Abstract
Smoldering multiple myeloma (SMM) is an asymptomatic precursor disease of multiple myeloma, and is defined by excess bone marrow plasma cells and monoclonal protein without evidence of end-organ damage (hypercalcemia, renal insufficiency, anemia, or bone lesions [CRAB]). The identification of SMM patients with more aggressive underlying disease remains a challenge. We hypothesize that SMM is a clinical entity comprised of both premalignant, high-risk MGUS and early multiple myeloma in transition to malignant disease, which may be differentiated with the use of the serum FLC (FLC) ratio.
This was a retrospective analysis of 586 patients with newly diagnosed SMM from 1970–2010 with available stored serum samples around the time of diagnosis to be utilized for quantification of FLC ratios. SMM was defined by the International Myeloma Working Group 2003 definition; serum M-protein ≥ 3 g/dL and/or ≥ 10% bone marrow plasma cells with no evidence of CRAB features. The immunoglobulin FLC assay (Binding Site, U.K.) was used for testing. The FLC ratio was calculated as κ/λ (reference range 0.26–1.65). The involved/uninvolved FLC ratio was recorded to simplify the reporting of data. Receiver Operating Characteristics (ROC) curves were created to assess the ability of the FLC ratio to discriminate patients who progressed to symptomatic multiple myeloma (MM) in the first 2 years or at any point during follow-up versus patients without evidence of progression. Patients with less than 24 months follow-up without progression were censored. The optimal diagnostic cut-point for FLC involved/uninvolved ratio to identify patients with progressive disease from the ROC curve was >88.6 (equivalent to <0.011 or >88.6). For ease of clinical application, the optimal value for involved/uninvolved FLC ratio was rounded to >100. Time to progression (TTP) from date of the initial FLC to active MM was calculated using Kaplan-Meier analysis and compared to patients with a high (>100) and low (<100) involved/uninvolved FLC ratio at time of SMM diagnosis. TTP within 24 months of the initial FLC was also calculated.
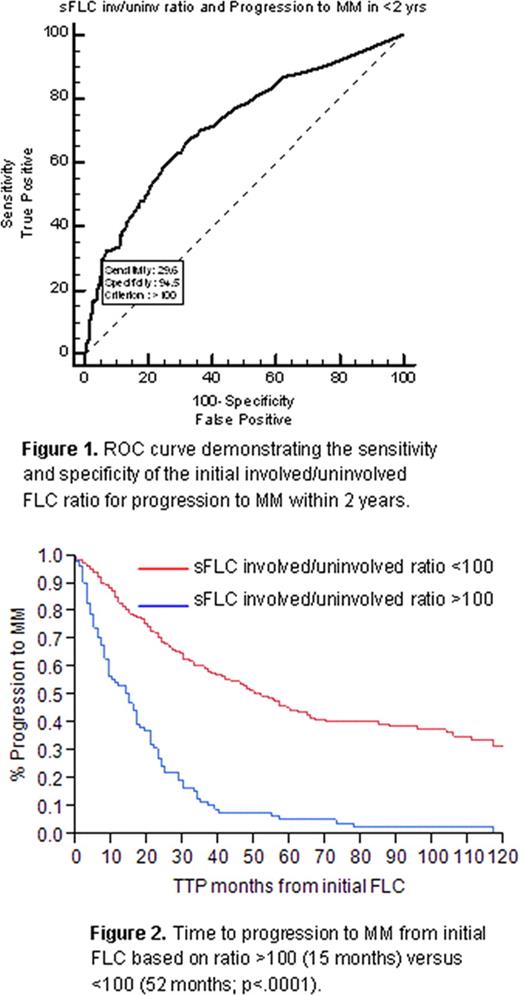
During the study period, 54% of patients progressed to active MM. On ROC analysis, a cut-point of >100 corresponded to a sensitivity of 25% (95% CI, 20.5–30.4) and specificity of 99.3% (97.3–99.9), with positive likelihood (+LR) ratio of 33.9 (38.1–41.0), negative likelihood ratio (−LR) of 0.75 (0.2–3.0), positive predictive value (PPV) of 97.6 (91.5–99.7) and negative predictive value of 53.0 (48.5–57.4). Using the ROC to assess progression to MM within 24 months (Figure 1), sensitivity was 29.6% (23.5–36.4), specificity 94.5% (91.7–96.5), +LR 5.36 (4.3–6.6), -LR 0.75 (0.5–1.1), PPV 85.8 (77.7–91.8), and NPV 54.3 (49.8–58.9). Median TTP to active MM in the FLC >100 group was 15 months (9–17) versus 52 months (44–60) in the FLC <100 group (p <.0001) [Figure 2]. In the FLC ratio >100 group, progression at 1 year was 47%, 76% at 2 years, and 90% at 3 years. Only 25% of the FLC <100 patients had progressed at 2 years. The most common progression event was bone disease (42%), followed by anemia (26%), renal impairment (23%), and hypercalcemia (5%).
Elevation of the FLC ratio >100 (or <0.01) is highly specific for the future development of active MM, with 76% of these patients developing end-organ damage requiring therapy within 2 years. Risk of transformation to MM in the FLC <100 group was similar to previously reported rates of 10% per year for the first 5 years. Development of an FLC ratio >100 is associated with increasing disease burden and in this study behaved in a malignant fashion rather than a precursor state. The FLC is a simple and useful predictor of progression to MM in SMM, and patients with FLC ratios of <0.01 or >100 within the first 2 years of SMM diagnosis should be monitored especially closely. Future studies are needed to determine optimum cutoffs for FLC ratio to where a change in definition of MM could be considered.
No relevant conflicts of interest to declare.

This icon denotes a clinically relevant abstract
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal