Comment on Groot et al, page 1957
In this issue of Blood, Groot and colleagues clarify the function of the 2 major types of dendritic cells in the transmission, propagation, and—importantly—the inhibition of infection with HIV-1.
Groot and colleagues have identified a new small anti-HIV factor secreted by one of the 2 major types of dendritic cells (DCs), adding to the armamentarium of natural antiviral compounds. Bench-to-bedside drug discovery programs have yet to take advantage of these innate constitutive and/or inducible molecules, despite their obvious attractiveness in terms of presumed efficacy and toxicity profiles.
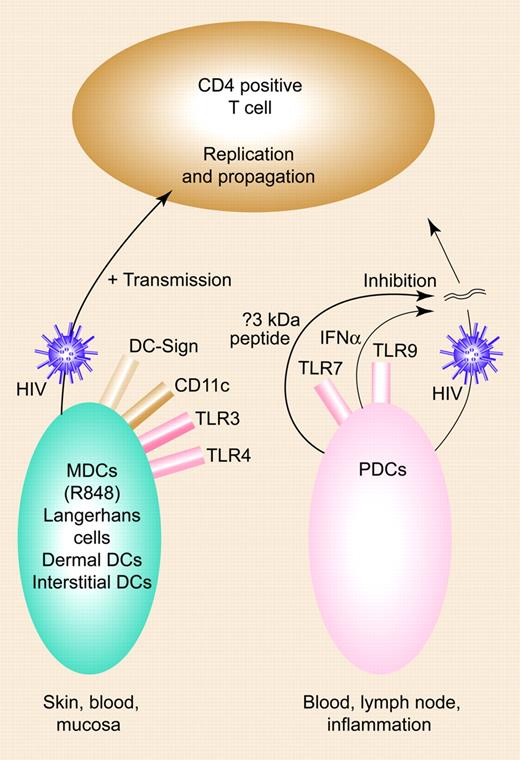
Previous data have shown that during HIV infection both myeloid DCs (MDCs; interleukin-12 [IL-12]–producing) and plasmacytoid DCs (PDC; interferon-α [IFN-α–producing) are reduced in number,1 that their function is impaired, and that through C-type lectins such as DC-specific intercellular adhesion molecule 3–grabbing nonintegrin (DC-SIGN) they capture and transmit the virus at the DC–T-cell interface, the infectious synapse.2,3
This study, which uses isolated donor-matched DCs rather than monocyte-derived cells, adds a number of new observations with functional consequences. MDCs enhance and PDCs inhibit HIV replication in T cells, a process mediated by different mechanisms for each DC subset: (1) MDCs actively transmit HIV to T cells, and factors that are secreted during MDC maturation or after T-cell encounter probably do not influence this process; and (2) secreted factors during PDC activation were completely responsible for the inhibition of HIV-1 infection of T cells. Here, PDC activation appears entirely responsible for the inhibition of HIV-1 replication.FIG1
Opposing roles of DC subsets in HIV infection. MDCs enhance HIV-1, with transmission of HIV-1 across the infectious synapse; PDCs inhibit this process via IFN-α and a new small molecule. Illustration by Paulette Dennis.
Opposing roles of DC subsets in HIV infection. MDCs enhance HIV-1, with transmission of HIV-1 across the infectious synapse; PDCs inhibit this process via IFN-α and a new small molecule. Illustration by Paulette Dennis.
MDCs, on the other hand, have differing transmission efficiencies (R-848 being the most superior), and this is not due to differences in expression of integrins (ICAM-1, -2, -3, or LFA-1), CD83/CD86 expression, or the amount of HIV capture. Furthermore, PDCs secrete high amounts of IFN-α, which inhibits HIV replication, but most interestingly, they secrete a small molecule less than 3 kDa in size that inhibits HIV replication. Since it is sensitive to heat, this molecule is likely to be a polypeptide. This molecule was found by passing supernatants through centrifugal filters and testing their capacity to inhibit HIV-1 infection in a single-cycle replication assay with LuSIV cells. The smallest fraction contained no IFN-α, and the addition of neutralizing antibodies against type 1 IFNs did not change inhibition.
Not only do Groot and colleagues discuss opposing roles of CD11c-positive and -negative DCs, their discovery of a novel, small, naturally occurring host molecule might lead to the discovery of a new unknown anti-HIV compound. While cause and effect remains to be established (is the molecule secreted as a consequence of HIV infection or is it constitutively produced?), it is notable that the passions and polemic surrounding similar discoveries in the past have not yet led to the identity of some naturally occurring anti-HIV secreted factors4 (though they tend to have a much higher molecular weight than the compound here). However, this is unquestionably a worthwhile endeavor. We learn an enormous amount from individuals and animals that become infected and do not develop uncontrollable viremia with suppression of CD4+ T cells.5 One appropriate step would be to identify this small molecule and determine its role in the long-term nonprogressor process. In addition, there are probably enough data now for a randomised DC-based therapeutic vaccine trial in HIV. Based on the paper herein by Groot et al, this should be based on the PDC model. ▪

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal