Abstract
Background: IL-1β secretion is tightly controlled at the transcriptional and post-translational levels. The NLRP3 inflammasome, a multiprotein complex composed of NOD-like receptor-containing pyrin 3 (NLRP3), apoptosis-associated speck-like protein (ASC) and pro-caspase-1, plays a critical role in IL-1β maturation at the latter stage. NLRP3 expression is a limiting factor for inflammasome activation, and therefore, negative regulation of this factor is necessary to control excessive IL-1β production during sepsis. Previously, we showed that the transcriptional repressor Gfi1 inhibits pro-IL-1β transcription and the IL-1β level is significantly higher in serum of LPS-treated Gfi1-/- than wild-type (WT) mice. The present study revealed that Gfi1 regulates IL-1β secretion through inhibiting NLRP3 inflammasome activation in macrophages.
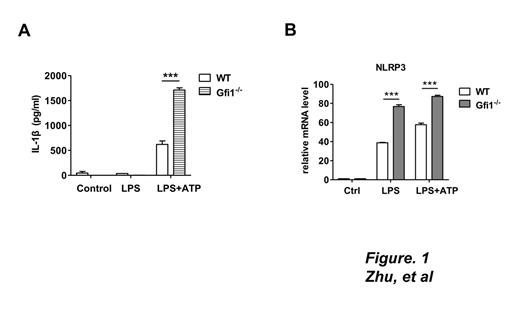
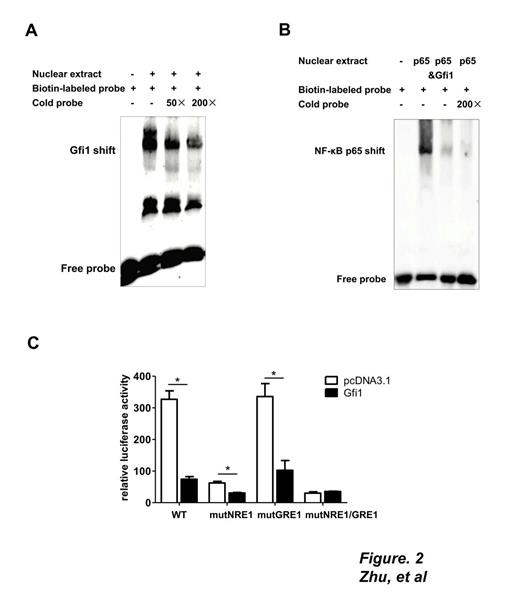
Methods and Results: Bone marrow-derived macrophages (BMDM) from WT and Gfi1-/- mice were primed with LPS and stimulated with ATP. Compared with WT cells, those lacking Gfi1 induced a significant increase in IL-1β in the culture medium (Figure 1A). Western blot disclosed moderate elevation of pro-IL-1β, along with a more dramatic increase in mature IL-1β and cleaved caspase-1 in Gfi1-deficient BMDMs, suggesting that Gfi1 inhibits IL-1β maturation. Consistently, real-time PCR findings showed increased NLRP3 mRNA in Gfi1-deficient macrophages (Figure 1B), implying that Gfi1 affects expression of NLRP3 at the transcriptional level. To determine the mechanism underlying the regulatory activity of Gfi1 on NLRP3 expression, the mouse NLRP3 promoter was screened, leading to the identification of a putative binding site for Gfi1 (GRE1, located at nt -1210/-1207). In dual luciferase reporter assays performed with WT and GRE1 mutant promoters, the inhibitory effect of Gfi1 on NLRP3 transcription was significantly reversed upon GRE1 mutation. EMSA and ChIP assays performed to further establish the function of GRE1 (Figure 2A) validated the in vivo and in vitro interactions between Gfi1 and the GRE1 element, and consequently, a direct transcriptional repression effect on the NLRP3 gene.
NF-κB p65 activates NLRP3 transcription through binding two elements in its promoter region, and Gfi1 interferes with activity through direct interactions with p65. Accordingly, we mutated the binding elements of p65 (NRE1 and NRE2) in the NLRP3 promoter. Dual luciferase reporter assays showed that mutation of NRE1 almost entirely suppressed activation of p65 while mutation of NRE2 exerted no effect, indicating that NF-κB p65 specifically interacts with NRE1 for the activation in this system. EMSA studies further confirmed that Gfi1 strongly competes for binding of NF-κB p65 with NRE1, antagonizing interactions between p65 and the NLRP3 promoter (Figure 2B). Maximum suppression of NLRP3 promoter activity by Gfi1 was observed with the NLRP3 promoter reporter containing a NRE1/ GRE1 double mutant (Figure 2C).
Conclusions: In summary, we propose a “dual repression model” mechanism of Gfi1 in the regulation of NLRP3 expression. Once activated by LPS, NF-κB promotes NLRP3 expression by binding to the cis-element, NRE1, in turn, promoting assembly of the NLRP3 inflammasome to generate biologically active IL-1β. Meanwhile, Gfi1 is induced to control LPS-induced inflammation. Gfi1 directly inhibits NLRP3 transcription by binding the GRE1 site or blocks NF-κB-mediated NLRP3 transcription via interactions with NF-κB p65. Both modes of action lead to suppression of IL-1β release from macrophages in response to LPS stimulation. The present data, along with previous reports showing that Gfi1 restricts pro-IL-1β transcription, indicate that Gfi1 plays pivotal roles in regulating IL-1β production at both transcriptional and post-translational levels. These findings provide novel evidence that should aid in the development of future anti-inflammatory therapies to prevent IL-1β-induced tissue injury and mortality during sepsis.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal