Abstract
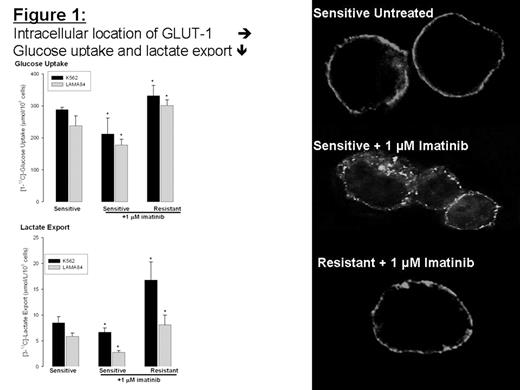
The development of imatinib resistance has become a significant therapeutic problem, in which the etiology appears to be multifactoral (Bcr-Abl mutation and/or overexpression, multidrug-resistance, protein overexpression, etc.) and poorly understood. As of today, there are no precise clinical criteria to predict the development of imatinib resistance in CML, other than relapse of the disease. However, there is evidence that the control of glucose-substrate flux is an important mechanism of the antiproliferative action of imatinib. Thus, imatinib-resistant gastrointestinal stromal (GIST) KIT-activated tumors reveal highly elevated glucose uptake in PET scans. Currently, there is no information about the changes in glucose cell metabolism during development of imatinib resistance in CML patients. We used nuclear magnetic resonance (NMR) spectroscopy and gas chromatography mass spectrometry (GC-MS) to assess 13C glucose uptake and metabolism (glycolysis, the TCA cycle, nucleic acid ribose synthesis) during imatinib treatment in cultured human CML cells with different sensitivity to imatinib. Our results demonstrate that imatinib-sensitive K562 and LAMA84 Bcr-Abl positive cells have decreased glucose uptake, decreased lactate production and improved oxidative TCA cycle upon imatinib treatment. The LAMA84-r (resistant to imatinib due to Bcr-Abl overexpression) and the K562-r (unknown mechanism of resistance) cells maintained high glycolytic metabolic phenotype with elevated glucose uptake and lactate production in the presence of imatinib (Figure 1). In addition, oxidative synthesis of RNA ribose from 13C-glucose via glucose-6-phosphate dehydrogenase was decreased and RNA synthesis via the non-oxidative transketolase pathway was increased in imatinib resistant cells. CML cells which exhibited an [oxidative/non-oxidative] flux ratio for nucleic acid ribose synthesis > 1, were sensitive to imatinib. The resistant K562-r and LAMA84-r exhibited the [oxidative/non-oxidative] flux ratio < 0.7. We correlated 13C glucose uptake and metabolism with intracellular expression and localization of GLUT-1 transporter protein in these cells. There was no difference in total protein load of GLUT-1 upon imatinib treatment between imatinib-sensitive and resistant cell lines. However, the changes in glucose uptake and metabolism were accompanied by intracellular trans-location of GLUT-1 from the plasma membrane to the cytoplasm in imatinib-sensitive treated cells, while GLUT-1 remained at the membrane surface in LAMA84-r and K562-r cells (Figure 1). In summary, elevated glucose uptake and non-oxidative glycolytic metabolic phenotype can be used as sensitive markers for imatinib resistance in Bcr-Abl positive cells with different origins of resistance.
Disclosure: No relevant conflicts of interest to declare.
Author notes
Corresponding author

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal