Abstract
Abstract 1532
The current standard initial treatment for APL is all-trans retinoic acid (ATRA) and anthracycline with or without citarabine, followed by ATRA maintenance. Although the treatment has greatly improved clinical outcomes of APL, a considerable proportion of patients still undergo disease relapse, requiring salvage therapy with such as arsenic trioxide (ATO). GO, an anti-CD33 monoclonal antibody conjugated with calicheamicin, is also highly effective on APL. Highly homogeneous expression of CD33, potential efficacy of calicheamicin and lower expression of P-glycoprotein on APL cells explain the high potency of GO on APL. We report here on the efficacy of GO monotherapy against relapsed/refractory APL in a post-marketing surveillance study of GO in Japan.
All patients who were treated with GO alone were obligatorily registered into a post-marketing surveillance study run by Wyeth Japan, Co. Ltd. under the guidance of the Pharmaceutical and Medical Devices Agency. We obtained permission to access patients' data from each institution and Pfizer Japan, Co. Ltd. The survey items included adverse events, treatment outcome, overall survival (OS) and relapse-free survival (RFS). Clinical and biological characteristics were analyzed in APL and compared by chi-square test or Fisher's exact test for categorical data, and Wilcoxon rank-sum test for continuous data. OS and RFS were estimated by the Kaplan-Meier method in APL, and then compared to those from the simultaneous post-marketing study in acute myeloid leukemia (AML) by the log-rank test.
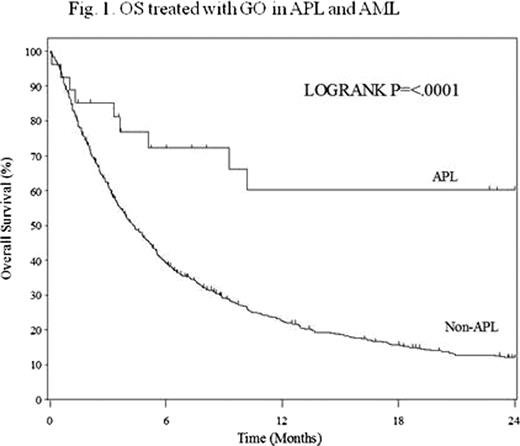
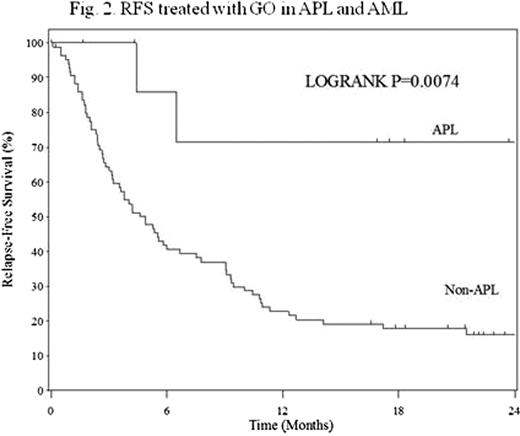
Between 2005 and 2008, 27 relapsed/refractory APL patients were enrolled in this surveillance. Of these, two patients were excluded from analysis because of insufficient data, while 726 AML patients were enrolled and 503 were evaluable. Twelve (48%) and 2 (9%) APL patients who were treated with GO alone achieved CR and CRp, respectively, while 42 (8%) and 41 (8%) of AML achieved CR and CRp, respectively. Remission duration of first CR and number of relapses influenced CR rates, but previous usage of ATO and age did not in APL. OS and RFS at 2 years were 63% and 71% in APL, and 14% and 20% in AML, respectively. These were shown in figures. OS and RFS in APL were better than those in AML (P<0.0001 and P=0.0074, respectively). Treatment related adverse events of GO (grade 3 or 4) in APL patients were infection (24%), bleeding (8%), lung damage (4%) and reversible veno-occlusive disease (4%); these were similar in AML patients.
Although other previous clinical studies failed to confirm clinical benefit of GO on AML, single use of GO is highly effective and relatively safe in relapsed/refractory APL. The efficacy was also observed in elderly patients and in relapsed ones after ATO therapy. Further studies are warranted, in which GO is included in addition to ATRA and ATO in the treatment of high-risk APL.
Takeshita:Novartis Pharma: Research Funding; Takeda Pharma: Research Funding. Naoe:Kyowa-Hakko Kirin.: Research Funding; Dainipponn-Sumitomo Pharma.: Research Funding; Chugai Pharma.: Research Funding; Novartis Pharma.: Honoraria, Speakers Bureau; Zenyaku-Kogyo: Research Funding; Otsuka Pharma.: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal