Abstract
Introduction: XPO1 (CRM1, Exportin 1) is the sole transporter of many tumor suppressor proteins (including MYC, BCL2, BCL6, BTK, IkB) and is elevated in non-Hodgkin Lymphoma. Selinexor (Sel, KPT-330) is an oral covalent inhibitor of XPO1, the first clinical molecule of the Selective Inhibitors of Nuclear Export drug class. The phase I clinical trial of Sel in hematologic malignancies showed promising early single-agent efficacy with modest toxicity in relapsed Diffuse Large B-cell Lymphoma (DLBCL, Gutierrez et al, ASCO 2014). DLBCL, the most common lymphoid malignancy, is currently cured in only 10% of relapsed patients, and consists of 2 subtypes based on putative cell of origin (COO): activated B-cell (ABC) and germinal center B-cell (GCB). We performed preclinical studies of Sel, modeling its single-agent efficacy in frontline and relapsed DLBCL and its potential synergy with other clinically relevant therapeutics.
Methods: To model drug resistant DLBCL, resistant subpopulations of 12 patient-derived DLBCL cell lines were created by in vitro intermittent exposure to active congeners of cyclophosphamide, doxorubicin, and vincristine (ivCHOP), approximating clinical practice. To determine if CHOP-resistant DLBCL is also resistant to other agents, we determined single-agent dose response curves and IC50 values for both parental and ivCHOP resistant (CHOP-res) subclones of 4 of these lines at submission (HBL1 & TMD8 of ABC subtype, OCI-Ly7 & HT of GCB subtype, with 8 lines in progress) with Sel, chemotherapy (CT, ivCHOP, DHAP, and ICE), and targeted therapy (TT, ibrutinib, ABT-199, idelalisib, everolimus, MLN0128, alisertib, lenalidomide, bortezomib, I-BET151, and ONC201). Viability was assessed with CellTiter-Glo (Promega) after a 3 day cell culture. IC50 values were determined using GraphPad Prism. Based on these results, we evaluated the ability of Sel to synergize with other agents or restore sensitivity in CHOP-res with a combination “checkerboard” (orthogonal dose titration for each drug). The Combination Index (CI) for pairs at all concentrations was calculated with ComboSyn, with CI values <1 indicating synergy. Gene expression profiling with Illumina HT12v4 arrays will compare parental and CHOP-res of 12 DLBCL lines.
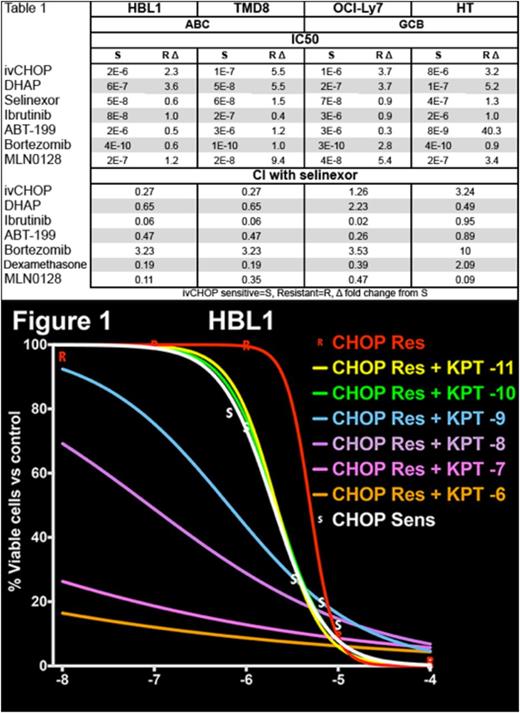
Results: All CHOP-res lines of both COO types had higher IC50 for both ivCHOP (mean, 3.7x) and DHAP (4.5x) as compared to parental cells (Table 1). In contrast, the IC50 of Sel is unchanged between parental and CHOP-res, for both COO types. Other targeted agents displayed variable activity between parental and CHOP-res and between COO types, with the IC50 of ibrutinib being nearly 2 log lower in ABC lines. CI values showed that Sel was generally a strong synergizer (Table 1), especially with TT and in ABC lines. Sel had lower CI values with CT, but restored sensitivity to ivCHOP in HBL1 (Figure 1). Bortezomib and Sel were moderately antagonistic, although further tests are ongoing. Gene expression profiling, comparing parental vs. CHOP-res and Sel synergizing vs. non-synergizing lines, is ongoing.
Conclusions: Our data suggest that Sel: 1) is equally active, and thus not cross-resistant, in cell lines made resistant to standard chemotherapeutics, 2) is a potent, broadly active synergizer with targeted therapy against lines modeling relapsed DLBCL, and 3) has greater synergy in ABC DLBCL, in which it may be able to reverse acquired resistance to frontline therapy. This behavior fits with the broad effects of XPO1 inhibition. The cross-resistance of CHOP-res lines to DHAP models clinical outcomes, and re-sensitization of CHOP-res lines with Sel suggests the potential for relapsed and frontline clinical trials. Further work with our model may discover more synergies of Sel, suggesting future clinical combinations and biomarkers associated with response.
| . | HBL1 . | TMD8 . | OCI-Ly7 . | HT . | ||||
|---|---|---|---|---|---|---|---|---|
| ABC | GCB | |||||||
| IC50 | ||||||||
| S | R Δ | S | R Δ | S | R Δ | S | R Δ | |
| ivCHOP | 2E-6 | 2.3 | 1E-7 | 5.5 | 1E-6 | 3.7 | 8E-6 | 3.2 |
| DHAP | 6E-7 | 3.6 | 5E-8 | 5.5 | 2E-7 | 3.7 | 1E-7 | 5.2 |
| Selinexor | 5E-8 | 0.6 | 6E-8 | 1.5 | 7E-8 | 0.9 | 4E-7 | 1.3 |
| Ibrutinib | 8E-8 | 1.0 | 2E-7 | 0.4 | 3E-6 | 0.9 | 2E-6 | 1.0 |
| ABT-199 | 2E-6 | 0.5 | 3E-6 | 1.2 | 3E-6 | 0.3 | 8E-9 | 40.3 |
| Bortezomib | 4E-10 | 0.6 | 1E-10 | 1.0 | 3E-10 | 2.8 | 4E-10 | 0.9 |
| MLN0128 | 2E-7 | 1.2 | 2E-8 | 9.4 | 4E-8 | 5.4 | 2E-7 | 3.4 |
| CI with selinexor | ||||||||
| ivCHOP | 0.27 | 0.27 | 1.26 | 3.24 | ||||
| DHAP | 0.65 | 0.65 | 2.23 | 0.49 | ||||
| Ibrutinib | 0.06 | 0.06 | 0.02 | 0.95 | ||||
| ABT-199 | 0.47 | 0.47 | 0.26 | 0.89 | ||||
| Bortezomib | 3.23 | 3.23 | 3.53 | 10 | ||||
| Dexamethasone | 0.19 | 0.19 | 0.39 | 2.09 | ||||
| MLN0128 | 0.11 | 0.35 | 0.47 | 0.09 | ||||
| ivCHOP sensitive=S, Resistant=R, Δ fold change from S to R | ||||||||
| . | HBL1 . | TMD8 . | OCI-Ly7 . | HT . | ||||
|---|---|---|---|---|---|---|---|---|
| ABC | GCB | |||||||
| IC50 | ||||||||
| S | R Δ | S | R Δ | S | R Δ | S | R Δ | |
| ivCHOP | 2E-6 | 2.3 | 1E-7 | 5.5 | 1E-6 | 3.7 | 8E-6 | 3.2 |
| DHAP | 6E-7 | 3.6 | 5E-8 | 5.5 | 2E-7 | 3.7 | 1E-7 | 5.2 |
| Selinexor | 5E-8 | 0.6 | 6E-8 | 1.5 | 7E-8 | 0.9 | 4E-7 | 1.3 |
| Ibrutinib | 8E-8 | 1.0 | 2E-7 | 0.4 | 3E-6 | 0.9 | 2E-6 | 1.0 |
| ABT-199 | 2E-6 | 0.5 | 3E-6 | 1.2 | 3E-6 | 0.3 | 8E-9 | 40.3 |
| Bortezomib | 4E-10 | 0.6 | 1E-10 | 1.0 | 3E-10 | 2.8 | 4E-10 | 0.9 |
| MLN0128 | 2E-7 | 1.2 | 2E-8 | 9.4 | 4E-8 | 5.4 | 2E-7 | 3.4 |
| CI with selinexor | ||||||||
| ivCHOP | 0.27 | 0.27 | 1.26 | 3.24 | ||||
| DHAP | 0.65 | 0.65 | 2.23 | 0.49 | ||||
| Ibrutinib | 0.06 | 0.06 | 0.02 | 0.95 | ||||
| ABT-199 | 0.47 | 0.47 | 0.26 | 0.89 | ||||
| Bortezomib | 3.23 | 3.23 | 3.53 | 10 | ||||
| Dexamethasone | 0.19 | 0.19 | 0.39 | 2.09 | ||||
| MLN0128 | 0.11 | 0.35 | 0.47 | 0.09 | ||||
| ivCHOP sensitive=S, Resistant=R, Δ fold change from S to R | ||||||||
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal