Abstract
Sickle Cell Disease (SCD) is an inherited disorder of the β-globin chain of hemoglobin, in which a single point mutation leads to decreased deformability of red blood cells (RBCs) and increased cellular adhesion to endothelium. The effect of this mutation on RBCs has been well characterized, and the interplay of endothelial cells, RBCs, and white blood cells (WBCs) have also been well characterized. However, few studies have specifically investigated how platelets interact with endothelial cells and other blood cells in the context of SCD and the role these cell fragments may have in vaso-occlusion.
To that end, we utilized microfluidic technology previously developed in our lab to perform a real time in vitro analyses of platelet-endothelial cell interactions in SCD patient samples. This microvasculature-on-a-chip enables the visualization of blood cell-endothelial cell interactions under a controlled hemodynamic environment (Tsai et al, JCI, 2012).
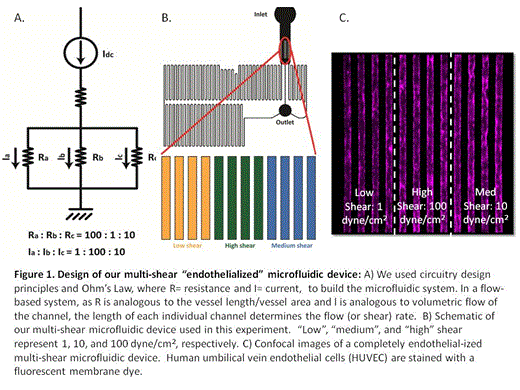
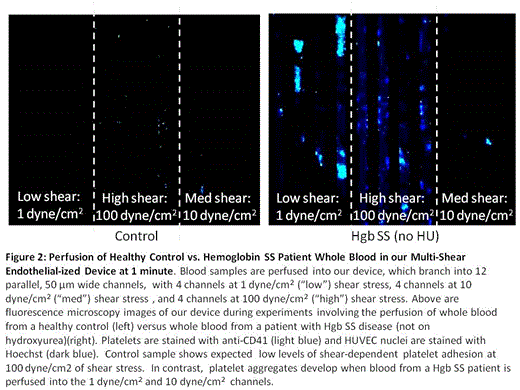
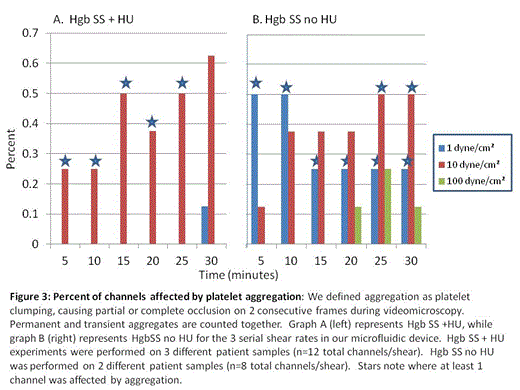
As shear stress can trigger platelet activation, we further modified and optimized our standard microfluidic devices to encompass 3 different physiologic shear rates. Our device features microchannels 50µm in diameter with human umbilical vein endothelial cells (HUVEC) confluently lining the channels; there are 12 channels in each device, grouped in 3 sets of 4 channels with graduating shear rates spanning 3 orders of magnitude (Figure 1). Our initial experiments were performed under normoxic conditions allowing characterization of platelet-endothelial interactions in an arterial in vitro environment. Whole blood samples were obtained from 3 patient populations: patients with HgbSS SCD on hydroxyurea (HgbSS+HU), patients with HgbSS SCD not on hydroxyurea (HgbSS-no HU), and normal healthy controls. Over 30 minutes, whole blood stained with fluorescently labeled CD41 to identify platelets and Hoeschst to identify HUVEC nuclei was perfused at a rate of 1.5µl/minute under videomicroscopy. Accumulation of platelets on the endothelialized channels and platelet aggregates were quantified based on anti-CD41 fluorescence. Within 1 minute of perfusion, HgbSS-no HU whole blood samples exhibited extensive platelet aggregates at 1 and 10 dyne/cm2 (Figure 2); this phenomenon did not occur under any of the shear conditions in blood samples from Hgb SS+HU or healthy control samples. In HgbSS-no HU blood samples, some of these thrombi-like aggregates were stable under flow, increased in size, and persisted for the remainder of the 30 minute experiments. In contrast, mild, uniform, platelet adhesion slowly developed at high shear conditions in Hgb SS+HU with fewer platelet aggregates forming as compared to patients with HgbSS- no HU. Healthy control samples did not exhibit this platelet aggregation. There appears to be an attenuating effect of hydroxyurea on platelets that prevents platelet clumping from occuring as frequently under various shear conditions that is not present in the Hgb SS-no HU samples (Figure 3).
In conclusion, using our novel in vitro system, we have demonstrated the platelets from Hgb SS-no HU patients have a significantly increased propensity to adhere, aggregate, and accumulate in endothelialized microvasculature-sized microchannels. Interestingly, this effect appears to be attenuated in blood samples from Hgb SS+HU patients and not present in healthy controls, demonstrating that hydroxyurea appears to be an important modifier of this phenomenon. Experiments investigating the underlying mechanisms of this phenomenon, the effects of deoxygenation and the potential role of platelets in vaso-occlusion, the effects of sickle cell platelet adhesion/aggregation on endothelial function, and how hydroxyurea may or may not affect any or all of these parameters, are all currently ongoing.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal