Abstract
Abstract 5048
Novel drugs, such as bortezomib, have significantly improved the response rates in multiple myeloma (MM), but little has been reported on bortezomib-based therapies in Chinese patients.
In the initial eight 28-day cycles, newly diagnosed ymptomatic patients were treated with combination therapy including bortezomib plus dexamethasone (PD) and the triplet combinations of PD with adriamycin (PAD), cyclophosphamide (PCD), thalidomide (PDT) between February 1, 2006 and May 31, 2012. Among the above regimens, bortezomib (1. 3 mg/m2) was given intravenously on days 1, 4, 8, 11, while dexamethasone (20 mg/m2/day) was given intravenously on days 1–2, 4–5, 8–9, 11–12, adriamycin (10 mg/m2) was given intravenously on days 1–4, cyclophosphamide (200 mg/m2) was given intravenously on days 1–4 and thalidomide (100 mg) was administered orally each day.
The overall response rate (¡Ý partial response, PR) of all the 151 eligible patients was 88. 7% (including 29. 8% very good partial response (VGPR) and 25. 8% complete response/near complete response (CR/nCR). The responses per IMWG criteria for patients are shown in Table 2. The median PFS was 20. 3 months (95% CI: 14. 8–25. 8 months) in the patients who received PDT, 24. 8 months (95% CI: 20. 0–30. 0 months) in the patients who received PCD, 22. 9 months (95% CI: 17. 6–28. 2 months) in patients who received PAD and 21. 8 months (95% CI: 15. 3–28. 3 months) in the patients who received PD with no significant differences between the groups. The median OS for PD arm was 42. 0(95% CI: 20. 1–63. 9 months) months while other arms were not reached, but the median OS for PDT, PCD and PAD was significant longer than PD (P=0. 042, 0. 039, 0. 010). PFS and OS for patients with favorable cytogenetics were significantly longer than those with unfavorable cytogenetics by FISH. The frequently observed hematologic toxicities (Grade 3/4) were: thrombocytopenia (17. 00%), neutropenia (15. 00%) and anemia (8. 61%). The most common non-hematologic toxicities included (all Grades) peripheral neuropathy(57. 61%), fatigue(27. 15%), infection(23. 84%), constipation(22. 52%), herpes zoster(17. 22%) and diarrhea(15. 23%).
Our experience indicated that bortezomib-based regimens were active and well-tolerated for MM patients, and triplet combinations were superior to PD. Serious Adverse events were rare in the Chinese patients with MM who received bortezomib-based chemotherapy.
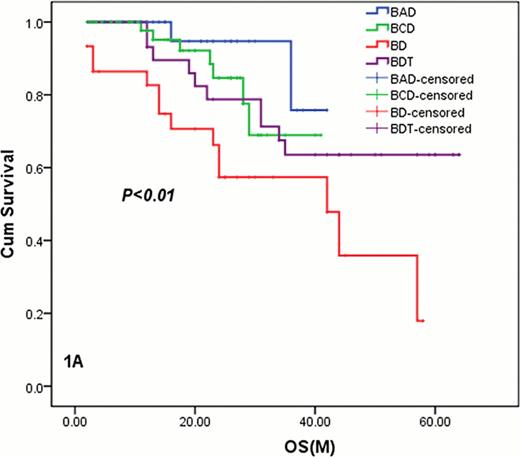
Kaplan-Meier survival curves for patients who received PDT, PCD, PAD and PD
1A: The OS of all the 151 eligible patients who received PDT, PCD, PAD and PD. The median OS for PD arm was 42. 0(95% CI: 20. 1–63. 9 months) months while other arms were not reached, but the median OS for PDT, PCD and PAD was significant longer than PD (P=0. 042, 0. 039, 0. 010).
Kaplan-Meier survival curves for patients who received PDT, PCD, PAD and PD
1A: The OS of all the 151 eligible patients who received PDT, PCD, PAD and PD. The median OS for PD arm was 42. 0(95% CI: 20. 1–63. 9 months) months while other arms were not reached, but the median OS for PDT, PCD and PAD was significant longer than PD (P=0. 042, 0. 039, 0. 010).
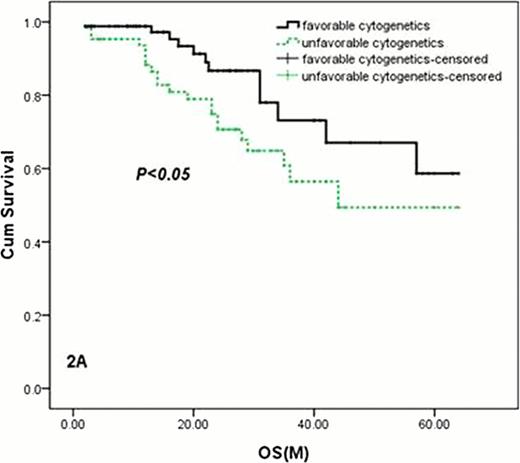
Kaplan-Meier survival curves for patients with favorable cytogenetics and unfavorable cytogenetics (with 1q21 amplification, 17p or 13q deletion, 14q32 rearrangement) by FISH.
2A: The OS of patients with favorable cytogenetics(FISH-negative) was longer than those with unfavorable cytogenetics (with 1q21 amplification, 17p or 13q deletion, 14q32 rearrangement) by FISH, P=0. 045.
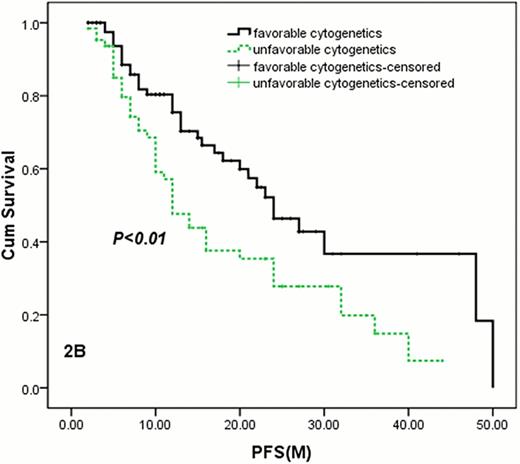
2B: The median PFS of patients with favorable cytogenetics(FISH-negative) was longer than those with unfavorable cytogenetics (with 1q21 amplification, 17p or deletion, 14q32 rearrangement) by FISH. 24. 0 months (95% CI: 18. 1–29. 9 months) vs 12. 0 months (95% CI: 8. 6–15. 4 months), P=0. 005.
Kaplan-Meier survival curves for patients with favorable cytogenetics and unfavorable cytogenetics (with 1q21 amplification, 17p or 13q deletion, 14q32 rearrangement) by FISH.
2A: The OS of patients with favorable cytogenetics(FISH-negative) was longer than those with unfavorable cytogenetics (with 1q21 amplification, 17p or 13q deletion, 14q32 rearrangement) by FISH, P=0. 045.
2B: The median PFS of patients with favorable cytogenetics(FISH-negative) was longer than those with unfavorable cytogenetics (with 1q21 amplification, 17p or deletion, 14q32 rearrangement) by FISH. 24. 0 months (95% CI: 18. 1–29. 9 months) vs 12. 0 months (95% CI: 8. 6–15. 4 months), P=0. 005.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal