Abstract
An irreversible proteasome inhibitor with decreased peripheral neuropathy compared to bortezomib, Carfilzomib (Cz) has potent anti-MM effects resulting in deep clinical responses and durable remissions. In this two-stage phase II trial of 45 planned patients, we treat newly diagnosed MM patients with Cz, lenalidomide(Ln), and dexamethasone (Dx) followed by 2 years of Ln maintenance.
Eight 28-day cycles of combination therapy comprises: Cz IV 20/36 mg/m2 on days 1, 2, 8, 9, 15, 16; Ln oral 25 mg days 1-21; and Dx IV or oral 20/10 mg (C1-4/5-8) on days 1, 2, 8, 9, 15, 16, 22, 23. Transplant eligible patients undergo stem cell collection after 4 cycles of CRd and continue with treatment. After 8 cycles of combination therapy, patients with SD or better receive up to 24 cycles of Ln extended dosing 10 mg days 1-21. Primary endpoint is ≥ grade 3 neuropathy. Bone marrow samples are collected at baseline, C1D2 (single agent Cz exposed), CR/end of cycle 8 (cycles 1-8), CR/end of cycle 20 (cycles 9-20), and CR/end of cycle 32/treatment termination (cycles 21-32). Patients are evaluated for clinical biomarkers, FDG-PET CT, and MRD studies (flow cytometry and PCR) at regular time points. Flow cytometry utilizes 8-color flow panel and analyzes ≥ 3 x 106 events (sensitivity 1 x 10-5).
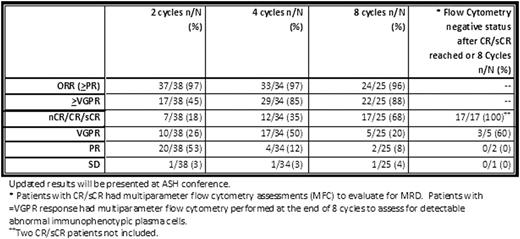
Forty-one patients meeting eligibility criteria have been enrolled (24 male, 17 female; median age 60; range 40-88). Among patients enrolled, median M-spike is 2.9 (1.0-7.6 g/dL) and isotypes included 26-IgG, 10 – IgA, 4 – free kappa, and 1 – free lambda. Thirty-eight patients are evaluable for response and toxicity. No patients have had ≥ grade 3 neuropathy. The mean decline in serum M-protein (n=33) was 72% after 1 cycle of CRd received. Best responses after a median of 9 cycles (range 2-20) completed, include 17–sCR/1-CR/6-nCR (63%), 10-VGPR (26%), 3-PR (8%), and 1-SD (3%). Among 18/38 (47%), median time to sCR/CR was 5 cycles. Among 14 CR/sCR + 3 nCR patients who underwent MRD assessment, all were negative. Among patients with ≤ VGPR at the end of 8 cycles of CRd, 5/8 (63%) had evidence of immunophenotypically abnormal plasma cells defined by flow cytometry, and 3 patients had undetectable abnormal plasma cells (1 achieved sCR after 8 additional Ln cycles). After median follow-up of 10 months (3-22), PFS rate was 83.3%. Twenty-five patients have completed 8 cycles of CRd, while 24/25 continued to Ln extension and 1 patient opted to exit study after CRd. Patients with non-hematologic toxicity events (≥ grade 3) include: electrolyte disturbances - 7(18%), LFT elevation - 5(13%), rash/pruritus – 4(11%), fatigue – 4(11%), cardiovascular – 3(8%), dyspnea/respiratory – 3(8%), constitutional symptoms – 2(5%), infections – 2(5%), VTE - 2(5%), and anxiety – 1(3%). Patients with hematologic toxicity events (≥ grade 3) include: lymphopenia –24(63%), anemia – 6(16%), leukopenia - 5(13%), and thrombocytopenia - 4(11%). All patients with baseline FDG avid lesions or extramedullary disease showed decrease or resolution of FDG avidity at MRD assessment time points.
Beyond traditional clinical response criteria, using a functional imaging and highly sensitive MRD assays, we consistently show high rates of deep remission and MRD negativity among newly diagnosed MM patients treated with carfilzomib, revlimid, dexamethasone (CRd) combination therapy. The results were very similar across age-groups and the oldest patient on this trial was 88 years old. If deep remissions are maintained with a delayed ASCT strategy incorporating extended dosing revlimid therapy, this may be a future strategy available to all patients irrespective of age.
Off Label Use: Abstract is in newly diagnosed Multiple Myeloma patients. Carfilzomib is approved for relapsed/refractory patients.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal