Abstract
Abstract 3762
The EUTOS Score was developed and validated as a prognostic tool for the achievement of complete cytogenetic response (CCR) at 18 months for chronic phase (CP) CML patients under imatinib therapy. The score identifies high-risk patients not reaching CCR at 18 months with a positive predictive value of 34% and a specificity of 92% using only two variables, peripheral blood basophils and spleen size at diagnosis (Hasford et al. Blood 2011). We sought to evaluate the clinical impact of the EUTOS score to predict molecular response. Therefore, we analyzed the EUTOS score with patients from the German CML-Study IV, a randomized 5-arm trial (imatinib 400 mg vs. imatinib 800 mg vs. imatinib in combination with interferon alpha vs. imatinib in combination with araC vs. imatinib after interferon failure).
From July 2002 to December 2010, 1,502 patients with BCR-ABL positive CML in CP were randomized. 129 patients with imatinib after interferon alpha and 36 other patients had to be excluded (14 due to incorrect randomization or withdrawal of consent, 22 with missing baseline information). 1,337 patients were evaluable for overall and progression-free survival (OS and PFS), 1,252 for molecular responses. 749 of these patients were part of the score development sample. Therefore cytogenetic analyses are not described here.
By EURO score, 36% of patients (n=475) were low risk, 51% (n=681) intermediate risk, and 12% (n=167) high risk. The EUTOS score was low risk in 88% (n=1163) and high risk in 12% (n=160). The high-risk patients differed between the two scores: EUTOS high-risk patients were classified according to EURO score in 12% as low (n=19), in 45% as intermediate (n=68) and in 43% as high risk (n=73).
Patients with high, intermediate, and low risk EURO score achieved MMR in 22, 16, and 13 months and CMR4 (BCR-ABL <=0.01%) in 59, 41, and 34 months. P-values for low vs. intermediate risk groups were borderline only (0.03 for MMR and 0.04 for CMR4), whereas p-values for high vs. low/intermediate risk groups were for both molecular response levels <0.001. At 12 months the proportion of patients in MMR was 38%, 46%, 54% for high, intermediate, and low risk patients, respectively. Similar results were observed with the Sokal score.
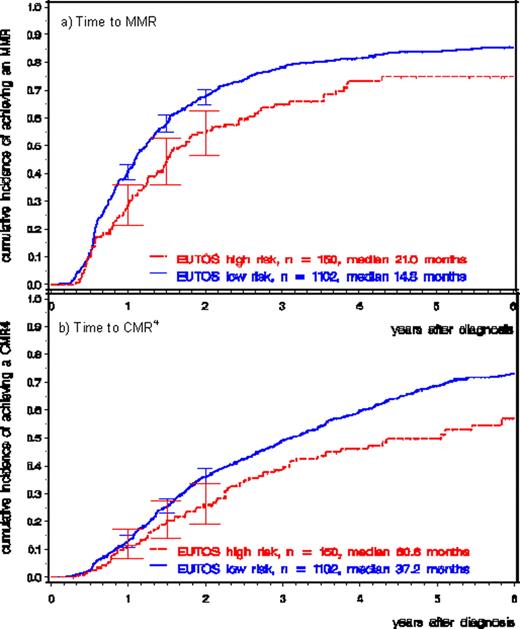
Patients with high risk EUTOS score achieved deep molecular responses (MMR and CMR4) significantly later than patients with low risk EUTOS score (MMR: median 21.0 vs. 14.8 months, p<0.001, Fig. 1a; CMR4: median 60.6 vs. 37.2 months, p<0.001, Fig. 1b).
The proportions of patients achieving MMR at 12 months were significantly lower in the EUTOS high-risk group than in the EUTOS low-risk group (30.8% vs. 50.6%, p<0.001).
OS after 5 years was 85% for high and 91% for low risk patients (p=n.s.), PFS was 85% and 90%, respectively.
The EUTOS score clearly separates CML patients also according to MMR and CMR4 (MR4). The new EUTOS score should be used in future trials with tyrosine kinase inhibitors in CML.
Neubauer:Novartis: Honoraria, Research Funding; Roche: Research Funding. Kneba:Hoffmann La Roche: Honoraria. Schnittger:MLL Munich Leukemia Laboratory: Employment, Equity Ownership. Hochhaus:Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees; Ariad: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. German CML Study Group:Deutsche Krebshilfe: Research Funding; Novartis: Research Funding; BMBF: Research Funding; EU: Research Funding; Roche: Research Funding; Essex: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal