Abstract
The use of allogeneic hematopoietic cell transplantation (HCT) in multiple myeloma (MM) remains controversial but the role of allogeneic HCT for consolidation of initial response in the era of novel agents has not been defined. Therefore, we conducted a phase 2 study of allogeneic HCT in MM patients achieving at least very good partial response (VGPR) after initial therapy.
Seventeen MM patients with first VGPR or complete remission (CR) received allogeneic HCT between 01/2010 and 04/2013 at Moffitt Cancer Center (NCT 00948922). Eligible patients were age ≤ 60, achieving ≥ VGPR, and have suitable HLA-matched donors. Six patients received bortezomib plus lenalidomide induction, 7 received bortezomib-based, and 4 received lenalidomide-based induction. Three patients (18%) underwent autologous HCT with melphalan plus bortezomib conditioning (melphalan 100 mg/m2 for 2 days followed by bortezomib 1.3 mg/m2) following induction therapies in order to achieve further cytoreduction. Conditioning regimen before allogeneic HCT consisted of fludarabine 30 mg/m2 for 4 days and melphalan 70 mg/m2 for 2 days followed by a single dose of bortezomib 1.3 mg/m2 (Flu/Mel/Vel). GVHD prophylaxis was tacrolimus plus either methotrexate (n=9), or mycophenolate mofetil (n=4), or sirolimus (n=4). No maintenance therapy was prescribed after allogeneic HCT.
The median age at transplant was 51 (25 – 57) years and the median time from initial therapy to transplant was 236 (126 – 418) days. Seven patients (41%) have high-risk cytogenetics/FISH. The median number of prior therapy was 1. Disease status at the time of allogeneic HCT was VGPR (n=8), CR (n=3), or stringent CR (n=6). All patients received unmanipulated peripheral blood stem cell grafts from HLA-matched sibling donors (n=7) or 8/8 HLA-matched unrelated donors (n=10). All patients achieved neutrophil engraftment with a median of 14 (11 - 18) days and platelet engraftment with a median of 17 (13 - 21) days. All patients established stable donor cell chimerism. Best responses after allogeneic HCT were CR (n=2), sCR (n=13), and disease progression (n=2). The 2-year progression-free survival estimate is 80% (95%CI: 51 – 98) for standard-risk group and 51% (95%CI: 15 – 87) for high-risk, respectively. With a median follow up of 18 (3 - 43) months for surviving patients, the 2-year overall survival estimate is 88% (95%CI: 68 – 99). The cumulative incidence of non-relapse mortality was 6% (95%CI: 0.0 – 22) at 100 days and 13% (95%CI: 1 – 34) at 1 year, respectively. The cumulative incidence of grades 2-4 acute GVHD at day 100 was 41% (95% CI: 20 – 65) and the cumulative incidence of moderate to severe chronic GVHD at 1 year was 8.0% (95% CI: 0.0 – 29).
These results indicate that allogeneic HCT for MM in VGPR or CR as consolidation achieves favorable disease control. The study is ongoing to assess long-term safety of this modality. A multicenter trial is planned to evaluate the utility of allogeneic HCT in high-risk MM.
Overall Survival
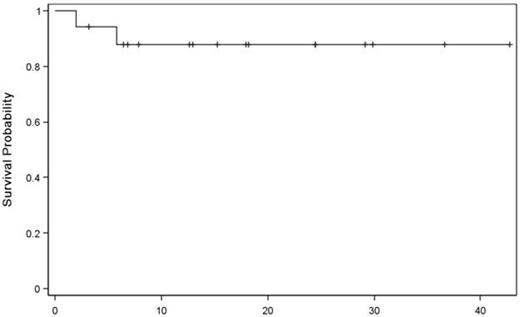
Months
Off Label Use: bortezomib - transplant conditioning. Alsina:Millennium: Membership on an entity’s Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal