Abstract
Abstract 2563
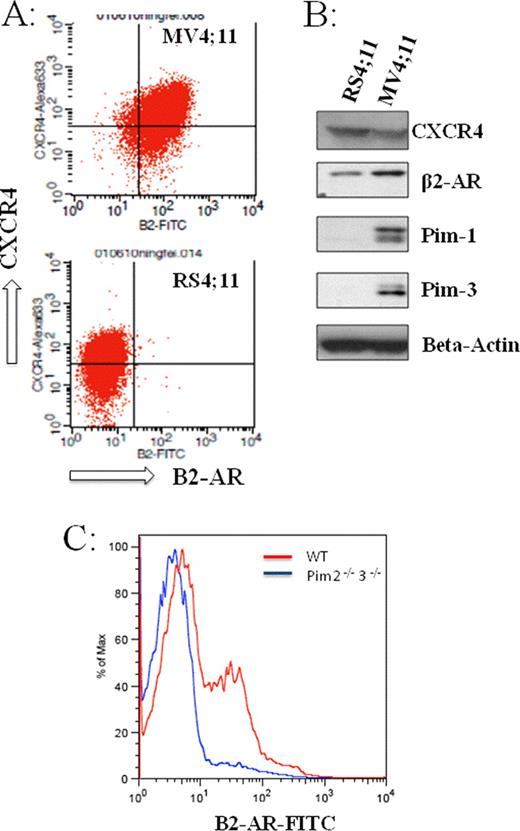
Acute myelogenous leukemia (AML) occurs with an incidence of 2.7 per 100,000 population in the year of 2009, and is associated with significant mortality and morbidity. Despite recent advances in molecular and cytogenetic analytic technologies, initial treatment for AML patients has remained essentially the same over the last 30 years; and the treatment outcome is dreadful with a 5-year survival rate of ∼25%. In an effort to gain a better understanding of AML cell biology and to develop more effective treatments for AML, we have been exploring the roles of the b2-adrenergic receptor (B2-AR) in AML. B2-AR is a G-protein- coupled catecholamine receptor and was recently found to play a direct role in the engraftment, proliferation and migration of hematopoietic stem cells [Spiegel, A., et al. Nat Immunol8, 1123–1131 (2007)]. However, very little is known about the roles of B2-AR in AML. We first screened a number of leukemia/hematopoietic cell lines, including K562, NBAL3, Jurkat, RPMI8226, U937, HEL, HL60, NB4, THP-1, and MV4;11, for B2-AR expression using flow cytometry. We found that B2-AR expression was not uniform in the leukemia/hematopoietic cell lines we tested. B2-AR was absent in acute non-myelogenous leukemia/hematopoietic cell lines such as K562, NBAL3, Jurkat or RPMI8226. In contrast, B2-AR was expressed on all acute myelogenous leukemia cell lines tested, with the highest expression on 2 myelomonocytic leukemia cell lines (THP-1 and MV4;11). We next examined the roles of B2-AR in leukemia cell survival, in leukemia cell responses to chemotherapy, and in leukemia cell migration in response to stromal derived factor-1 (SDF-1). We found that treatment with a B2-AR antagonist (ICI115881) modestly inhibited leukemia cell growth. Interestingly, treatment with a B2-AR agonist (i.e., isoproteronol or clenbuterol) inhibited leukemia cell migration to SDF-1. Additionally, combined treatment of MV4-11 cells with Isoproterenol and SDF-1 increased downstream ERK phosphorylation synergistically, suggesting a potential interaction or reciprocal regulation between B2-AR and CXCR4 receptor. To further understand the regulation and functional role of B2-AR in AML cells, we have been focusing on two biphenotypic leukemia cell lines, that is, MV4;11 and RS4;11 cells. Both MV4;11 and RS4;11 cells carry the t(4;11)(q21;q23) chromosomal translocation. While RS4;11 cells bear only the wild-type version of the Flt3 gene, MV4;11 cells express exclusively the mutated Flt3-ITD gene. Interestingly, we found that in contrast to MV4;11 leukemia cells, RS4;11 cells did not express surface B2-AR (panel A), although the total amount of B2-AR in the whole cell lysate was comparable between these two cell lines (panel B). This significantly different B2-AR expression pattern between these 2 cell lines may be related to the difference in the expression level of proviral insertion in murine lymphoma (Pim) kinases (panel B); RS4;11 has reduced/absent expression of Pim kinases compared to MV4;11 cells. Consistent with the potential regulation of B2-AR expression by Pim kinases, we found that Pim 2 and 3 double knockout mice had reduced B2-AR surface expression in peripheral blood mononucleated cells. Additional experiments are currently ongoing to further dissect the interaction between B2-AR and Pim kinases. Taken together, our current studies demonstrated a novel role of B2-AR in AML and a potential functional interaction between B2-AR, CXCR4, Pim kinase, and Flt3 gene.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal