Abstract
Hodgkin lymphoma (HL) is characterized by the presence of CD30-positive Hodgkin Reed-Sternberg cells. The standard of care for patients (pts) with relapsed or refractory HL is salvage chemotherapy followed by autologous stem cell transplant (auto-SCT). However, approximately 50% of pts experience relapse of HL after auto-SCT and this population represents a pronounced unmet need. In a 756-pt retrospective analysis, the median overall survival (OS) in HL pts who relapsed after auto-SCT was 2.4 years, as measured from the time of auto-SCT, with shorter time to relapse after auto-SCT being the most predictive factor for shortened survival (Arai 2013). Brentuximab vedotin (ADCETRIS®) comprises an anti-CD30 antibody conjugated by a protease-cleavable linker to monomethyl auristatin E (MMAE), a microtubule-disrupting agent. A pivotal phase 2 study was conducted to determine the efficacy and safety of brentuximab vedotin in 102 pts with relapsed or refractory HL after auto-SCT (ClinicalTrials.gov #NCT00848926). Data representing approximately 3 years of follow up from this ongoing trial are described, including characterization of patients who experienced long-term remissions.
Pts received 1.8 mg/kg brentuximab vedotin every 3 weeks as a 30-minute outpatient IV infusion for up to 16 cycles. The primary endpoint was the objective response rate (ORR) per independent review according to the Revised Response Criteria for Malignant Lymphoma (Cheson 2007). Long-term follow-up assessments to determine survival and disease status occurred every 3 months (mos) for 2 years, every 6 mos during years 3 to 5, and annually thereafter.
In this high-risk population with poor prognosis, the median time to relapse after auto-SCT was 6.7 mos (range, 0–131 mos). Pts received a median of 9 cycles of brentuximab vedotin and the ORR was 75% (76 of 102 pts), with complete remissions (CRs) in 33% of pts (n=34). As previously reported, the most common (≥15%) brentuximab vedotin-related adverse events of any grade were peripheral sensory neuropathy, nausea, fatigue, neutropenia, and diarrhea. Adverse events of Grade 3 or higher that occurred in ≥5% of pts were neutropenia, peripheral sensory neuropathy, thrombocytopenia, and anemia.
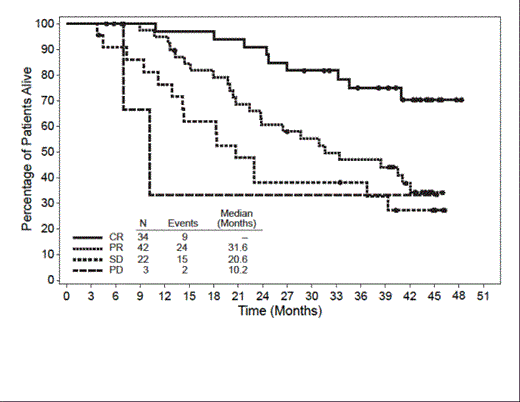
At the time of this analysis (May 2013), the median observation time from first dose was 32.7 mos (range, 1.8 to 48.3 mos). Of the 102 pts enrolled, 51 (50%) were alive at the time of last follow up. The median OS was 40.5 mos (95% CI: 28.7, – [range, 1.8 to 48.3+ mos]) and the estimated 36-mo survival rate was 54% (95% CI: 44%, 64%). Median OS by best clinical response was CR (n=34): median not yet reached; partial remission (PR, n=42): 31.6 mos; stable disease (SD, n=22): 20.6 mos; and progressive disease (PD, n=3): 10.2 mos. Of the 51 pts who were alive at the time of this analysis, 14 remain in remission and have not started a new anti-cancer therapy other than 5 pts who received consolidative allo-SCT following brentuximab vedotin. Demographics/baseline characteristics of these 14 pts show that the majority are female (10/14; 71%) and white (12/14; 86%), with a median age of 26.5 years (range, 15–54). Eleven of the 14 pts had CRs and 3 had PRs following brentuximab vedotin; all 3 of the pts with PRs received subsequent allo-SCT. The observation time for the 14 pts who remain in remission ranges from 31.5 mos to 44.4 mos and their progression-free survival ranges from 27.2+ mos to 44.4+ mos. Additional characterization of the pts who achieved long-term remissions following brentuximab vedotin will be presented at the meeting.
Overall Survival by Best Clinical Response
After a median observation time of approximately 3 years from first dose of brentuximab vedotin, 50% of pts with relapsed or refractory HL were alive at the time of last follow up. The median OS was 40.5 mos. Fourteen patients remain in follow up with no evidence of lymphoma progression, providing early suggestion that a fraction of these patients may be cured. A randomized phase 3 study is being conducted to evaluate brentuximab vedotin in combination with AVD (doxorubicin, vinblastine, and dacarbazine) versus ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) for frontline treatment of HL (ClinicalTrials.gov #NCT01712490).
Gopal:Biogen, Idec: Research Funding; Merck: Research Funding; BioMarin: Research Funding; Gilead: Research Funding; Emergent/Abbott: Research Funding; Pfizer: Research Funding; Cephalon/Teva: Research Funding; Janssen: Research Funding; Millennium: Honoraria, Research Funding; Sanofi-Aventis: Consultancy, Honoraria; Seattle Genetics, Inc.: Consultancy, Honoraria, Research Funding; Piramal: Research Funding; Spectrum: Research Funding. Chen:Seattle Genetics, Inc.: Consultancy, Research Funding, Speakers Bureau, Trave expenses Other. Smith:Seattle Genetics, Inc.: Research Funding; Spectrum: Consultancy; Cephalon: Consultancy, Speakers Bureau; Celgene: Consultancy, Speakers Bureau; GlaxoSmith Kline: Speakers Bureau. Ansell:Seattle Genetics, Inc.: Research Funding. Rosenblatt:Seattle Genetics, Inc.: Research Funding; University of Miami: Employment. Savage:Seattle Genetics, Inc.: Consultancy, Honoraria, Research Funding. Connors:F Hoffmann-La Roche: Research Funding; Roche Canada: Research Funding. Engert:Seattle Genetics, Inc.: Honoraria, Research Funding; Millennium: Honoraria, Research Funding; Takeda: Honoraria. Larsen:Seattle Genetics, Inc.: Employment, Equity Ownership. Huebner:Takeda Cambridge US: Employment; Takeda: Equity Ownership. Sievers:Seattle Genetics, Inc.: Employment, Equity Ownership. Younes:Seattle Genetics, Inc.: Advisory/Scientific Board Membership Other, Honoraria, Research Funding; Sanofi-Aventis: Honoraria; Novartis: Honoraria; Celgene: Honoraria; Incyte: Honoraria; Millennium: Honoraria; Pharmacyclics: Honoraria; Curis: Honoraria; Genentech: Research Funding; Gilead: Research Funding; Johnson and Johnson: Research Funding; Infinity: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal