Abstract
Background:
Bone marrow (BM) microenvironment plays an important role in development of drug resistance in acute myeloid leukemia (AML) by supporting survival of therapy resistant leukemic cells that eventually may lead to disease relapse. Consideration of tumor stroma factors is therefore critically important when assessing the efficacy of drugs in the ex vivo drug testing of primary AML cells. To study the effects of tumor stroma factors on the response of AML patient cells ex vivo to a panel of cancer drugs, we evaluated the effects of stroma-derived conditioned medium (CM) on the sensitivity of genomically defined primary AML cells.
Methods:
Primary AML cells were isolated by Ficoll gradient separation from BM aspirates or peripheral blood of AML patients (n=13). For drug testing, 303 small molecule inhibitors were plated on 384-well plates in 5 different concentrations over a 10,000-fold concentration range. The stromal-cell conditioned medium (CM) was made by culturing human bone marrow (BM) stromal cell line HS-5 (American Type Culture Collection) in RPMI 1640 medium for 3 days. AML cells were added to the plates in either CM diluted with RPMI 1640 medium (25% CM) or in mononuclear cell medium (MCM, Promocell), which was used as the standard medium comparison. Cell viability was measured after 72 h and dose response curves generated for each drug. Drug sensitivity scores (DSS) were calculated as described previously (Yadav et al, 2014). Phosphorylation profiles of 43 proteins were analyzed with a human phospho-kinase array (R&D Systems). In addition, somatic mutations were identified by exome sequencing using DNA from the leukemia cells and matched skin biopsies, while expressed fusion genes were identified by transcriptome sequencing.
Results:
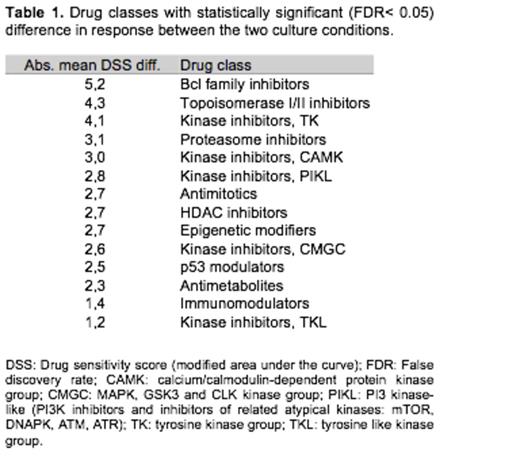
AML samples with activating mutations to kinases such as FLT3 or PDGFRB exhibited more sensitive ex vivo drug response profiles, particularly to broad-spectrum kinase inhibitors, compared to samples driven by other types of mutations. When the same AML samples were compared between the two conditions, CM or MCM, the drug sensitivities were different for many classes of drugs (Table 1). In CM, samples typically lost sensitivity to many of the tested drugs, such as topoisomerase II inhibitors, BCL2 inhibitors and several other classes of tyrosine kinase inhibitors (TKIs). The loss of TKI sensitivity in CM was particularly striking in the FLT3 and PDGFRB mutated cases. Cluster analysis of overall drug responses for AML samples tested in MCM resulted in a tight group of most TKIs, reflecting their overlapping target profiles. However, when the analysis was applied to responses from the same cells tested in CM, the TKI grouping was more dispersed. Thus, these results indicate that tyrosine kinase signaling is stringently regulated in standard medium, whereas CM helps to support cell survival resulting in lower responses to a range of TKIs. To test this hypothesis, phosphorylation of 43 different kinases was measured with AML samples incubated in either CM or MCM. CM induced phosphorylation of multiple proteins including p38α, HSP27, Src, Lyn, Hck and STAT6 proving the activation of other signaling pathways.
Conclusions:
Our dataindicate that stromal cell conditioned medium may have prominent effects on ex vivo drug responses of AML cells. BM factors likely provide survival cues that make primary patient-derived AML cells resistant to several targeted agents, such as topoisomerases and TKIs. This underscores the need to develop drug testing methods that take into account tumor-microenvironment interactions.
Gjertsen:BerGenBio AS: Membership on an entity's Board of Directors or advisory committees; Boehringer Ingelheim : Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Kinn Therapeutics AS: Equity Ownership. Porkka:Bristol-Myers Squibb: Honoraria, Research Funding; Novartis: Honoraria, Research Funding. Kallioniemi:Medisapiens: Consultancy, Membership on an entity's Board of Directors or advisory committees. Wennerberg:Pfizer: Research Funding. Heckman:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal