Abstract
Background: The last decade has seen impressive advances in identifying the genetic landscape and clinical heterogeneity of MDS. Diverse cytogenetic abnormalities and specific aberrations in RNA splicing, cell-signaling, transcription regulation and tumor suppressor genes are increasingly being applied for the prognostic stratification of these pts at diagnosis. Despite these advances, treatment options are limited to HMA therapy and lenalidomide; the survival advantage of these agents is established, but most pts eventually relapse. Furthermore, the prognosis for pts in whom HMA therapy has failed is grim, with a median OS of 4.3 to 5.6 months (Jabbour et al, Cancer, 2010; Prébet et al, J Clin Oncol, 2011). The clonal architecture and evolution of molecular changes has been chronicled in newly diagnosed MDS pts but the assessment of these abnormalities in pts who have failed or relapsed after HMAs is limited. Here we document for the first time the very high incidence of these molecular changes in higher-risk MDS patients after failure of HMAs and assess the relationship between the genetic and cytogenetic abnormalities and response to a novel agent, rigosertib. We correlate the results of cytogenetic abnormalities in HMA failures with response to rigosertib in the context of a clinical trial that compared this treatment with best supportive care.
Methods: Genomic DNA was isolated from single microscopic slides from 153 pts from Study 04-21 and subjected to sequence analysis of a myeloid panel comprising of 24 selected loci known to be frequently mutated in MDS and AML. Standardized cytogenetic investigations were performed using G banding and centrally reviewed. Whenever possible, 25 metaphases were analysed. Description of chromosome aberrations and clone definition followed the International System for Cytogenetic Nomenclature. FISH for deletion 5q was included. Depending on the aberrations detected during karyotyping, further probes were applied. A complex karyotype was defined as ≥3 independent aberrations within 1 clone.
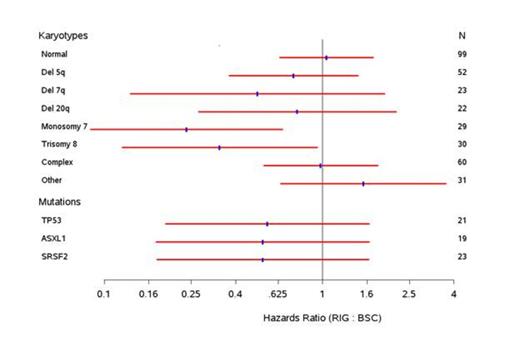
Results: Adequate DNA samples were obtained from 92 (60%) of 153 patients. All but 8 of the 92 samples carried at least 1 mutation (91%), with 16 of the 24 myeloid mutations detected. The most frequently mutated loci were TP53 (23%, mutations were detected at multiple coding regions of the protein), SRSF2 (17%), U2AF1 (16%). SF3B1 (13%), ASXL1 (13%) and TET2 (10%). Mutations were found in RUNX1 (5 samples); 4 samples each carried a mutation in ETV6 (4), EZH2 and N- and K-ras. All but 1 of the mutations were represented at >10% of the alleles, with a range of 9.2-94%. Sixty-two percent of mutations detected in rigosertib patients who did not respond to initial HMA therapy (primary HMA failure, 61% of the study population) carried single or multiple mutations. The effect of single and multiple mutations on OS is summarized in Figure 1. Patients carrying mutations in TP53, ASXL1, and SRSF2 showed a trend toward increased survival benefit of rigosertib therapy. It is noteworthy that pts with monosomy 7 and trisomy 8 mutations demonstrated a survival benefit with rigosertib therapy compared to BSC (monosomy 7: HR = 0.24, p = 0.0033; trisomy 8: HR = 0.34, p = 0.035). The significance of individual and combined mutations, in the context of founder and subclonal lesions is being evaluated further.
Conclusions: We have investigated the role of karyotype and point mutations in MDS patients after failure of HMA therapy and evaluated these changes to response in a clinical trial. Certain karyotypes were linked to enhanced survival benefit of rigosertib. The majority of second-line MDS patients carry mutations including those associated with poor prognosis. These results have important implications on designing therapeutic approaches and trials for MDS pts after failure of HMAs.
Mufti:Onconova Therapeutics, Inc: Research Funding. Best:Onconova Therapeutics, Inc: Research Funding. Lea:Onconova Therapeutics, Inc: Research Funding. Azarnia:Onconova Therapeutics, Inc: Employment. Wilhelm:Onconova Therapeutics, Inc: Employment, Equity Ownership. Goehring:Onconova Therapeutics, Inc: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal