hitz:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal