Abstract
Abstract 425
FDA and EMA have approved Deferasirox for iron chelation therapy for transfusion dependent MDS patients after a limited experience in selected MDS patients. Therefore we felt the need of a large, prospective, trial to verify Deferasirox safety, compliance, efficacy and effect on erythroid function in a large, “real world,” MDS population.
One hundred fifty-two consecutive MDS IPSS lower risks (62 low and 90 intermediate1) transfusion dependent (minimum transfusion history 20 units received) patients were enrolled in 37 Italian centres. Patients received Deferasirox at the starting dose of 20 mg/kg. Characteristics of patients have been summarized by means of cross-tabulations for categorical variables and by means of median and range for continuous data. Adverse events (AEs) were defined according to CTCAE.3 (2003) definition. Disease progression was defined as advance to higher IPPS score group, development of acute leukemia or death. Efficacy of the treatment was measured by monitoring monthly serum ferritin levels. Transfusion independence was defined as three consecutive months without PRBC requirement with a minimum stable Hb level of 9 g/dl. The probability of drop out and transfusion independence were estimated using the appropriate non-parametric method, considering death and progression disease as a competing risk. Differences in univariate analysis were evaluated by Gray test. Fine and Gray model was used to evaluated prognostic factors in multivariate analysis. FriedmanÕs test was performed to evaluate differences in serum ferritin levels, hemoglobin level and transfusions input over time.
Ninety-six males and 56 females were enrolled. Median age was 72 years (66–77). Median interval diagnosis-enrolment was 32 months (17–54). Patients have been receiving regular blood transfusions for a median of 21 months (10–36) with a median number of units received of 37 (22–63). At baseline median CIRS co-morbidity score was 0 (0–1), CIRS severity score 0 (0–1), and Charlson co-morbidity score 0(0–1).
Eighty-four patients (55%) prematurely interrupted the study while 68 (45%) completed the planned year of treatment. Risk of drop out was 18.8% and 28% at 6 and 12 months, respectively. Risk of disease progression or death was 12.7% and 25% at 6 and 12 months, respectively. In multivariate analyses significant risk factors for treatment interruption were shorter diagnosis-enrolment interval (P=0.0008), lower Deferasirox dose (p=0.008) and higher serum ferritin level (P=0.004).
During the study 304 AEs were reported in 107 distinct patients. Of these events 93 (66 patients = 43%) were defined as possible-probably-certain drug related. Of these 93 related events fourteen in 11 distinct patients (7%) were superior to grade 2. Features of AEs were similar to those already reported.
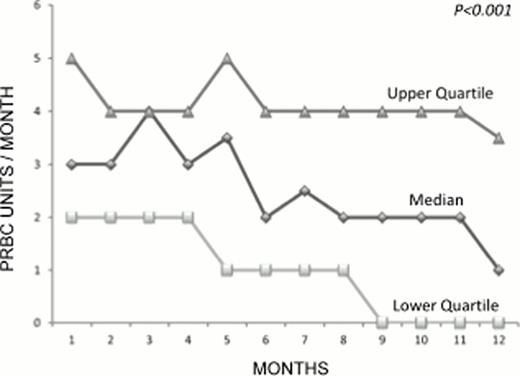
Serum ferritin significantly decreased during the study from a median starting value of 1966 ng/ml (1416–2998) to a median final value of 1475 ng/ml (915–2010), (P<0.0001). With equivalent pre-transfusional haemoglobin level (p=0.27) transfusion requirement decreased during the study. Median number of transfusions received per month was 3 (2–5) at starting the study and 1 (0–4) after one year (P= 0.0001) (figure 1). Twenty-two patients acquired transfusion independence with a probability of acquiring transfusion independency of 5.5% (95%CI 5.4–5.6), 15.7% (95%CI 15.4–15.9) and 19.7% (95% CI 19.4–20) after 6, 9 and 12 months of treatment, respectively.
Less than 50% of the enrolled population was able to complete the planned year of treatment because of drop out (risk 28%) and progression (risk 25%) despite a limited number of >2 grade AEs. Noteworthy >69% of the AEs was not drug related indicating a basic vulnerability of this population. Deferasirox was effective in reducing serum ferritin levels. Clinical benefit was also related to erythroid improvement that was clinically significant reducing transfusion need in a relevant percentage of patients. ClinicalTrials.gov Identifier: NCT00469560
Angelucci:Novartis : Chair of Steering Committee of the Telesto trial Other. Vallisa:CELGENE CORPORATION: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal