Abstract
Abstract 1958
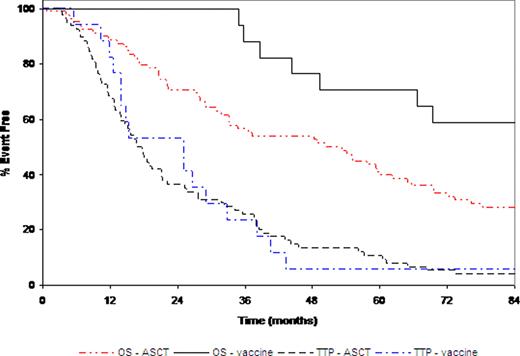
Vaccines may offer additional benefit as consolidation therapy after plateau phase or autologous stem cell transplant (ASCT) for multiple myeloma (MM). We have reported previously the results of another phase II trial showing that using APC8020 (Mylovenge™) after ASCT improved the overall survival (OS 5.3 yrs, 95% CI 4.0 yrs – NA) compared to ASCT alone (3.4 yrs, 95% CI 2.7 – 4.6 yrs; p = 0.02; Lacy, AJH (84): 799). We now report the results from a randomized, phase II trial using the same vaccine plus adjuvant cytokines in MM patients in plateau phase after either chemotherapy or ASCT. Twenty patients were enrolled between 2001 and 2003 and were randomized to receive APC8020 and adjuvant cytokine of either interferon-g (IFNg, arm A, n = 10) or interleukin-2 (IL2, arm B, n = 10). Each cycle of treatment included five days of daily subcutaneous injections of either IFNg (106 IU, arm A) or IL2 (200 mg, arm B) followed by APC8020 injection. Each patient received a cycle of treatment every 2 weeks for 4 cycles. One patient in each arm was in plateau phase post-chemotherapy. Time from diagnosis to treatment was 11.9 mos for the patient in arm A and 16.2 mos for arm B. Time to progression (TTP) was 3.7 and 68 months respectively the patient in arm A and B. Both patients were alive at five years. For the 9 post-ASCT patients in each arm, no statistically significant differences were seen in the baseline characteristics between the 2 arms. The median time from diagnosis to treatment was 15.4 months. No grade 4 or 5 adverse events (AEs) were reported. Two patients in each arm had grade 3 AE, including lymphopenia, thrombocytopenia, hemorrhage, infection, and fatigue. One patient in arm B had grade 3 autoimmune disorder that was deemed possibly related to study treatment. The most common AEs were grade 1 fatigue (n = 7) and anemia (n = 5) in arm A and grade 1 anemia (n = 7) and injection site reaction (n = 5) in arm B. The median TTP was 9.3 months (95% CI: 3.9 – 19.3 months) for arm A and 6.6 months for arm B (95% CI: 6.3 – 29.6 months) with no statistically significant difference between the two arms (p = 0.89). The median OS had not been reached for either arm; the 5-year OS rate was 67% and 56% for arms A and B respectively (p = 1.0). Interestingly, similar to our previous study where TTP was not different while OS was improved with vaccine, when we compared the clinical outcome for all patients in this vaccine trial to that of 78 matched control patients who only received ASCT during the same time frame, we did not see significant improvement in TTP (Figure 1). However, the 5-year overall survival for all patients in this trial is significantly improved at 71% (95%CI: 51–96%) compared to ASCT control patients (41%, 95% CI: 31 – 54%; p = 0.03; Figure 1). This trial suggests improved OS with vaccine plus cytokine. It is possible that modulation of patients’ immunity may help them live longer with multiple myeloma. Further investigation is needed to compare the approach in this trial with dendritic cell vaccine alone to better understand optimal strategy for vaccine therapy in MM.
Dispenzieri:Celgene: Honoraria, Research Funding; Binding Site: Honoraria. Kumar:Celgene: Consultancy, Research Funding; Millennium: Research Funding; Merck: Consultancy, Research Funding; Novartis: Research Funding; Genzyme: Consultancy, Research Funding; Cephalon: Research Funding. Lacy:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal