Abstract
Abstract 2352
The administration of highly purified haploidentical peripheral blood CD34+ cells combined with an unrelated cord blood transplant results in earlier neutrophil engraftment than is typically seen with a cord blood transplant alone. Chimerism data from pilot trials evaluating this strategy have reported 3 phases of engraftment: 1) early myeloid engraftment from transplanted haplo-CD34+ cells followed by 2) cord blood engraftment resulting in dual chimerism and 3) the subsequent disappearance of haploidentical donor cells with resultant full donor cord chimerism. The mechanism accounting for the disappearance of haploidentical cells has not been defined. Here the clinical results and an in vitro assessment of alloreactivity in three patients that underwent combined haploidentical CD34+ cell and cord blood transplantation for severe aplastic anemia (SAA) are described. The conditioning regimen consisted of cyclophosphamide (60mg/kg/day on days -7 and -6), fludarabine (25mg/m2/day on days -5 to -1), horse ATG (40mg/kg/day on days -5 to -2), and total body irradiation (200cGy on day -1). GVHD prophylaxis consisted of tacrolimus and mycophenolate mofetil. PCR of STRs was used to assess chimerism in T-cell and myeloid lineages and mixed lymphocyte reaction assays(MLR) were performed on peripheral blood samples collected at different time-points post-transplant to assess for alloreactivity against the recipient, the haploidentical donor, or the cord unit. Stimulator cord blood cells for the MLR were obtained from residual cord blood cells remaining in the infusion bag after patient administration and expanded in vitro using anti-CD28/CD3 Dynabeads.
Prior to transplantation, all three pts had transfusion dependent SAA associated with severe neutropenia that was refractory to conventional immunosuppressive therapy. Pt 1 had an early transient myeloid recovery (ANC 400 on day+11) from the haploidentical donor followed by engraftment of the cord unit (Cord ANC > 500) on day 21. The patient is currently 2 years post transplant and has 100% cord blood chimerism and is transfusion independent. An MLR assay performed when donor T-cell chimerism was 100% cord, showed evidence for rejection of the haploid cells by cord blood T-cells, with the MLR response to haploidentical donor cells being seven fold higher than the response to fully HLA-mismatched 3rd party cells. In pt 2, neutrophil recovery from the transplanted haploidentical donor occurred on day +10, with chimerism studies showing no evidence for cord engraftment in either myeloid or T-cell lineages at any point post-transplant. The patient is currently 15 months post transplant and is transfusion independent with normal blood counts and sustained “split” chimerism (T-cells recipient in origin with myeloid cells being 100% haploidentical donor). MLR assays showed that the recipient was tolerant to the haploid donor, with no statistically significant difference in the alloreactive response to the haploid donor compared to self. In pt 3, neutrophil recovery from the transplanted haploidentical donor occurred on day +10, with chimerism studies showing split chimerism (T-cell chimerism >90% cord and myeloid chimerism 88–100% haploid donor in origin). MLR assays again showed evidence of rejection of the haploid cells by cord blood T-cells, with a trend towards greater alloreactivity against the haploid donor compared to an HLA mismatched 3rd party on post-transplant day +63.
Combined haploidentical CD34+ cell and unrelated cord blood transplantation following highly immunosuppressive conditioning represents a viable treatment option for patients with SAA who lack an HLA-matched donor. Using this approach, 2 of 3 pts had cord blood engraftment associated with early neutrophil recovery from the haploidentical donor. In one pt, the cord unit failed to engraft. Remarkably, sustained engraftment from the haploidentical donor in this pt resulted in transfusion independence. MLR appears to be a useful approach to assess the in vitro alloreactivity of this unique stem cell graft source. In the two pts who had cord engraftment, in vitro MLR assessments established that the disappearance of haploid cells occurred as a consequence of rejection of the haploidentical cells by engrafting cord blood T-cells, rather than from non-immunological haploidentical cell graft failure.
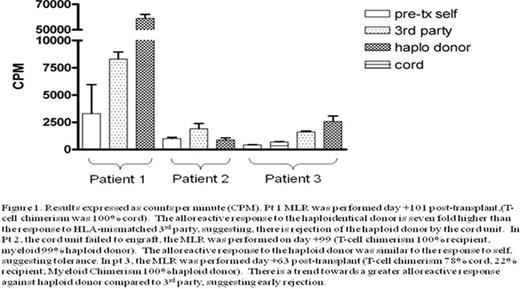
Results expressed as countsper minute (CPM). Pt 1 MLR was performed day + 101 post-transplant. (T-cell chemerism was 100% cord). The alloreactiveresponse to the haploidentical donor is seven fold higher than the response to HLA-mismatched 3rd party, suggesting, there is rejecttion of the haploid donor by the cordunit. In Pt 2. the cord unit failed to engraft, the MLR was performed on day +99 (T-cell chimerism 100% recipient, myeloid 99% hapolied donor). The alloreactiver esponse to the haploid donor was similar to the response to self. suggestomg tolerance. In pt 3. thee MLR was performed day +63 post-transplant (T-cell chimerism 79% cord. 22% recipient; Myeloid Chimerism 100% haploid donor). There is a trend to wards a greater alloreactive response against haploid donor compared to 3rd party, suggesting early rejection.
Results expressed as countsper minute (CPM). Pt 1 MLR was performed day + 101 post-transplant. (T-cell chemerism was 100% cord). The alloreactiveresponse to the haploidentical donor is seven fold higher than the response to HLA-mismatched 3rd party, suggesting, there is rejecttion of the haploid donor by the cordunit. In Pt 2. the cord unit failed to engraft, the MLR was performed on day +99 (T-cell chimerism 100% recipient, myeloid 99% hapolied donor). The alloreactiver esponse to the haploid donor was similar to the response to self. suggestomg tolerance. In pt 3. thee MLR was performed day +63 post-transplant (T-cell chimerism 79% cord. 22% recipient; Myeloid Chimerism 100% haploid donor). There is a trend to wards a greater alloreactive response against haploid donor compared to 3rd party, suggesting early rejection.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal