Abstract
Infantile hemangiomas are composed of endothelial cells (ECs), endothelial progenitor cells (EPCs), as well as perivascular and hematopoietic cells. Our hypothesis is that hemangioma-derived EPCs (HemEPCs) differentiate into the mature ECs that comprise the major compartment of the tumor. To test this, we isolated EPCs (CD133+/Ulex europeus– I+) and mature ECs (CD133–/Ulex europeus–I+) from proliferating hemangiomas and used a previously described property of hemangioma-derived ECs (HemECs), enhanced migratory activity in response to the angiogenesis inhibitor endostatin, to determine if HemEPCs share this abnormal behavior. Umbilical cord blood–derived EPCs (cbEPCs) were analyzed in parallel as a normal control. Our results show that HemEPCs, HemECs, and cbEPCs exhibit increased adhesion, migration, and proliferation in response to endostatin. This angiogenic response to endostatin was consistently expressed by HemEPCs over several weeks in culture, whereas HemECs and cbEPCs shifted toward the mature endothelial response to endostatin. Similar mRNA-expression patterns among HemEPCs, HemECs, and cbEPCs, revealed by microarray analyses, provided further indication of an EPC phenotype. This is the first demonstration that human EPCs, isolated from blood or from a proliferating hemangioma, are stimulated by an angiogenesis inhibitor. These findings suggest that EPCs respond differently from mature ECs when exposed to angiogenic or antiangiogenic signals.
Introduction
Infantile hemangioma, the most common tumor of infancy, is characterized by rapid proliferation of the endothelial cells followed by slow spontaneous involution.1 This transition from the proliferating phase to the involuting phase represents a gradient of cellular activity from unregulated proliferation to apoptosis. The molecular events that control the evolution of hemangioma remain obscure. Both intrinsic and extrinsic anomalies have been suggested to underlie the primary defect.2-6 Recent studies showing that hemangioma-derived endothelial cells (HemECs) are clonal provide support for an intrinsic defect.4,6 We and others have identified endothelial progenitor cells (EPCs) in hemangioma tissue and circulating blood of patients with hemangiomas, respectively.7,8 These findings suggest that a hemangioma may arise from the clonal expansion of an EPC.
An angioblastic origin for hemangiomas has been envisioned for over a century. In 1863, Virchow9 questioned whether hemangiomas represented sequestered embryonic mesoderm. Pack and Miller10 suggested that hemangiomas arise from the localized growth of angioblastic cells. Similarly, Malan11 imagined hemangiomas as an activation of dormant angioblasts. Histologic examination of hemangioma ECs reveals an immature phenotype with large nuclei and scant cytoplasm.12 During the rapid growth phase of hemangiomas, syncytial EC hyperplasia, with and without lumens, is seen. In the involuting phase, hyperplasia diminishes and mature vascular channels with defined lumens become prominent.1 Cultured cells from proliferating-phase hemangiomas resemble fetal ECs more than neonatal ECs.13 The diffuse cytoplasmic localization of CD31 also suggests an immature/precursor state of the ECs isolated from hemangiomas.13
Our working hypothesis is that hemangiomas are composed of proliferating angioblasts that arise from a single progenitor and the involuting phase represents a delayed and possibly defective differentiation path to mature endothelium. If so, EPCs isolated from the hemangioma tissues (HemEPCs) should exhibit similar aberrant properties in vitro when compared with HemECs. We demonstrated previously that HemECs display increased vascular endothelial growth factor (VEGF)–induced migration in response to the angiogenesis inhibitor endostatin (ES); this is in direct contrast to the inhibitory response of human dermal microvascular endothelial cells (HDMECs) from newborn foreskin.4 In this study, we investigated whether EPCs derived from hemangioma exhibit the same altered cellular behavior in response to ES.
Materials and methods
Isolation and culture of EPCs and ECs
Hemangioma specimens were obtained with approval of the Committee on Clinical Investigation, Children's Hospital Boston. The clinical diagnosis was confirmed in the Department of Pathology, Children's Hospital Boston. Neonatal foreskin and umbilical cord blood were obtained with institutional review board (IRB) approval from the Brigham and Women's Hospital (Boston, MA). HemEPCs and mature HemECs were isolated from a proliferating-phase specimen (Hem88; 7-month-old female, untreated) by modification of the method described previously.4,14 The tissue was digested with 0.2% collagenase A (Roche Diagnostics, Indianapolis, IN) in Dulbecco modified Eagle medium (Invitrogen, Carlsbad, CA) supplemented with 2% FBS, Ca++Mg++ (1.26 mM calcium chloride/0.93 mM magnesium sulfate), and PSF (Invitrogen; 100 U/mL penicillin, 100 μg/mL streptomycin, 0.25 μg/mL amphotericin) for 30 minutes at 37°C. The tissue homogenate was filtered through a 100-μm cell strainer (Fisher Scientific, Hampton, NH). Red blood cells (RBCs) were lysed by incubating the sample in NH4Cl (0.8% NH4Cl/0.1 mM EDTA; StemCell Technologies, Vancouver, BC, Canada) for 10 minutes on ice. Cells were filtered through a 40-μm strainer to obtain a single-cell suspension from which the cells expressing the stem-cell antigen CD133 were selected. CD133+ and CD133– cells were isolated using anti-CD133–coated magnetic beads (Miltenyi Biotec, Auburn, CA) and cultured separately on fibronectin (FN; 1 μg/cm2) coated plates with Endothelial Basal Medium (EBM-2; CC-3156; Cambrex Bio Science, Walkersville, MD). EBM-2 was supplemented with 20% FBS, SingleQuot (CC-4176; Cambrex Bio Science; contains human epidermal growth factor, VEGF, human basic fibroblast growth factor, insulin-like growth factor, hydrocortisone, heparin, ascorbic acid, and gentamicin/amphotericin B), and PSF. Hereafter this supplemented medium is called EBM-2/20% FBS. To purify the endothelial cells, subconfluent CD133+ and CD133– cells were trypsizined and incubated with Ulex europaeus–I (Ulex) lectin-coated beads. HemEPCs (CD133+Ulex+) and mature HemECs (CD133–Ulex+) were cultured and expanded in the EBM-2/20% FBS on FN-coated plates as described for CD133+ cells. Hence, HemEPCs were so designated based on expression of the stem-cell marker CD133, which is downregulated quickly as the cells are cultured in vitro.15
Human dermal microvascular ECs (HDMECs) and cord-blood EPCs (cbEPCs) were isolated, as described previously, but cultured in EBM-2/20% FBS as noted above.14,15 In addition, cbEPCs were isolated by a non–CD133-dependent modification described by Lin et al,16 which takes advantage of the high proliferative potential of EPCs. In brief, mononuclear cells were seeded on 1% gelatin–coated tissue culture plates for 48 hours. Unbound cells were removed following the 48 hours of culture and the bound fraction was cultured for another 2 weeks in EBM-2/20% FBS. Colonies of endothelial-like cells were trypsinized and then selected using either Ulex- or CD31-coated magnetic beads (Dynal Biotech, Brown Deer, WI). Similar results were obtained with cord-blood EPCs isolated by CD133 selection,15 CD31 selection, or Ulex selection. For long-term studies, cells were cultured in 100 × 20-mm Falcon (Heidelberg, Germany) culture plates; passaged 1:3; and examined at passages 6, 12, 18, and 24. Consistent cell-culture conditions were maintained for all endothelial preparations.
Endothelial characterization
Flow cytometry. EPCs and ECs were labeled with fluorescein isothiocyanate (FITC)–conjugated mouse anti–human CD31 (Ancell, Bayport, MN), FITC-mouse anti–human CD146 (Chemicon International, Temecula, CA), phycoerythrin (PE)–conjugated mouse anti–human CD34 (Miltenyi Biotec), PE-mouse anti–human VEGF-R2 (R&D Systems, Minneapolis, MN), FITC-mouse anti–human CD45 (BD PharMingen, San Jose, CA), and FITC-mouse anti–human CD14 (BD PharMingen) antibodies. CD34 and VEGF-R2 antibody incubation was carried out following permeabilization of cells with 0.5% saponin. The cells were fixed with 4% paraformaldehyde and permeabilized with saponin for 10 minutes. Antibody labeling was carried out for 20 minutes on ice followed by 3 washes with PBS, 1% BSA, and 0.2 mM EDTA.
Indirect immunofluorescence. Immunofluorescent analysis of cells was carried out with goat anti–human CD31 (Santa Cruz Biotechnology, Santa Cruz, CA), mouse anti–human VE-cadherin (Immunotech, Westbrook, ME), and rabbit anti–human von Willebrand factor (VWF; DakoCytomation, Carpinteria, CA). FITC-conjugated secondary antibodies (Vector Laboratories, Burlingame, CA) were used for detection.
RT-PCR. Total RNA was isolated from cultured EPCs and ECs using RNeasy Mini Kit (Qiagen, Valencia, CA). Purity of the RNA samples was assessed by determining the optical density at 260:280 nm. Three micrograms of total RNA was used for cDNA synthesis with iScript cDNA Synthesis Kit (Bio-Rad Laboratories, Hercules, CA). The reverse transcriptase–polymerase chain reaction (RT-PCR) mixture (20 μL volume) consisted of 15 μL Platinum Taq Supermix (Invitrogen), 1 μL of each forward and reverse 10-μM primers (Table 1), 2 μL cDNA template, and 1 μLH2O. The reaction was performed for 30 cycles with the following temperature profile: 95°C for 3 minutes (initiation; 30 seconds/cycle thereafter), 55°C for 30 seconds, and 72°C for 45 seconds. Ten microliters of the RT-PCR products were run on a 3% agarose gel.
Oligonucleotide primer sequences for RT-PCR
Gene . | Primers, 5′→3′ . | Amplicon, bp . | Reference . |
|---|---|---|---|
| CD31 | TCCCACGCCAAAATGTTAAGTGAG, GATCAAGAGAGCAATGATCACTCC | 405 | Unger et al17 |
| CD34 | GCGCTTTGCTTGCTGAGTTTGC, GCCTCATTGCCATGTTGAGACAC | 191 | Unger et al17 |
| VEGF-R2 | TCAAAGGAGAAGCAGAGCCATGTG, GCACTCTTCCTCCAACTGCCAATA | 186 | Yu et al18 |
| VWF | CACCGTTTGCCCACCCTTCG, GCCCACTGGGAGCCGACACT | 435 | Murga et al19 |
| VE-cadherin | CCTTGGGTCCTGAAGTGACCT, CAGGGCCTTCCTTCTGCAA | 512 | — |
| S9 | GATGAGAAGGACCCACGGCGTCTGTTCG, GAGACAATCCAGCAGCCCAGGAGGGACA | 431 | BD Biosciences PT3391-1 |
Gene . | Primers, 5′→3′ . | Amplicon, bp . | Reference . |
|---|---|---|---|
| CD31 | TCCCACGCCAAAATGTTAAGTGAG, GATCAAGAGAGCAATGATCACTCC | 405 | Unger et al17 |
| CD34 | GCGCTTTGCTTGCTGAGTTTGC, GCCTCATTGCCATGTTGAGACAC | 191 | Unger et al17 |
| VEGF-R2 | TCAAAGGAGAAGCAGAGCCATGTG, GCACTCTTCCTCCAACTGCCAATA | 186 | Yu et al18 |
| VWF | CACCGTTTGCCCACCCTTCG, GCCCACTGGGAGCCGACACT | 435 | Murga et al19 |
| VE-cadherin | CCTTGGGTCCTGAAGTGACCT, CAGGGCCTTCCTTCTGCAA | 512 | — |
| S9 | GATGAGAAGGACCCACGGCGTCTGTTCG, GAGACAATCCAGCAGCCCAGGAGGGACA | 431 | BD Biosciences PT3391-1 |
— indicates not previously published.
Endostatin experiments
Endostatin experiments were conducted on cells that were growth factor and serum starved for 6 hours prior to the assays.
Adhesion assay. Adhesion of cells was analyzed on 48-well culture plates coated with 0.5% BSA (negative control) or 50 μg/cm2 FN overnight at 37°C. Trypsinized cells were counted and seeded at a density of 5000 cells/cm2. Four treatments, in serum-free media, were tested (control, 10 ng/mL ES, 5 ng/mL VEGF [R&D Systems], and 5 ng/mL VEGF + 10 ng/mL ES). Human recombinant ES was obtained from EntreMed (Rockville, MD) and was provided to us by Dr Kashi Javaherian, Children's Hospital Boston. ES pretreatment of the cells was carried out for 30 minutes before seeding the cells with or without VEGF. After 30 minutes at 37°C, the wells were washed twice with PBS to remove nonadherent cells. The attached cells were counted and expressed, relative to control cells.
Migration assay. Cellular migration was assayed as described.4 Rat tail collagen type I (100 μg/mL; 0.02 N acetic acid) was used to coat 6.5-mm Transwells with 8.0-μm-pore polycarbonate membrane inserts (Corning Life Sciences, Acton, MA) for 24 hours at 37°C, followed by one wash with PBS. Cells were trypsinized and counted. ES pretreatment of cells (10 ng/mL) was performed in suspension for 30 minutes.4 For dose-response experiments, ES pretreatment was performed with 1 ng/mL, 10 ng/mL, 100 ng/mL, 1 μg/mL, and 10 μg/mL ES. Cells were seeded in the upper chamber of the transwell plate at a density of 10 000/well (100 μL volume). Control media (600 μL; EBM-2/serum and growth factor free) or media with VEGF (5 ng/mL) was added to the lower chamber. Cells were then allowed to migrate for 4 hours at 37°C. The cells in the upper chamber were gently scraped using a cotton swab. Chilled methanol was added to the lower chamber and the cells on the underside of the insert were fixed. Cells were visualized by DAPI fluorescence (Vector Laboratories) and counted. Migration assays were performed on cells cultured for 6, 12, 18, and 24 passages to determine how the VEGF-induced migratory activity was affected by long-term culture.
Proliferation assay. To determine the effect of ES on cellular proliferation, ECs were seeded on 1 μg/cm2 FN-coated 48-well plates and assayed for cell number following 24 hours of treatment. Exposure to ES was tested at 2 time points: (1) ES treatment initiated at the time of seeding, and (2) ES treatment initiated after allowing cells to attach in the presence of 2% serum for 30 minutes. Following attachment, media with 2% serum was removed and the cells were covered with basal serum-free media, 10 ng/mL ES, 5 ng/mL VEGF, or 10 ng/mL ES + 5 ng/mL VEGF. Cells were incubated for 24 hours and washed twice with PBS, counted, and expressed relative to control.
Phospho-protein profiling. Cells were grown to confluence on FN-coated 100 × 20-mm Falcon tissue culture plates. Whole-cell extracts were prepared with 500 mM Tris (pH 7.5), 4 M urea, 5 mM EDTA, 0.5% SDS, and 0.5% nonidet P40 with protease inhibitor cocktail (Roche Diagnostics). Protein concentrations were determined by Bio-Rad Protein Assay (Bio-Rad Laboratories). Custom antibody arrays (Hypromatrix, Worcester, MA; Table 2) were blocked for 1 hour at room temperature with 5% dry milk in TBST (150 mM NaCl; 25 mM Tris; 0.05% Tween-20, pH 7.5). Arrays were then incubated with 500 μL (0.5 μg/μL) of cell extracts for 2 hours at room temperature. The arrays were washed with TBST and incubated with horseradish peroxidase (HRP)–conjugated antiphosphotyrosine or antiphosphoserine antibodies (Hyperpmatrix) for 2 hours. ECL Plus Substrate (Amersham Biosciences, Piscataway, NJ) was used for detection. Phosphorylation levels were quantitated by densitometry using Image J software (http://rsb.info.nih.gov/ij/).
Integrin phosphorylation levels: ratio of HemEPCs/HDMECs
. | Control . | . | ES . | . | ||
|---|---|---|---|---|---|---|
| Integrin . | Tyrosine . | Serine . | Tyrosine . | Serine . | ||
| α1 | 0.12 | 0.11 | 0.14 | 0.17 | ||
| α5 | 1.89* | 2.52* | 1.53* | 2.31* | ||
| αv | 0.86 | 0.38 | 0.94 | 0.31 | ||
| β1 | 0.10 | 4.61* | 0.08 | 5.98* | ||
| β3 | 0.29 | 0.32 | 0.26 | 0.42 | ||
. | Control . | . | ES . | . | ||
|---|---|---|---|---|---|---|
| Integrin . | Tyrosine . | Serine . | Tyrosine . | Serine . | ||
| α1 | 0.12 | 0.11 | 0.14 | 0.17 | ||
| α5 | 1.89* | 2.52* | 1.53* | 2.31* | ||
| αv | 0.86 | 0.38 | 0.94 | 0.31 | ||
| β1 | 0.10 | 4.61* | 0.08 | 5.98* | ||
| β3 | 0.29 | 0.32 | 0.26 | 0.42 | ||
Higher phosphorylation in HemEPCs.
E-selectin expression. Endothelial cells were treated with TNF-α (10 ng/mL) or ES (10 ng/mL) for 5 hours, removed from the culture plates with trypsin, incubated with anti–human E-selectin monoclonal antibody (mAb; Clone 5G11),20 and analyzed by flow cytometry.
Oligo microarrays
cDNA from endothelial cells was used for synthesis and amplification of biotin-16-uridine-5′-triphosphate (Roche Diagnostics) labeled cRNA with TrueLabeling-AMP Kit (Superarray Bioscience, Frederick, MD). A total of 10 μg biotin-labeled cRNA was hybridized to the oligo arrays (OHS-013; 113 genes encoding matrix proteins and proteins involved in cell-cell, cell-tissue, and cell-matrix interactions; Superarray Biosciences) for 24 hours. Chemiluminescent detection was performed with alkaline phosphate–conjugated streptavidin and CDP-Star chemiluminescent substrate (Superarray Bioscience). Data were analyzed with GEArray Expression Analysis Suite (Superarray Bioscience).
Statistical analysis
The data were expressed as means ± SEM and analyzed by analysis of variance (ANOVA) followed by Student t test. Differences were considered significant at P values of less than .05.
Results
Isolation and characterization of EPCs and ECs
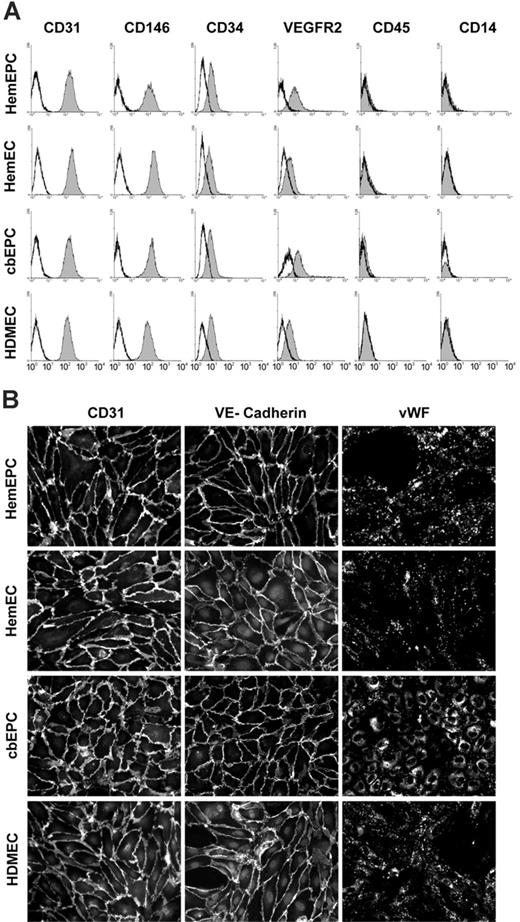
Flow cytometric analysis of isolated cells showed remarkably uniform expression of EC markers CD31, CD146, CD34, and VEGF-R2 (Figure 1A). In addition, the cells were negative for hematopoietic markers CD45 and CD14. Indirect immunofluorescent staining was performed to further examine the expression of EC markers in isolated cells. The results show that the cells express CD31, VE-cadherin, and VWF (Figure 1B). The localization of CD31 and VE-cadherin at the cell-cell borders and VWF in a punctate pattern in the cytoplasm were consistent among the sources of ECs. This confirmed that when examined for expression of a widely accepted panel of EC markers, the cells are nearly indistinguishable from one another.
EC marker expression and characterization. (A) Cytometric analysis of ECs and hematopoietic markers in cultured HemEPCs, HemECs, cbEPCs, and HDMECs. Solid gray histograms represent cells stained with fluorescence-conjugated antibodies. Black lines show cells stained with fluorescence-conjugated isotype control antibodies. (B) Indirect immunofluorescent staining of cells, grown in monolayers, with CD31, VE-cadherin, and VWF antibodies showing positive staining for EC markers (all cells were passage 8). Images were taken with a Nikon Eclipse TE300 (Nikon, Melville, NY) using Spot Advanced 3.5.9 software (Diagnostic Instruments, Sterling Heights, MI) and a 20×/0.45 objective lens.
EC marker expression and characterization. (A) Cytometric analysis of ECs and hematopoietic markers in cultured HemEPCs, HemECs, cbEPCs, and HDMECs. Solid gray histograms represent cells stained with fluorescence-conjugated antibodies. Black lines show cells stained with fluorescence-conjugated isotype control antibodies. (B) Indirect immunofluorescent staining of cells, grown in monolayers, with CD31, VE-cadherin, and VWF antibodies showing positive staining for EC markers (all cells were passage 8). Images were taken with a Nikon Eclipse TE300 (Nikon, Melville, NY) using Spot Advanced 3.5.9 software (Diagnostic Instruments, Sterling Heights, MI) and a 20×/0.45 objective lens.
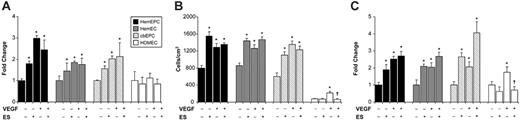
ES induces increased adhesion, migration, and proliferation in HemEPCs, HemECs, and cbEPCs
We previously reported the altered response of HemECs to ES when tested in a VEGF-mediated migration assay.4 We used this altered response as a “marker” for the hemangioma phenotype to test whether HemEPCs exhibit the same phenotype as HemECs in this assay. We examined adhesion, migration, and proliferation of normal and hemangioma-derived EPCs and ECs in the presence and absence of ES and VEGF. We found that 10 ng/mL ES increased the adhesion of HemEPCs and HemECs to FN-coated culture plates (Figure 2A). Treatment of the HemEPCs and HemECs with VEGF (5 ng/mL) also increased adhesion to FN, but pretreatment with ES had no additional effect on the VEGF-induced adhesion (Figure 2A). HDMECs showed no significant changes in adhesion when treated with ES or VEGF (Figure 2A). We also demonstrated increased adhesion of normal cbEPCs to FN when treated with ES or VEGF. These findings indicate ES may differentially regulate adhesion of EPCs compared with mature ECs from infantile foreskin.
Next, we examined how ES regulates the migration of the ECs to VEGF. First, the cell counts (Figure 2B) demonstrate the higher basal migration of HemEPCs, HemECs, and cord blood–derived EPCs compared with mature HDMECs. Migration of HemEPCs, HemECs, and cbEPCs but not HDMECs was increased by pretreatment with 10 ng/mL ES. All ECs exhibited VEGF-induced migration as expected. For HemEPCs, HemECs, and cbEPCs, ES pretreatment and exposure to VEGF did not further increase cellular migration. Nevertheless, as reported by others, ES inhibited VEGF-induced migration of normal mature HDMECs, consistent with its function as an angiogenesis inhibitor.
Previous studies have shown a concentration-dependent response of ECs to ES.21-23 In order to determine whether the altered behavior of HemEPCs, HemECs, and cbEPCs is concentration dependent, we carried out the migration assay with varying ES concentrations. This dose-response analysis showed no significant changes in the migration rate of the cells upon treatment with 1 ng/mL to 10 μg/mL ES (data not shown). A similar pattern was observed using cbEPCs.
We employed a third angiogenic assay to evaluate cellular proliferation in response to ES and VEGF. We assayed ES response either directly after seeding the cells or after allowing cell attachment in the presence of 2% serum for 30 minutes to minimize potential differences in adhesion between the cell types. In both assays, the numbers of HemEPCs, HemECs, and cbEPCs in the ES-treated group were significantly higher than the control group (Figure 2C); whereas ES had no effect on HDMECs. For all ECs, VEGF induced an approximately 2-fold increase in cell number. Basal proliferation was 2- to 3-fold higher in HemEPCs, HemECs, and cbEPCs compared with HDMECs (data not shown). Consistent with previous studies, ES inhibited VEGF-induced proliferation of HDMECs at 24 hours.
ES-induced adhesion, migration, and proliferation of EPCs. Increased adhesion (A), migration (B), and proliferation (C) of HemEPCs, HemECs, and cbEPCs in response to ES. (For the adhesion and the proliferation assay, data are presented relative to control. *P < .05 compared with control; †P < .05 compared with VEGF treatment group; n = 3/treatment; all cells were passage 9). Error bars represent SEM.
ES-induced adhesion, migration, and proliferation of EPCs. Increased adhesion (A), migration (B), and proliferation (C) of HemEPCs, HemECs, and cbEPCs in response to ES. (For the adhesion and the proliferation assay, data are presented relative to control. *P < .05 compared with control; †P < .05 compared with VEGF treatment group; n = 3/treatment; all cells were passage 9). Error bars represent SEM.
ES-induced migration in EPCs is independent of isolation procedures
HemEPCs were isolated by CD133/Ulex selection whereas cbEPCs were isolated by CD34/CD133 selection. Therefore, we tested whether or not EPCs isolated by a different method would also respond to ES. We subjected cbEPCs, purified by Ulex selection or CD31 selection, to the ES-mediated migration assay. This showed that cbEPCs purified by either method show increased migration in response to ES (data not shown). Thus, human EPCs, isolated from hemangioma or cord blood, are stimulated rather than inhibited by ES in these assays of in vitro angiogenesis.
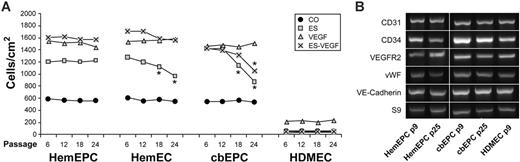
HemEPCs retain the angiogenic response to ES in long-term culture whereas normal EPCs shift toward a mature EC response
We hypothesized that HemEPCs in the proliferating hemangioma differentiate into the apparently mature ECs that line the prominent vascular channels seen in the involuting phase. To test whether such maturation might take place in vitro, we assayed the response to ES in the cells at various time points, corresponding to 2 months' in vitro culture. We defined EC maturation as acquisition of the normal inhibitory response to ES, as seen in HDMECs. Cells were grown in culture; passaged every 4 to 6 days under identical conditions; and cryopreserved at passages 6, 12, 18, and 24. Cells were then thawed and assayed simultaneously for migration in response to ES, VEGF, and ES + VEGF (Figure 3A). The angiogenic response to ES was consistently seen in HemEPCs at passages 6 to 24 and in HemECs and cbEPCs at passages 6 and 12. However, at passages 18 and 24, statistically significant decreases in ES-stimulated cell migration of HemECs and cbEPCs were measured (Figure 3A ##). Furthermore, a statistically significant decrease in the number of cells migrated in response to VEGF plus ES was seen in cbEPCs at passage 24 (Figure 3A gray ×). Hence, cbEPCs and HemECs appear to undergo a shift in ES response toward a mature EC phenotype in culture. In contrast, HemEPCs, which were selected based on the expression of the stem-cell marker CD133, retained the angiogenic response to ES under these conditions, perhaps reflecting a defect or delay in endothelial maturation. Basal and VEGF-induced migration were remarkably constant in all EC types from passage 6 to 24. As seen previously, basal and VEGF-induced migration of HDMECs was lower than HemECs, and VEGF-induced migration was inhibited by ES.4,21,22
To determine if relative levels of well-known endothelial markers increased or decreased with growth in culture, RT-PCR (30 cycles) was performed on cells at passage 9 and passage 25: levels of CD31, CD34, VEGF-R2, VWF, and VE-cadherin did not change in either HemEPCs or cbEPCs (Figure 3B). PCR primers used for this analysis are shown in Table 1.
Effect of long-term culture on the ES response. (A) Analysis of the ES- and VEGF-induced migration of the cells at passages 6, 12, 18, and 24. (*P < .05 compared with passage 6 and 12; n = 3/treatment.) (B) RT-PCR analysis of EC markers at passage 9 and passage 25. HDMECs served as a positive control.
Effect of long-term culture on the ES response. (A) Analysis of the ES- and VEGF-induced migration of the cells at passages 6, 12, 18, and 24. (*P < .05 compared with passage 6 and 12; n = 3/treatment.) (B) RT-PCR analysis of EC markers at passage 9 and passage 25. HDMECs served as a positive control.
Analysis of potential ES binding partners
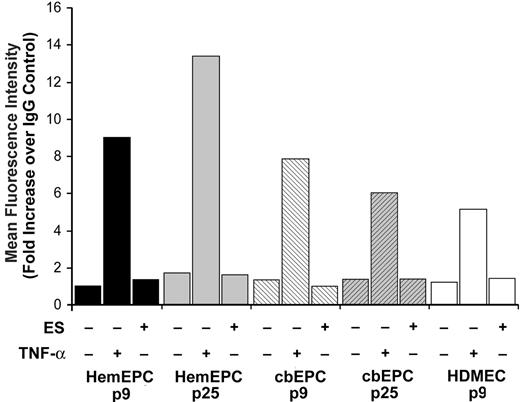
To date there is no clearly defined signaling receptor for ES. However, cell-surface moieties such as integrins, α5β1 in particular,24,25 glypican-1,26 and E-selectin22 have been shown to interact with ES on the cell surface directly or indirectly and to be involved in the inhibitory effects of ES. To determine if E-selectin might be functioning in the ES stimulatory response, we measured basal, TNF-α–induced, and ES-induced E-selectin levels in HemEPCs and cbEPCs at low and high passage. HDMECs were also analyzed as a positive control. TNF-α induced E-selectin above basal levels in all endothelial populations tested, whereas ES did not (Figure 4). This result indicates that (1) the EPCs express cytokine-inducible E-selectin as expected for human ECs, and (2) ES does not induce E-selectin expression. These results suggest that E-selectin is not involved in the stimulatory response of the EPCs to ES because basal levels of expression were not appreciably above background.
To assess possible functions of integrins, the tyrosine and serine phosphorylation status of 3 α integrin subunits and 2 β integrin subunits in control and ES-treated HemEPCs versus HDMECs was measured. In Table 2, the results are summarized and expressed as the ratio of HemEPCs/HDMECs. The α5 integrin showed increased tyrosine and serine phosphorylation in HemEPCs while the β1 integrin showed increased serine phosphorylation. The levels were not changed appreciably with ES treatment, with the exception of a small increase in serine phosphorylation of the β1 integrin. Integrins α1 and β3 were more highly phosphorylated in HDMECs compared with HemEPCs. Taking phosphorylation status as indication of integrin activation, these results suggest that α5β1 may play a role in the ES stimulatory response. However, further experiments will be required to pursue this possibility.
E-selectin expression in EPCs. Flow cytometric analysis of E-selectin expression in HemEPCs, cbEPCs, and HDMECs; HemEPCs and cbEPCs were assayed at passage 9 and passage 25. Cells were treated with media alone, TNF-α (10 ng/mL), or ES (10 ng/mL) for 5 hours prior to harvesting for analysis.
E-selectin expression in EPCs. Flow cytometric analysis of E-selectin expression in HemEPCs, cbEPCs, and HDMECs; HemEPCs and cbEPCs were assayed at passage 9 and passage 25. Cells were treated with media alone, TNF-α (10 ng/mL), or ES (10 ng/mL) for 5 hours prior to harvesting for analysis.
mRNA profiling reveals similarity between hemangioma cells and cbEPC
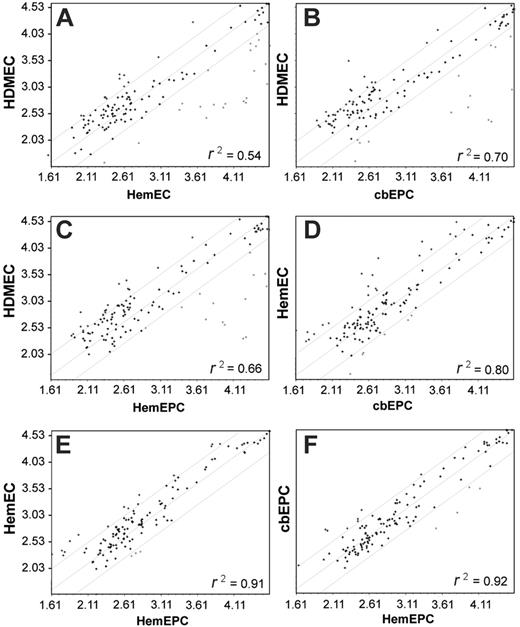
Transcriptional profiling has been used to analyze global gene-expression patterns among different cell populations to determine lineage relationships. We used microarray analysis to test our hypothesis that hemangioma begins with an intrinsic defect in an EPCs, giving rise to HemEPCs which then slowly differentiate into HemECs. We determined the transcriptional profiles of a selected group of genes involved in cell-cell and cell-matrix adhesion molecules. Analysis of 113 adhesion molecule genes, not strictly EC-specific genes, revealed a similarity among HemEPCs, HemECs, and cbEPCs but not with HDMECs. Linear regression analysis of the microarray expression data indicated a close relationship between HemEPCs and HemECs (Figure 5E) but also a close relationship between HemEPCs and cbEPCs (Figure 5F) and HemECs and cbEPCs (Figure 5D). These findings demonstrate a correlation between gene-expression patterns and the property of being stimulated by ES that is not found in fully differentiated ECs. In Table 3, adhesion molecules that were coordinately up-regulated or down-regulated in cbEPCs, HemEPCs, and HemECs are shown.
Genes up-regulated or down-regulated at least 3-fold in different endothelial-cell populations analyzed inFigure 5
Cell type . | Increase/decrease . | Gene products . |
|---|---|---|
| HDMECs | Increase | Collagen, type I, alpha 1; collagen, type V, alpha 3; collagen, type VI, alpha 1; collagen, type VI, alpha 2; chondroitin sulfate proteoglycan 2; hyaluronan synthase 1; tissue inhibitor of metalloproteinase 3 |
| HemEPCs/HemECs/cbEPCs | Increase | Fibronectin; integrin, alpha 5; matrix metalloproteinase 1 |
| HemECs/HDMECs | Increase | Matrix metalloproteinase 2 |
| HemEPCs | Decrease | Laminin, alpha 4 |
| HemEPCs/HemECs/cbEPCs | Decrease | Collagen, type IV, alpha 2; collagen, type XII, alpha 1; connective tissue growth factor; osteonectin |
Cell type . | Increase/decrease . | Gene products . |
|---|---|---|
| HDMECs | Increase | Collagen, type I, alpha 1; collagen, type V, alpha 3; collagen, type VI, alpha 1; collagen, type VI, alpha 2; chondroitin sulfate proteoglycan 2; hyaluronan synthase 1; tissue inhibitor of metalloproteinase 3 |
| HemEPCs/HemECs/cbEPCs | Increase | Fibronectin; integrin, alpha 5; matrix metalloproteinase 1 |
| HemECs/HDMECs | Increase | Matrix metalloproteinase 2 |
| HemEPCs | Decrease | Laminin, alpha 4 |
| HemEPCs/HemECs/cbEPCs | Decrease | Collagen, type IV, alpha 2; collagen, type XII, alpha 1; connective tissue growth factor; osteonectin |
Gene expression-profiling. Linear regression analysis of mRNA levels in HDMECs/HemECs (A), HDMECs/cbEPCs (B), HDMECs/HemEPCs (C), HemECs/cbEPCs (D), HemECs/HemEPCs (E), and HemEPCs/cbEPCs (F). (A-F) Mean and 2 SD limits are indicated by gray lines. The array contained 113 genes encoding matrix proteins and proteins involved in cell-cell and cell-matrix interactions.
Gene expression-profiling. Linear regression analysis of mRNA levels in HDMECs/HemECs (A), HDMECs/cbEPCs (B), HDMECs/HemEPCs (C), HemECs/cbEPCs (D), HemECs/HemEPCs (E), and HemEPCs/cbEPCs (F). (A-F) Mean and 2 SD limits are indicated by gray lines. The array contained 113 genes encoding matrix proteins and proteins involved in cell-cell and cell-matrix interactions.
Discussion
We showed that HemEPCs and normal healthy cord blood–derived EPCs share the stimulatory response to endostatin (ES), which was first described for HemECs.4 ES increased adhesion, migration, and proliferation in HemEPCs, HemECs, and cbEPCs but inhibited these cellular properties in the mature HDMECs. The ES response was maintained following long-term culture of HemEPCs, whereas the degree to which HemECs and cbEPCs were stimulated diminished following long-term culture. E-selectin, an adhesion molecule needed for antiangiogenic activity of ES,22 was induced normally upon cytokine treatment in all EPCs, indicating that altered regulation of E-selectin is not involved. Integrins α5 and β1 showed increased phosphorylation in HemEPCs versus HDMECs, suggesting that α5β1, a previously reported receptor for ES,24,25 might play a role in the stimulatory response to ES. Finally, mRNA profiling of hemangioma and normal blood-derived EPCs demonstrated a strong association among HemEPCs, HemECs, and cbEPCs, indicating similar transcriptional and cellular activity.
The presence of EPCs in proliferating hemangiomas8 and increased circulating EPC levels in hemangioma patients,7 in combination with the appearance of endothelial-lined vascular channels in the involuting phase, support our hypothesis that HemECs arise from HemEPCs. We used the ES response to compare the HemEPCs to HemECs and normal cbEPCs. We found that both CD133+ HemEPCs and CD133– HemECs are stimulated by ES in 3 different in vitro assays, indicating that the absence of cell-surface CD133 does not correspond to full loss of an immature endothelial phenotype. When examined at various time points, corresponding to 2 months of in vitro cell culture, HemEPCs maintained the same degree of stimulatory response whereas HemECs and cbEPCs gradually lost this capacity. Perhaps with further time in culture, these cells would adopt an inhibitory response to ES, indicative of a mature EC phenotype. Clearly, the ES stimulatory response is maintained much longer in HemEPCs compared with normal cbEPCs, suggesting a possible defect in EPC maturation in hemangioma. The mRNA profiling data indicated that both types of endothelial cells from hemangioma, CD133+ EPCs and CD133– ECs, are similar to CD133+ cells derived from the cord blood.
An important and intriguing finding in this study is the stimulatory effect of ES on normal blood-derived EPCs. ES has been reported to inhibit endothelial cellular activities via interaction with VEGF-R2,27 tropomyosin,28 integrin receptors,24,25 and glypicans,26 and indirectly with E-selectin,22 suggesting ES may interact with cells through multiple mechanisms. The mechanism by which ES exerts its stimulatory response on EPCs is likely to differ from the antiangiogenic mechanisms exerted on mature ECs. It has been suggested that ES treatment decreases the levels of circulating ECs in the blood.29 However, little is known about how ES affects cellular responses in circulating or bone marrow–derived EPCs. Differential effects of ES on cell proliferation in mature umbilical vein–derived ECs and mouse embryonic stem-cell–derived ECs have been reported.30 ES decreased the proliferation and growth factor–induced migration in the mature umbilical vein ECs, as expected, but did not affect embryonic stem-cell–derived ECs. These findings suggest that ES may modulate the angiogenic responses depending on the maturation state of the ECs. This unique property of EPCs may provide an invaluable cellular “marker” of the differentiative steps involved in endothelial maturation. In addition, this unique effect on EPCs could have important implications for clinical use of ES. Finally, this study provides further insights into the cellular origins and possible mechanisms leading to formation of infantile hemangioma.
Prepublished online as Blood First Edition Paper, March 30, 2006; DOI 10.1182/blood-2006-03-006478.
Supported by a National Institutes of Health (NIH) grant (P01 AR048564).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Kristin Johnson for preparation of the figures.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal