Abstract
BACKGROUND: The role of rituximab (R) as a component of therapy for relapsed and refractory diffuse large B cell lymphoma (DLBCL) is controversial. We have previously reported that the addition of rituximab to ICE (RICE) second-line chemotherapy improved outcomes (Kewalramani, 2004). Furthermore, a small phase II study performed at Stanford University (Horwitz et al, 2004) suggested improvement in event free survival (EFS) and overall survival (OS) with the addition of rituximab maintenance therapy following high dose therapy and autologous stem cell rescue (HDT/ASCR) in rituximab naïve patients. However, it is unknown if there is benefit of rituximab maintenace in patients (pts) receiving rituximab as part of pre-HDT cytoreduction.
METHODS: We retrospectively identified pts with relapsed and refractory DLBCL who underwent uniform pre-HDT/ASCR cytoreduction with RICE between 1999 and 2006. Pts proceeded to HDT/ASCR if they had a response to RICE chemotherapy. The earlier pts (n=38) did not receive any maintenance (NM) and a subsequent cohort (n=43) was treated with post ASCR rituximab (MR) on one of two regimens (R wkly x 4 at day + 42 and day + 180, n=26 or R q8 wks x 6 starting on day+29, n=17).
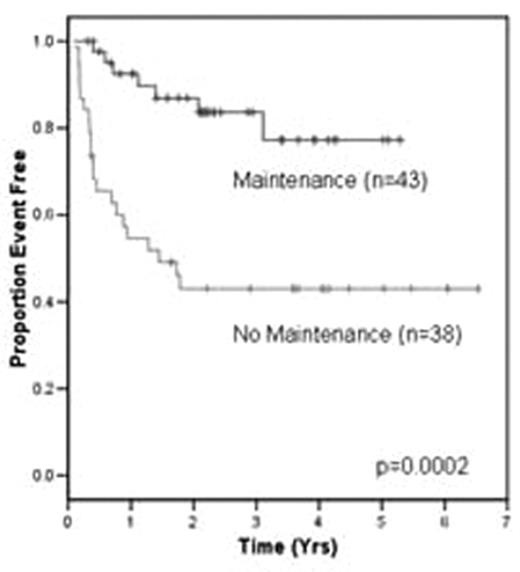
RESULTS: Median follow up for surviving pts is 2.9 yrs: 4 yrs in the NM group; and 2.2 yrs in the MR group. The 3 yrs EFS is 43% and 84% and OS is 59% and 89% for the NM and MR groups, respectively. A Cox regression model was constructed to include disease status (relapsed v refractory), maintenance rituximab (yes v no) and second-line IPI (LR v LI-HR). Disease status (p = .04) and rituximab maintenance (p = .003) were significant for OS. Only maintenance rituximab was significant for EFS (p < .001) (Figure 1). In analyzing patients who did not receive maintenance therapy by second-line IPI low risk (0–1) and low-intermediate risk-high risk (2–5), as expected, there is a survival difference which approaches significance (p = 0.074) in favor of the low risk group. Maintenance rituximab eliminated the adverse impact of the second-line IPI on EFS and OS for pts with LI to HR disease. For pts with LR disease maintenance rituximab did not influence outcome. It has been previously reported that patients who receive post-transplant rituximab maintenance experience high rates of neutropenia. We noted 16 patients who experienced asymptomatic afebrile neutropenia (Grade III/IV). One pt (2.3%) required hospital readmission post transplant related to fever and neutropenia. Neutropenia was noted early in the treatment course of both regimens. Growth factors were used at the discretion of the physician.
CONCLUSION: Maintenance rituximab therapy post HDT/ASCR was well tolerated by patients and in this small sample significantly associated with improvement in EFS and OS. A prospective trial is currently underway to fully evaluate maintenance therapy versus observation (CORAL 50-03B).
Figure
Patient characteristics
| . | . | RICE Alone . | RICE + Maintenance . |
|---|---|---|---|
| Total | 38 | 43 | |
| Male | 23 | 22 | |
| Female | 15 | 21 | |
| Median Age (range) | 52 (26–73) | 48 (22–69) | |
| Disease Status | |||
| Relapsed | 27 | 18 | |
| Refractory | 11 | 25 | |
| IPI | |||
| LR | 11 | 24 | |
| LIR-HR | 27 | 19 |
| . | . | RICE Alone . | RICE + Maintenance . |
|---|---|---|---|
| Total | 38 | 43 | |
| Male | 23 | 22 | |
| Female | 15 | 21 | |
| Median Age (range) | 52 (26–73) | 48 (22–69) | |
| Disease Status | |||
| Relapsed | 27 | 18 | |
| Refractory | 11 | 25 | |
| IPI | |||
| LR | 11 | 24 | |
| LIR-HR | 27 | 19 |
Author notes
Disclosure:Consultancy: Dr. Andrew Zelenetz has consulted for GSK; Favril; Novartis; and sanofi-aventis. Research Funding: Dr. Craig Moskowitz and Dr. Andrew Zelenetz have research funding from Genentech, Inc. Dr. Andrew Zelenetz has research funding from Biogen Idec and GSK. Off Label Use: Rituximab use for patients with relapsed or refractory diffuse large B cell lymphoma is outside of the approved indication

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal