Abstract
Activation of canonical and noncanonical NF-kB pathways plays a key role in multiple myeloma (MM) pathogenesis. Recent studies have shown that constitutive activation of NF-kB pathways is present in ∼15-20 % of newly diagnosed MM patients. Bortezomib is a potent selective inhibitor of NF-kB activation; however its cytotoxicity is mainly due to inhibition of the noncanonical NF-kB pathway. We have previously shown that the achievement of very good partial response (VGPR) after induction therapy prior to frontline autologous stem cell transplantation (ASCT) was a favourable prognostic factor for progression-free survival (Moreau et al, Blood 2011;117:3041-3044). Our aim was to correlate NF-kB activity, reflected by a gene signature of well-known NF-kB targets, with the response rate achieved with a bortezomib-based induction regimen prior to high-dose therapy and ASCT performed as part of frontline treatment in patients with symptomatic multiple myeloma.
One hundred and ninety-nine patients with symptomatic MM were enrolled in the prospective randomized IFM2007-02 trial comparing 4 cycles of bortezomib-dexamethasone (VD) induction therapy versus 4 cycles of the triplet combination bortezomib-thalidomide-dexamethasone (VTD) prior to ASCT. Among these 199 patients, 114 included in the present study were available for gene expression profile testing and therefore analysed for NF-kB activity. For each patient, the NF-kB(10) index, a reliable measure of NF-kB activity in MM tumor cells (Demchenko et al, Blood 2010; 115: 3541-3552) was calculated. NF-kB(10) index is based on a transcription signature of 10 genes: IL2RG, NFKB2,TNFAIP3, NFKBIE, NFKBIA, RELB, CD74, PLEK, MALT1 and WNT10. Gene expression signature was obtained from Affymetrix Exon1.0 normalized data. A high level of NF-kB activity was defined by the NF-kB(10) index found in a cohort of 20 MM patients with biallelic deletions, identified by using Affymetrix SNP6.0 data, that inactivate negative regulators (cIAP1/2) of NF-kB pathways. Response to induction therapy was evaluated according to the IMWG criteria.
Responses to induction therapy are shown in Table 1. In this subgroup analysis, the CR plus VGPR rates were not statistically different in both arms of the trial: 24 / 54 (44%) in the VTD arm versus 21 / 60 (35%) in the VD arm, P = .35. We subsequently analyzed the correlation between NF-kB activity and response in the whole cohort of 114 patients regardless of induction treatment.
Table patient's response
| . | VD (n = 60) . | VTD (n = 54) . |
|---|---|---|
| CR | 14 | 14 |
| VGPR | 7 | 10 |
| PR | 30 | 23 |
| stable | 6 | 3 |
| progression | 3 | 4 |
| . | VD (n = 60) . | VTD (n = 54) . |
|---|---|---|
| CR | 14 | 14 |
| VGPR | 7 | 10 |
| PR | 30 | 23 |
| stable | 6 | 3 |
| progression | 3 | 4 |
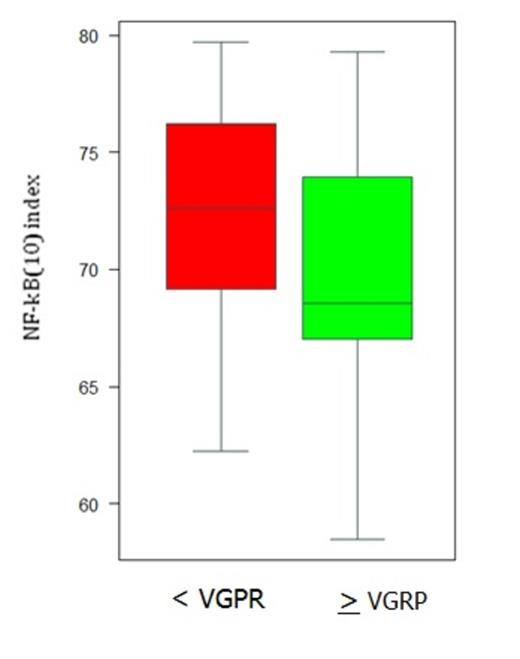
We found that the level of NF-kB activity, based NF-kB(10) index, tested as a continuous variable, was strongly correlated with the quality of response, i.e. VGPR or better (P = .007 Wilcoxon test). We also investigated the cut-off value of NF-kB(10) index that could impact the response rate. Since high NF-kB(10) index was found in MM cells with known NF-kB mutations, we calculated the NF-kB(10) index in a control cohort of MM patients deleted in cIAP1/2. Patients of IFM2007-02 trial with a NF-kB(10) index (< 70.5) lower than the index of the control cohort were assigned to the low NF-kB activity group (50/114, 44%); therefore the remaining cases (64/114, 56%) presented with a high NF-kB activity. We found that patients with a reduced NF-kB(10) index displayed a significantly higher response rate (27/50; 54% vs 18/64; 28%, P= .007), as shown in Figure 1, indicating that the vast majority of patients with high NF-kB were not able to achieve at least VGPR.
Our results show that a low level NF-kB activity is associated with a higher response rate (>VGPR) to bortezomib-based induction regimen. Patients with low NF-kB(10) index represent 44% of the cohort studied. Since NK-kB activity is related to both canonical and noncanonical pathways, and knowing that bortezomib-induced cytotoxicity is mostly due to the inhibition of the noncanonical pathway only, our data strongly suggest that bortezomib should be combined with drug also targeting the canonical pathway in order to induce a high response rate in patients with increased NF-kB activity, as shown in preclinical studies (Fabre et al, Clin Cancer Res. 2012;18:4669-4681.
Moreau:CELGENE: Honoraria, Speakers Bureau; JANSSEN: Honoraria, Speakers Bureau. Off Label Use: FRONTLINE TREATMENT WITH CARFIZOMIB. Attal:CELGENE: Honoraria, Speakers Bureau; JANSSEN: Honoraria, Speakers Bureau. Facon:Janssen and Celgene: Speakers Bureau; Millennium, Onyx, Novartis, BMS, Amgen: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal