Abstract
Imatinib mesylate (imatinib) is a potent inhibitor of defined tyrosine kinases (TKs) and is effective in the treatment of malignancies characterized by constitutive activation of these TKs such as chronic myeloid leukemia and gastrointestinal stromal tumors. TKs also play an important role in T-cell receptor (TCR) signal transduction. Inhibitory as well as stimulating effects of imatinib on T cells and dendritic cells have been described. Here, we analyzed the effects of imatinib treatment on antiviral immune responses in vivo. Primary cytotoxic T-cell (CTL) responses were not impaired in imatinib-treated mice after infection with lymphocytic choriomeningitis virus (LCMV) or after immunization with a tumor cell line expressing LCMV glycoprotein (LCMV-GP). Similarly, neutralizing antibody responses to vesicular stomatitis virus (VSV) were not affected. In contrast, secondary expansion of LCMV-specific memory CTLs was reduced in vitro and in vivo, resulting in impaired protection against reinfection. In addition, imatinib treatment delayed the onset of diabetes in a CTL-induced diabetes model. In summary, imatinib treatment in vivo selectively inhibits the expansion of antigen-experienced memory CTLs without affecting primary T- or B-cell responses. Therefore, imatinib may be efficacious in the suppression of CTL-mediated immunopathology in autoimmune diseases without the risk of acquiring viral infections.
Introduction
Imatinib mesylate (STI571, Glivec; Novartis Pharma AG, Basel, Switzerland) selectively inhibits the tyrosine kinases (TKs) ABL, BCR/ABL, ARG, PDGF-R α and β, and c-KIT. Constitutive activation of these TKs has been documented in chronic myeloid leukemia (CML), Philadelphia chromosome–positive (Ph+) acute lymphocytic leukemia, myeloproliferative disorders due to chromosomal rearrangements in PDGF-R, and gastrointestinal stromal tumors with mutations in c-KIT.1-4 In these diseases, blocking of TKs with imatinib is very efficient and substantially improves clinical outcome.
Since TKs are involved in various intracellular signaling pathways, it is not surprising that imatinib treatment affects immune responses. Clinical observations have suggested that imatinib treatment correlates with a reversible dose-dependent lymphopenia and hypogammaglobulinemia.5 Experimental in vitro studies have demonstrated that imatinib inhibited the development of human CD34+ progenitor cell–derived dendritic cells (DCs). In addition, DCs exposed to imatinib were less potent in inducing cytotoxic T-cell (CTL) responses against tumor and recall antigens.6 However, results of the effect of imatinib on DC maturation are controversial. Other experiments have suggested a normal maturation but reduced expansion of DCs in mice when stimulated with Flt3L.7 Further, it has been shown that treating DCs in vitro with imatinib enhanced antigen-presenting cell function and overcame tumor-induced CD4+ T-cell tolerance.8 In addition, several in vitro studies using T cells isolated from human peripheral blood have demonstrated a dose-dependent reduction of T-cell proliferation in the presence of imatinib.1,9,10 These results raise the possibility that imatinib could affect normal immune functions through TK inhibition. TKs play a prominent role in T-cell receptor (TCR) and B-cell receptor (BCR) signal transduction, and, thus, it is conceivable that imatinib may interfere with this process. TCR ligation triggers a signaling cascade that includes activation of the TKs Lck, ZAP70, and Ltk. A recent study has reported the requirement of c-ABL and ARG TKs for TCR-dependent transcriptional activation.11 c-ABL is activated by Lck and then leads to the phosphorylation of ZAP70. It remains unclear whether c-ABL activates only ZAP70 or also activates other downstream proteins.12 However, primary T cells lacking functional ABL TKs showed decreased IL-2 production and cell proliferation in response to TCR stimulation.11 Comparably, it has been shown that ABL phosphorylates the BCR coreceptor CD19, suggesting a role for ABL in the regulation of B-cell proliferation.13 Taken together, these experiments suggest that imatinib treatment in vivo may crucially influence antiviral CD8+ T- and B-cell responses. However, physiologic consequences of imatinib treatment on protective immune response have not yet been demonstrated.
Primary infection with lymphocytic choriomeningitis virus (LCMV), a noncytopathic RNA virus, is controlled almost exclusively by CTLs.14 As αβ TCR transgenic CD8+ T cells (p14) are available against a major CTL target, the LCMV glycoprotein (LCMV-GP) peptide amino acids 33-41 (gp33),15 LCMV infection offers a good model for assessing beneficial or detrimental effects of immunomodulation by imatinib treatment. On the other hand, vesicular stomatitis virus (VSV), a highly cytopathic RNA virus, can be used to study effects on B-cell responses. An early neutralizing antibody response against VSV is required to prevent lethal encephalitis, and, in addition, VSV infection induces lifelong protective B-cell memory.16
In the present study, we analyzed the effects of imatinib on B cells, on T-cell subpopulations, and on DCs during antiviral immune responses in vivo. We found that imatinib did not impair efficient priming of T- and B-cell responses in vivo but inhibited secondary expansion of memory CTLs.
Materials and methods
Mice
C57BL/6 mice were from Harlan (Amsterdam, The Netherlands). The p14 TCR transgenic mice, with approximately 60% specific CD8+ T cells (Vα2+) for the LCMV-GP15 and H8 transgenic mice,17 ubiquitously expressing gp33, were from the Institute for Laboratory Animals (Switzerland). CD45.1+ mice were from C. Mueller (Switzerland) and RIP-GP18 mice were from G. Holländer (Switzerland). Animal experiments were performed with sex- and age-matched mice and approved by the Experimental Animal Committee of the canton of Berne and performed according to Swiss law for animal protection.
Imatinib mesylate and treatment of mice
Imatinib mesylate was from Novartis Pharma AG. Fresh stock solutions were prepared in sterile water. To reach steady-state concentrations, imatinib administration was started 7 days prior to immunization in all in vivo studies. Twice-daily administration of imatinib is required in murine models because the half-life of imatinib in mice is shorter than in humans.19 Either 25 mg/kg imatinib was given by gavage twice daily (indicated as 25/25) or 50 mg/kg was given every morning and 100 mg/kg every evening (indicated as 50/100). These doses of imatinib have been shown to reach plasma concentrations comparable to those in CML patients treated with 400 mg imatinib daily.20-22
For in vitro studies, 10 mM imatinib stock solutions in dimethylsulfoxide (DMSO; Fluka, Buchs, Switzerland) were frozen at –20°C. Indicated concentrations were added to the cell cultures.
Viruses, virus detection, peptide, and 51Cr-release assay
LCMV-WE was received from R.M. Zinkernagel (Switzerland) and propagated on L929 fibroblast cells. VSV-Indiana and recombinant baculovirus, expressing the glycoprotein of VSV (VSV-G), were also from R.M. Zinkernagel. LCMV titers of centrifuged spleen homogenates were determined in a focus-forming assay as described.23 The VSV neutralization assay was performed as described.24 The LCMV-GP, amino acids 33-41 (gp33; KAVYNFATM), was purchased from NeoMPS SA (Strasbourg, France). Primary ex vivo and secondary 51Cr-release assays after 5 days of in vitro restimulation have been described earlier.25
DC generation
DCs were generated from murine bone marrow as described.26
Antibodies and flow cytometry
αCD8-FITC, -PE, –PE-Cy5, and -APC; αCD4-biotin and -PE; αB220-biotin; αI-Ab–major histocompatibility complex (MHC) class II–biotin and –PE-Cy5; αCD45.1-PE; αCD11c-FITC and -PE; αCD11b-PE; αCD70-PE; CD86-PE; αIL-2–APC; αIFNγ-FITC; αTNFα-FITC, –streptavidin-FITC, –PE-Cy5, and –APC were purchased from eBioscience (San Diego, CA). αCD8–PerCP-Cy5.5; αCD4–PerCP-Cy5.5; αCD80-FITC; and αVα2-biotin and -PE were purchased from BD Pharmingen (San Diego, CA). Annexin V–PE was produced and used as previously described.27 MHC class I (H-2Db) tetramers PE-complexed with gp33 were purchased from ProImmune (Oxford, United Kingdom) and used according to the manufacturer's protocol. Intracellular staining was performed as described.28 Relative fluorescence intensities were measured on a BD LSRII (BD Biosciences, San Jose, CA) and analyzed using FlowJo software (Tree Star, Ashland, OR).
T-cell proliferation assays
Memory p14 T cells were generated by adoptive transfer of 106 naive p14 splenocytes into naive C57BL/6 recipients. Twenty-four hours later, the mice were infected intravenously with 200 plaque-forming units (pfu) LCMV. Six weeks later, memory p14 splenocytes were isolated.
For in vitro proliferation, naive and memory p14 splenocytes were purified for CD8+ T cells by magnetic cell sorting (MACS; Miltenyi Biotec, Bergisch Gladbach, Germany). MACS-purified CD8+ T cells (2 × 105) were restimulated for 4 days with 2 × 105 irradiated (10 Gy [1000 rad]) gp33-pulsed or unpulsed naive C57BL/6 splenocytes or with αCD3ϵ (wells coated overnight at 4°C with 2 μg/mL αCD3ϵ in Tris buffer 50 mM, pH = 9). 3H-thymidine (Amersham Biosciences, Little Chalfont, United Kingdom) was added for the last 14 hours. Then, cells were collected with a Packard Filtermate 196-Harvester (Packard, Meriden, CT) on UniFilter-96, GF/C plates (PerkinElmer, Groningen, The Netherlands), and the incorporation of radioactivity was measured after adding Microscint 0 (PerkinElmer) on a Top Count microplate scintillation counter (PerkinElmer, Zaventem, Belgium).
For in vivo proliferation, 1 × 107 naive p14 splenocytes containing approximately 6 × 106 gp33-specific naive CD8+ T cells (CD45.1–) or 3.6 × 106 memory p14 splenocytes containing 2 × 105 gp33-specific memory CD8+ T cells (Vα2+CD45.1–) were adoptively transferred into imatinib-treated and nontreated CD45.1+ recipients. Sixteen hours later, the mice were infected intravenously with 104 pfu LCMV. On days 5, 8, and 14 after infection the in vivo expansion of the transferred T cells (CD45.1–CD8+) was determined by flow cytometry. The absolute number of expanded T cells was calculated based on the count of total splenocytes.
Cell-cycling analysis in vitro and in vivo
Single-cell suspensions of naive and memory p14 spleens were prepared and erythrocytes removed by Puregene red blood cell (RBC) Lysis Solution (Bioconcept, Basel, Switzerland). The cells were washed twice with PBS, adjusted to 2 × 106 cells/mL, and labeled with carboxy-fluorescein diacetate succinimidyl ester (CFSE; Molecular Probes, Eugene, OR) at a final concentration of 5 μM for 10 minutes at 37°C. After labeling, FCS was added up to a final concentration of 10% and cells were washed with PBS at 4°C.
For in vitro cell-cycling analysis, 4 × 106 CFSE-labeled naive and memory p14 splenocytes were restimulated with 2 × 106 irradiated gp33-pulsed or unpulsed naive C57BL/6 splenocytes in the presence of 50 U/mL recombinant murine IL-2 (rmIL-2) and 1 or 5 μM imatinib or DMSO as a solvent control.
For in vivo cell-cycling analysis, 5.1 × 106 CFSE-labeled memory p14 splenocytes containing 3.3 × 105 gp33-specific memory CD8+ T cells (Vα2+CD45.1–) were injected intravenously into imatinib-treated and nontreated CD45.1+ recipients. Sixteen hours later, the mice were infected intravenously with 1200 pfu LCMV, and on days 2, 3, and 4 after infection CFSE dilution and annexin V staining of CD8+Vα2+CD45.1– T cells in blood and spleen were analyzed by flow cytometry.
LCMV protection experiments
Eight weeks after intravenous infection of naive C57BL/6 mice with 200 pfu LCMV, single-cell suspensions of the spleens were prepared. The number of gp33-specific memory CD8+ T cells was determined by Db-gp33–tetramer staining. Imatinib-treated and nontreated naive C57BL/6 mice were infected intravenously with 104 pfu LCMV, and 10 hours later 2.3 × 106 splenocytes containing 5 × 104 gp33-specific memory CD8+ T cells were injected intravenously. Imatinib-treated and nontreated naive C57BL/6 mice infected intravenously at the same time point with 104 pfu LCMV but not receiving memory CTLs served as controls. At different time points, virus titers in spleen were measured as described in virus detection.
Induction of diabetes
Memory p14 splenocytes (8.2 × 106) containing 2 × 105 gp33-specific memory CD8+ T cells (Vα2+) were adoptively transferred into imatinib-treated and nontreated RIP-GP mice. One and 5 days later, mice were immunized intravenously with 2 × 105 H8-derived DCs. At different time points, the glucose concentration in blood obtained from a tail vein was measured using a Freestyle mini (Abbott, Baar, Switzerland) according to the manufacturer's guide. Mice were considered diabetic with values greater than 14 mM at 2 consecutive measurements.
Statistical analyses
Statistical significance was determined by Student t test, unpaired.
Results
Primary cytotoxic T-cell responses are not impaired in imatinib-treated mice
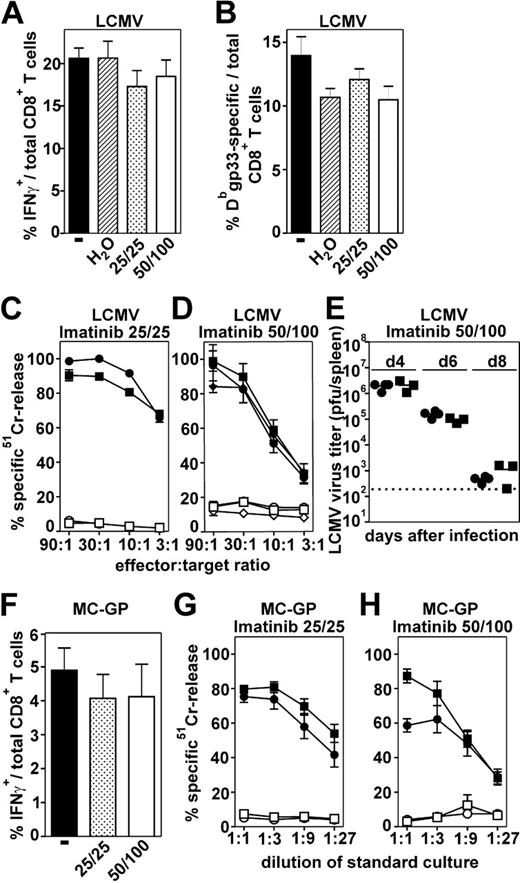
Antiviral CTL responses in imatinib-treated and nontreated naive C57BL/6 mice were analyzed after infection with LCMV. Eight days after infection, the frequency of LCMV-gp33–specific CTLs was analyzed by intracellular IFNγ production after in vitro restimulation (Figure 1A) or by tetramer staining (Figure 1B). The frequency of the specific CTLs in imatinib-treated mice was similar to the frequency in nontreated mice. In addition, CTLs isolated from imatinib-treated and nontreated mice lysed peptide-pulsed target cells to a comparable extent (Figure 1C-D), irrespective of the imatinib dose used for the treatment. Control mice treated twice daily with sterile water and nontreated mice both lysed peptide-pulsed target cells identically. These experiments indicate that primary CTL expansion and function is not reduced in response to LCMV infection in imatinib-treated mice. The unimpaired CTL function in imatinib-treated mice resulted in an elimination kinetic of LCMV similar to that in nontreated mice (Figure 1E).
LCMV rapidly replicates in mice and causes a general inflammation leading to a strong activation of CTLs. Differences in CTL expansion in the presence of imatinib may be more important in response to less-potent stimuli. Therefore, we analyzed CTL responses in vivo after immunization with a tumor cell line expressing LCMV-GP (MC-GP29 ). The frequency of gp33-specific CTLs was analyzed by intracellular IFNγ production. In imatinib-treated mice, the frequency of the specific CTLs was comparable to the frequency in nontreated mice (Figure 1F). In addition, these CTLs were functionally not affected and efficiently lysed peptide-pulsed target cells after 5 days of in vitro restimulation, irrespective of the imatinib dose used for the treatment (Figure 1G-H). Taken together, antiviral primary CTL responses were not impaired in imatinib-treated mice.
Neutralizing antibody responses after a viral infection are not impaired in imatinib-treated mice
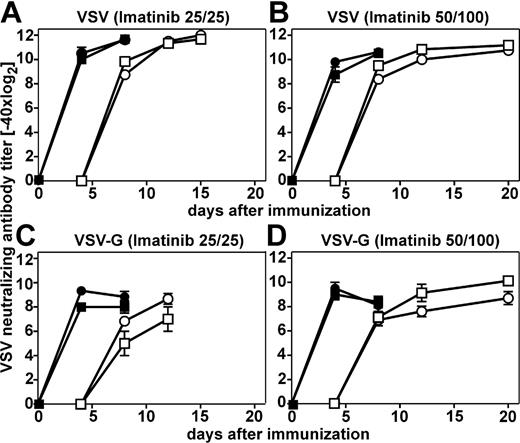
VSV induces strong neutralizing antibody responses. The early IgM antibody response is CD4+ T-cell help independent, whereas the later IgG antibody response, starting 6 to 7 days after infection, is CD4+ T-cell help dependent.24 IgM and IgG neutralizing antibody responses after infection with VSV in imatinib-treated mice were comparable to the responses in nontreated mice, irrespective of the imatinib dose used for the treatment (Figure 2A-B). To investigate a possible influence of imatinib treatment on antibody responses to nonreplicating antigens, mice were immunized with baculo-derived VSV-G protein. Again, neutralizing antibody responses in imatinib-treated mice were similar to non-treated mice (Figure 2C-D). Thus, imatinib treatment influenced neither the activation of B cells and their differentiation into plasma cells nor the activation of CD4+ T cells to a detectable level.
Primary CTL responses in imatinib-treated mice. Imatinib-treated (low-25/25 and high-dose 50/100 regimen) and nontreated naive C57BL/6 mice were infected intravenously with 200 pfu LCMV (A-E) or immunized intraperitoneally with 2 × 106 MC-GP cells (F-H). Eight days after LCMV infection the frequency of gp33-specific CTLs was analyzed in the spleen by intracellular IFNγ staining after in vitro restimulation (A) and by tetramer staining (B). (C-D) To determine CTL activity, splenocytes were isolated 8 days after LCMV infection and analyzed in a 51Cr-release assay. • indicates imatinib-treated mice; ▪, nontreated mice; and ♦, mice treated with sterile water. Filled symbols represent gp33-pulsed target cells and open symbols represent unpulsed target cells. CTL activity is given as mean ± SEM of 3 to 5 mice per group. (E) Virus titers were measured in the spleen 4, 6, and 8 days after LCMV infection. Virus titers are expressed as log10 plaque-forming units (pfu) of virus per spleen. (F) Nine days after immunization with MC-GP cells, the frequency of gp33-specific CTLs was analyzed by intracellular IFNγ staining after in vitro restimulation. (G-H) Splenocytes of MC-GP–immunized mice were isolated 9 days after immunization and the induction of a CTL response was assessed in a 51Cr-release assay after 5 days of in vitro restimulation. CTL activity is given as mean ± SEM of 3 to 5 mice per group. One of 2 representative experiments is shown (A-D, F-H).
Primary CTL responses in imatinib-treated mice. Imatinib-treated (low-25/25 and high-dose 50/100 regimen) and nontreated naive C57BL/6 mice were infected intravenously with 200 pfu LCMV (A-E) or immunized intraperitoneally with 2 × 106 MC-GP cells (F-H). Eight days after LCMV infection the frequency of gp33-specific CTLs was analyzed in the spleen by intracellular IFNγ staining after in vitro restimulation (A) and by tetramer staining (B). (C-D) To determine CTL activity, splenocytes were isolated 8 days after LCMV infection and analyzed in a 51Cr-release assay. • indicates imatinib-treated mice; ▪, nontreated mice; and ♦, mice treated with sterile water. Filled symbols represent gp33-pulsed target cells and open symbols represent unpulsed target cells. CTL activity is given as mean ± SEM of 3 to 5 mice per group. (E) Virus titers were measured in the spleen 4, 6, and 8 days after LCMV infection. Virus titers are expressed as log10 plaque-forming units (pfu) of virus per spleen. (F) Nine days after immunization with MC-GP cells, the frequency of gp33-specific CTLs was analyzed by intracellular IFNγ staining after in vitro restimulation. (G-H) Splenocytes of MC-GP–immunized mice were isolated 9 days after immunization and the induction of a CTL response was assessed in a 51Cr-release assay after 5 days of in vitro restimulation. CTL activity is given as mean ± SEM of 3 to 5 mice per group. One of 2 representative experiments is shown (A-D, F-H).
DCs generated in vitro in the presence of imatinib are not functionally impaired in vivo
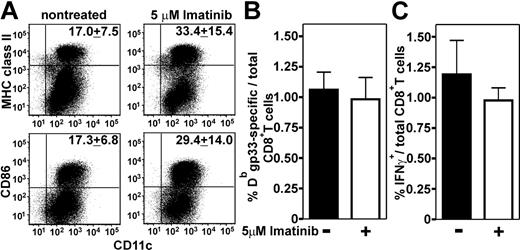
The normal induction of antiviral CTL and B-cell responses in vivo suggested an unimpaired differentiation and maturation of professional antigen-presenting cells in the presence of therapeutic concentrations of imatinib. To determine the effect of imatinib on the differentiation of murine DCs in more detail, we generated DCs from C57BL/6 bone marrow cells in the presence of 1 μM imatinib (data not shown), 5 μM imatinib, or DMSO as a solvent control (Figure 3). The maximum plasma concentration measured in CML patients after a single administration of 400 mg imatinib is about 3.2 μM but can reach 6.2 μM at steady state with 400 mg imatinib administered twice daily.30 Therefore, the imatinib concentrations used in our experiments are in the therapeutic range. In the presence of imatinib, significantly lower numbers of DCs were generated (1.1 × 106 ± 0.2 × 106) compared with nontreated controls (4.0 × 106 ± 0.4 × 106)(P < .001). Surprisingly, DCs raised in the presence of imatinib displayed higher maturation. The expression of MHC class II, CD86, CD80, and CD70 was increased (Figure 3A; not shown). To analyze whether these phenotypic differences in DC maturation affect the induction of antigen-specific primary CTL responses, bone marrow–derived DCs from H8 transgenic mice were generated in the presence or absence of 5 μM imatinib. DCs (2 × 105) were injected into naive C57BL/6 mice. Ten days later, the frequency of gp33-specific IFNγ-secreting and tetramer+ CD8+ T cells was determined. Although DCs generated in the presence of imatinib displayed higher maturation, they induced the similar frequency of antigen-specific CD8+ T cells as nontreated DCs (Figure 3B). In addition, CTLs induced by imatinib-treated and nontreated DCs produced comparable levels of IFNγ in response to antigenic stimulation (Figure 3C). Therefore, DCs generated in vitro in the presence of imatinib induced normal CTL responses in vivo that were quantitatively and qualitatively similar to those in nontreated control mice.
Neutralizing antibody responses after immunization with VSV and VSV-G in imatinib-treated mice. Imatinib-treated (•) (low-25/25 and high-dose 50/100 regimen) and nontreated naive C57BL/6 mice (▪) were infected intravenously with 2 × 106 VSV pfu (A-B) or immunized intravenously with 20 μg VSV-G (C-D). Neutralizing antibody responses were determined 4, 8, 12, and 15 or 20 days later. Filled symbols represent total Ig, open symbols represent IgG. Results are given as the mean ± SEM of 3 to 5 mice per group.
Neutralizing antibody responses after immunization with VSV and VSV-G in imatinib-treated mice. Imatinib-treated (•) (low-25/25 and high-dose 50/100 regimen) and nontreated naive C57BL/6 mice (▪) were infected intravenously with 2 × 106 VSV pfu (A-B) or immunized intravenously with 20 μg VSV-G (C-D). Neutralizing antibody responses were determined 4, 8, 12, and 15 or 20 days later. Filled symbols represent total Ig, open symbols represent IgG. Results are given as the mean ± SEM of 3 to 5 mice per group.
Maturation and function of DCs generated in the presence of imatinib. (A) DCs were generated from C57BL/6 bone marrow cells in the presence of 5 μM imatinib or DMSO as solvent control. Mature DCs were stained with αCD11c-FITC and αMHC class II–PE-Cy5 or αCD86-PE antibody. Numbers indicate the mean ± SEM of 5 independent cultures. One representative dot plot is shown. (B-C) DCs were generated from H8 bone marrow and 2 × 105 imatinib-treated or nontreated DCs were injected intravenously into naive C57BL/6 mice. Ten days later, the frequency of gp33-specific CD8+ T cells was determined by tetramer staining (B) and by intracellular IFNγ staining after in vitro restimulation with gp33 (C). Results are given as means ± SEM of 3 mice per group.
Maturation and function of DCs generated in the presence of imatinib. (A) DCs were generated from C57BL/6 bone marrow cells in the presence of 5 μM imatinib or DMSO as solvent control. Mature DCs were stained with αCD11c-FITC and αMHC class II–PE-Cy5 or αCD86-PE antibody. Numbers indicate the mean ± SEM of 5 independent cultures. One representative dot plot is shown. (B-C) DCs were generated from H8 bone marrow and 2 × 105 imatinib-treated or nontreated DCs were injected intravenously into naive C57BL/6 mice. Ten days later, the frequency of gp33-specific CD8+ T cells was determined by tetramer staining (B) and by intracellular IFNγ staining after in vitro restimulation with gp33 (C). Results are given as means ± SEM of 3 mice per group.
Imatinib inhibits secondary expansion of CTLs
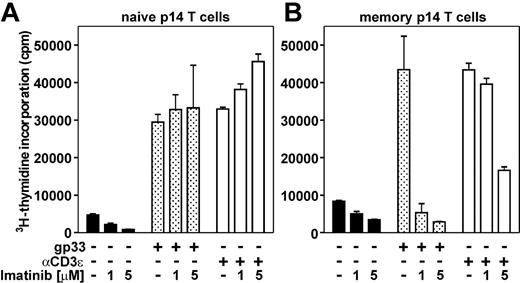
So far, our experiments suggested that imatinib does not affect primary antiviral immune responses in vivo. This finding was surprising, since various in vitro studies have shown an impaired expansion of T cells in response to TCR stimulation by antigen, allogeneic mature DCs, phytohemagglutinin, or αCD3/αCD28.1,9,10 The use of a TCR transgenic system allows analysis of in vitro and in vivo expansion of naive and memory CD8+ T cells in response to stimulation with a defined antigenic stimulus. 3H-thymidine incorporation of naive and memory p14 T cells was measured after 4 days of in vitro stimulation in the presence of 1 or 5 μM imatinib or DMSO as a solvent control. Proliferation of naive p14 T cells in the presence of 1 or 5 μM imatinib was similar or slightly increased compared with proliferation of nontreated naive p14 T cells (Figure 4A). This was irrespective of whether the cells were stimulated with gp33-pulsed splenocytes or with αCD3 monoclonal antibody. Therefore, the in vitro expansion of naive CD8+ T cells in response to TCR stimulation is not affected in the presence of therapeutic imatinib concentrations. In contrast, the in vitro expansion of memory p14 T cells was reduced 8-fold in the presence of 1 or 5 μM imatinib after antigen-specific stimulation with gp33-pulsed splenocytes (Figure 4B). Expansion of memory p14 T cells after unspecific TCR stimulation by αCD3 was not impaired in the presence of 1 μM imatinib and only reduced 3-fold in the presence of 5 μM imatinib. This difference in proliferation after unspecific TCR stimulation compared with the antigen-specific stimulation of memory CTLs may be explained by the fact that gp33 stimulates exclusively antigen-experienced memory CTLs. In contrast, αCD3 stimulates gp33-specific memory CTLs (which have a frequency of 23.9% ± 7.1% in memory p14 mice; data not shown) and in large parts naive CTLs. Since the expansion of naive CTLs was not impaired in the presence of imatinib, the expansion of CD8+ T cells from memory p14 mice in response to αCD3 stimulation is not affected to the same degree as in response to specific TCR stimulation.
Expansion of naive and memory p14 T cells in vitro. Naive p14 T cells (A) and memory p14 T cells (B) were restimulated in vitro for 4 days with irradiated nonpulsed splenocytes (▪), gp33-pulsed splenocytes (▦), or αCD3 (□) in the presence of 1 or 5 μM imatinib or DMSO as a solvent control. 3H-thymidine incorporation was measured during the last 14 hours of culture. Results are given as mean ± SEM of 3 to 4 samples per group. One of 2 representative experiments is shown.
Expansion of naive and memory p14 T cells in vitro. Naive p14 T cells (A) and memory p14 T cells (B) were restimulated in vitro for 4 days with irradiated nonpulsed splenocytes (▪), gp33-pulsed splenocytes (▦), or αCD3 (□) in the presence of 1 or 5 μM imatinib or DMSO as a solvent control. 3H-thymidine incorporation was measured during the last 14 hours of culture. Results are given as mean ± SEM of 3 to 4 samples per group. One of 2 representative experiments is shown.
The in vivo expansion of naive and memory p14 T cells was analyzed in the spleen after adoptive transfer into imatinib-treated and nontreated recipients. The expansion of naive p14 CTLs after infection with LCMV in imatinib-treated mice was comparable to nontreated mice. This resulted in similar numbers of CTLs on day 14 after infection (Figure 5A). In contrast, secondary expansion of memory p14 CTLs was reduced by a factor of 2 in imatinib-treated mice compared with the expansion in nontreated mice at the peak of expansion on day 8, resulting in a 3-fold lower number of CTLs 14 days after infection (Figure 5A). Activated naive and memory p14 CTLs in imatinib-treated mice were not functionally impaired compared with those in nontreated controls. Most of the gp33-specific T cells produced the inflammatory cytokines IFNγ and TNFα in response to antigenic restimulation 8 days after infection, whereas only a few cells produced IL-2 at a detectable level (Figure 5B). This indicates that imatinib selectively impaired the expansion of memory CTLs without affecting their differentiation into effector CTLs.
Impaired survival of expanding memory CTLs in vivo in the presence of imatinib
A reduced 3H-thymidine incorporation could reflect either an impaired cell-cycle activity or a reduced number of cells participating in the response. To distinguish between these 2 possibilities and to visualize cell proliferation, p14 T cells were labeled with the fluorescent dye CFSE. After cell division, CFSE is equally distributed between the 2 daughter cells, thus allowing resolution of cell division by flow cytometry.
Naive and memory p14 splenocytes were restimulated with irradiated gp33-pulsed splenocytes in vitro, and 1, 2, 3, and 4 days later CFSE dilution of CD8+Vα2+ T cells was analyzed by flow cytometry. The CFSE dilution of naive p14 T cells in imatinib-treated cultures was similar to that in nontreated cultures (Figure 6A; only day 4 is shown). Nontreated memory p14 T cells efficiently proliferated in response to antigen-specific restimulation. In contrast, only a few memory p14 T cells entered cell cycling in the presence of imatinib (Figure 6B).
To analyze cell proliferation in vivo, memory p14 T cells were CFSE labeled and transferred to recipients. After LCMV infection, the CFSE dilution was analyzed 2, 3, and 4 days later (Figure 6C-D; only day 4 is shown). In marked contrast to our results (Figure 6B) and published in vitro experiments,1,10 proliferation and cell division of memory CTLs in response to a viral infection in vivo was identical in both imatinib-treated and nontreated mice (Figure 6C). Antigen-specific restimulated memory CTLs underwent up to 9 cell divisions during the 4 days after LCMV infection, irrespective of the imatinib treatment. However, if we looked at the absolute number of specific CTLs in the spleen, we found a 3-fold reduction of dividing cells in imatinib-treated mice compared with nontreated mice (Figure 6D). The identical number of cell divisions of memory CTLs in imatinib-treated and nontreated mice with an up to 3-fold reduced number of cells in each division step suggested a reduced survival of proliferating cells. This was confirmed by a higher percentage of annexin V–positive specific CTLs in imatinib-treated mice (Figure 6D, inset). Therefore, the expansion of memory CTLs in vitro seems to be limited by a block in cell-cycling activity. In contrast, the reduced expansion of memory CTLs in imatinib-treated mice in vivo was due to reduced survival of proliferating cells.
Expansion of naive and memory p14 T cells in vivo. (A) Naive (1 × 107) p14 splenocytes (•) containing approximately 6 × 106 gp33-specific naive CD8+ T cells (Vα2+CD45.1–) or 3.6 × 106 memory p14 splenocytes (▪) containing 2 × 105 gp33-specific memory CD8+ T cells (Vα2+CD45.1–) were injected intravenously into imatinib-treated (high-dose 50/100 regimen; filled symbols) and nontreated (open symbols) CD45.1+ recipients. Sixteen hours later, the mice were infected intravenously with 104 pfu LCMV. On days 5, 8, and 14 after infection, the number of CD45.1–CD8+ T cells in the spleen was analyzed by flow cytometry. Symbols represent the means ± SEM of 3 to 4 mice per group and time point. (B) The frequencies of IFNγ-, TNFα-, and IL-2–secreting CD45.1–CD8+ T cells were analyzed in the spleen by flow cytometry on day 8 after infection after in vitro restimulation with gp33. Results are given as means ± SEM of 3 to 4 mice per group.
Expansion of naive and memory p14 T cells in vivo. (A) Naive (1 × 107) p14 splenocytes (•) containing approximately 6 × 106 gp33-specific naive CD8+ T cells (Vα2+CD45.1–) or 3.6 × 106 memory p14 splenocytes (▪) containing 2 × 105 gp33-specific memory CD8+ T cells (Vα2+CD45.1–) were injected intravenously into imatinib-treated (high-dose 50/100 regimen; filled symbols) and nontreated (open symbols) CD45.1+ recipients. Sixteen hours later, the mice were infected intravenously with 104 pfu LCMV. On days 5, 8, and 14 after infection, the number of CD45.1–CD8+ T cells in the spleen was analyzed by flow cytometry. Symbols represent the means ± SEM of 3 to 4 mice per group and time point. (B) The frequencies of IFNγ-, TNFα-, and IL-2–secreting CD45.1–CD8+ T cells were analyzed in the spleen by flow cytometry on day 8 after infection after in vitro restimulation with gp33. Results are given as means ± SEM of 3 to 4 mice per group.
Cell-cycling analysis of memory p14 T cells in vitro and in vivo. CFSE-labeled naive (4 × 106) (A) and memory p14 splenocytes (4 × 106) (B) were restimulated in vitro with irradiated gp33-pulsed splenocytes in the presence of 5 μM imatinib or DMSO as a solvent control. Four days later, CFSE dilution of Vα2+CD8+ was assessed by flow cytometry. (C-D) CFSE-labeled memory p14 splenocytes (5.1 × 106) containing 3.3 × 105 gp33-specific memory CD8+ T cells (Vα2+CD45.1–) were injected intravenously into imatinib-treated (high-dose 50/100 regimen) and nontreated CD45.1+ recipients. Sixteen hours later, the mice were infected intravenously with 1200 pfu LCMV. CFSE dilution of specific T cells (Vα2+CD8+CD45.1–CFSE+) was assessed by flow cytometry 4 days after infection. (C) Percentage of proliferating specific T cells in imatinib-treated and nontreated mice. (D) Absolute number of gp33-specific T cells in imatinib-treated and nontreated mice per spleen. (Inset) Percentage of annexin V+ cells within Va2+CD8+CD45.1– cells in imatinib-treated and nontreated mice. One representative histogram of 4 to 6 samples per group is shown.
Cell-cycling analysis of memory p14 T cells in vitro and in vivo. CFSE-labeled naive (4 × 106) (A) and memory p14 splenocytes (4 × 106) (B) were restimulated in vitro with irradiated gp33-pulsed splenocytes in the presence of 5 μM imatinib or DMSO as a solvent control. Four days later, CFSE dilution of Vα2+CD8+ was assessed by flow cytometry. (C-D) CFSE-labeled memory p14 splenocytes (5.1 × 106) containing 3.3 × 105 gp33-specific memory CD8+ T cells (Vα2+CD45.1–) were injected intravenously into imatinib-treated (high-dose 50/100 regimen) and nontreated CD45.1+ recipients. Sixteen hours later, the mice were infected intravenously with 1200 pfu LCMV. CFSE dilution of specific T cells (Vα2+CD8+CD45.1–CFSE+) was assessed by flow cytometry 4 days after infection. (C) Percentage of proliferating specific T cells in imatinib-treated and nontreated mice. (D) Absolute number of gp33-specific T cells in imatinib-treated and nontreated mice per spleen. (Inset) Percentage of annexin V+ cells within Va2+CD8+CD45.1– cells in imatinib-treated and nontreated mice. One representative histogram of 4 to 6 samples per group is shown.
Imatinib impairs CTL-mediated protection against reinfection
So far, we have shown that the secondary expansion of antiviral CTLs in vivo is impaired in imatinib-treated mice. To determine whether this finding is of physiologic relevance, antiviral protection against reinfection was analyzed. To exclude the possibility that soluble factors such as neutralizing antibodies or cytokines play a major role in the protection against challenge infections with LCMV, antiviral protection was studied after adoptive transfer of splenocytes from LCMV-immunized mice into imatinib-treated and nontreated recipients. Virus titers in spleens were analyzed 3 and 4 days after the challenge infection (Figure 7A). The mean reduction of the virus titer in the spleen after transfer of memory CTLs in nontreated mice was reduced 1600-fold on day 3 and 30 000-fold on day 4 compared with control C57BL/6 mice. In contrast, memory CTLs in imatinib-treated mice reduced the virus titer only 140-fold on day 3 and 800-fold on day 4 compared with control C57BL/6 mice. Imatinib has been shown to reduce vaccinia virus replication.31 In contrast, LCMV titers in the spleen 4 days after infection were comparable in imatinib-treated and nontreated mice. In addition, imatinib did not impair LCMV replication in vitro in MC57-infected cells (S.M., unpublished results, August 2005). In summary, the reduced secondary expansion of memory CTLs in the presence of imatinib resulted in impaired protection against reinfection.
Imatinib delays the onset of autoimmune diabetes
An impaired secondary expansion of CTLs in the presence of imatinib may be useful in the treatment of CTL-mediated autoimmune diseases. We analyzed this possibility in a murine model of diabetes. RIP-GP mice express the LCMV-GP under the rat insulin promoter in the pancreatic β cells.18 In these mice, activation of LCMV-GP–specific CTLs results in the destruction of the pancreatic islets and the onset of diabetes. Memory p14 T cells were adoptively transferred to imatinib-treated and nontreated RIP-GP mice. One and 5 days later, the mice were immunized with LCMV gp33-expressing DCs generated from H8 mice, and the blood-glucose concentration was measured thereafter (Figure 7B). Imatinib treatment delayed the onset of diabetes, and blood-glucose levels were lower in imatinib-treated mice compared with non-treated mice. In one imatinib-treated mouse, diabetes was prevented completely during the treatment and blood-glucose concentration only increased 4 days after imatinib treatment had been stopped (data not shown). In contrast, all nontreated mice died from diabetes within 10 days after DC immunization. Therefore, imatinib treatment delayed the onset of CTL-mediated immunopathological sequel and diabetes.
CTL-mediated protection against reinfection with LCMV and the onset of autoimmune diabetes in imatinib-treated mice. (A) Splenocytes (2.3 × 106) from LCMV immune C57BL/6 mice containing 5 × 104 gp33-specific memory CD8+ T cells were adoptively transferred into imatinib-treated (•; high-dose 50/100 regimen) and nontreated (▪) C57BL/6 recipients. The recipients had been infected intravenously with 104 pfu LCMV 10 hours earlier. Imatinib-treated and nontreated C57BL/6 mice infected intravenously at the same time point with 104 pfu LCMV, but not receiving memory CTLs, served as controls. LCMV titers in the spleens were measured 3 and 4 days after adoptive transfer, and the reduction of virus titer compared with controls was calculated. Results are given as the mean ± SEM of 3 to 4 mice per group. (B) Memory p14 splenocytes (8.2 × 106) containing 2 × 105 gp33-specific memory CD8+ T cells (Vα2+) were adoptively transferred into imatinib-treated (•; high-dose 50/100 regimen) and nontreated (▪) RIP-GP mice. One and 5 days later, RIP-GP mice were immunized intravenously with 2 × 105 H8-derived DCs, and blood-glucose concentration was measured at the time points indicated. Results are given as the mean ± SEM of 3 to 4 mice per group. Blood-glucose levels were statistically analyzed in imatinib-treated and nontreated mice 9 and 10 days after immunization. †Death of mice due to diabetes.
CTL-mediated protection against reinfection with LCMV and the onset of autoimmune diabetes in imatinib-treated mice. (A) Splenocytes (2.3 × 106) from LCMV immune C57BL/6 mice containing 5 × 104 gp33-specific memory CD8+ T cells were adoptively transferred into imatinib-treated (•; high-dose 50/100 regimen) and nontreated (▪) C57BL/6 recipients. The recipients had been infected intravenously with 104 pfu LCMV 10 hours earlier. Imatinib-treated and nontreated C57BL/6 mice infected intravenously at the same time point with 104 pfu LCMV, but not receiving memory CTLs, served as controls. LCMV titers in the spleens were measured 3 and 4 days after adoptive transfer, and the reduction of virus titer compared with controls was calculated. Results are given as the mean ± SEM of 3 to 4 mice per group. (B) Memory p14 splenocytes (8.2 × 106) containing 2 × 105 gp33-specific memory CD8+ T cells (Vα2+) were adoptively transferred into imatinib-treated (•; high-dose 50/100 regimen) and nontreated (▪) RIP-GP mice. One and 5 days later, RIP-GP mice were immunized intravenously with 2 × 105 H8-derived DCs, and blood-glucose concentration was measured at the time points indicated. Results are given as the mean ± SEM of 3 to 4 mice per group. Blood-glucose levels were statistically analyzed in imatinib-treated and nontreated mice 9 and 10 days after immunization. †Death of mice due to diabetes.
Discussion
Previous studies have shown that the expansion of CD8+ T cells in response to polyclonal T-cell stimuli such as αCD3 or phytohemagglutinin as well as in response to antigenic stimulation was impaired in the presence of imatinib.1,9,10 Since peripheral-blood lymphocytes were used as responders in these experiments, proliferating cells may largely represent antigen-experienced effector and memory T cells.32 In contrast, naive CD8+ T cells, expressing homing receptors for secondary lymphoid organs,32 are resident in the secondary lymphoid organs and therefore may have been excluded from these analyses. Thus, the use of TCR transgenic mice allowed a differentiated study of the expansion of naive and memory CD8+ T cells. Surprisingly, the expansion of naive CTLs in vitro and in vivo was not impaired in the presence of imatinib. More importantly, this resulted in an unimpaired control of primary LCMV infection in vivo. In addition, the unimpaired generation of neutralizing antibodies in response to VSV infection indicates a normal activation and expansion of CD4+ T cells and a normal B-cell function in the presence of therapeutic imatinib concentrations. This coincides with the clinical observation that patients under imatinib medication are not abnormally susceptible to viral infections.4,33
Previous studies have documented controversial effects of imatinib on DC maturation and function ranging from impaired maturation to enhanced stimulation of tolerant CD4+ T cells.6-8 These differences were attributed to different culture conditions, various dosages, different time of exposure to imatinib, and differences in the DC progenitors analyzed (eg, bone marrow–versus monocyte-derived DCs).34 All these previous studies have analyzed the DC expansion and phenotype in response to maturation stimuli such as TNFα or Flt3L in vitro and in vivo.6-8 On the other hand, DC maturation in response to a physiologic stimulus such as a viral infection has not been analyzed previously. Primary CD8+ and CD4+ T-cell responses depend on the presentation of the specific peptides on MHC class I and II, respectively, on professional antigen-presenting cells.35,36 Therefore, the unaffected expansion of specific CTLs in response to LCMV infection in the presence of imatinib suggested an unimpaired differentiation of DCs in vivo. This conclusion was further supported by a normal CTL induction in vivo by bone marrow–derived DCs that have been generated in vitro in the presence of imatinib.
To be able to differentiate between effects of imatinib treatment on naive versus memory virus–specific T cells, TCR transgenic CTLs had to be analyzed. This has the advantage that specific T-cell populations can be followed in vivo but the disadvantage that the high frequency of specific T cells may impact on virus control. In contrast to the unimpaired primary CTL, B-cell, and DC functions in response to viral infections in vivo, the secondary expansion of CTLs in response to an antigen rechallenge was significantly reduced. With these results, the following question is raised: how could imatinib inhibit the expansion of antigen-experienced CD8+ T cells without affecting primary CTL expansion? The inhibitory effect of imatinib on memory CD8+ T-cell expansion results most likely from the inhibition of one or more TKs involved in TCR signal transduction. While the sequence of intracellular events in TCR signaling transduction in naive T cells is well characterized, the intracellular signaling events following memory TCR stimulation are much less clear.37 It has been demonstrated that lipid rafts of memory CD8+ T cells contain more phosphoproteins such as LAT. In addition, they are more efficiently phosphorylated compared with naive CD8+ T cells, suggesting a higher TK activity in memory CTLs.38 A recent study has described the requirement of ABL kinases in TCR signal transduction.11 Imatinib, by inactivating c-ABL, led to decreased phosphorylation of ZAP70 and LAT. This resulted in reduced activation of transcription factors, causing a decreased T-cell expansion. The involvement of ABL kinases in TCR signal transduction and T-cell viability is further confirmed by ABL-deficient mice, which show thymic and splenic atrophy and substantial T- and B-cell lymphopenia.9,11 However, there are currently no indications that c-ABL may be regulated differently in naive and memory CTLs. In contrast, another study has reported that in memory CD8+ T cells Lck expression is increased and associated with the CD8 coreceptor, whereas in naive CD8+ T cells Lck is homogenously distributed in the cytoplasm and must first be recruited to the CD8 coreceptor upon TCR stimulation.39 This may explain why memory CD8+ T cells reach the threshold for proliferation more rapidly than naive T cells. Interestingly, Seggewiss et al1 recently showed that Lck shares sequence homologies in the ATP-binding pocket with known imatinib targets. This suggests that imatinib affects TCR signaling through inhibition of Lck. Lck as a target of imatinib could explain the preferential inhibition of memory CTL expansion.
The total number of T cells present in the antigen-specific pool is determined by 3 cell-intrinsic parameters: the proportion of T cells entering into the proliferating pool, their cycle activity, and their survival. In accordance with published data,9,10 we found that imatinib inhibited 3H-thymidine incorporation. Earlier studies have suggested that the reduced expansion was largely due to inhibition of cell-cycle activity.1,9,10 Our in vitro experiments with CFSE-labeled naive and memory p14 T cells confirmed these earlier findings. In contrast, we found that imatinib does not affect cell-cycle entry or activity of memory CD8+ T cells in vivo. Yet, imatinib reduced the number of CTLs in vivo by inhibiting the survival of proliferating T cells. These contradictory observations may be explained by the fact that the requirements for T-cell expansion in vitro and in vivo differ substantially. T-cell expansion in vitro is induced by a defined TCR signal, often by αCD3 or phytohemagglutinin stimulus. On the other hand, cell cycling and survival of in vivo–restimulated T cells is a net result of TCR signals in conjunction with costimulatory signals such as CD28, CD27, 4-1BB, and cytokines secreted by bystander cells in secondary lymphoid organs.40,41 In vivo, these costimulatory signals together with the TCR stimulation seem to be sufficient for memory T cells to enter cell cycling. However, survival of proliferating cells becomes limiting in this situation, resulting in up to 3-fold lower frequencies of specific CTLs.
A selective inhibition of memory CTL expansion without affecting primary immune response to acute viral infections is quite a unique characteristic for an immunosuppressive drug. It may result in an impaired control of persistent infections, which are mainly CD8+ T-cell controlled. This knowledge could be important for the design of future clinical trials. Imatinib is increasingly used for the prevention of relapses in Ph+ leukemia after allogeneic stem cell transplantation.42,43 Cytomegalovirus (CMV) reactivation is a major problem in allogeneic bone marrow transplantations and CMV is mainly CD8+ T-cell controlled.44,45 Therefore, imatinib treatment may increase CMV reactivation in this setting. In addition, as suggested previously,1 imatinib may cause failure of leukemia control after stem cell transplantation by inhibiting the graft-versus-leukemia effect. In contrast, the selective inhibition of memory CTL expansion may be therapeutically useful in the treatment of immunopathologic sequels such as autoimmune diseases. Indeed, we could provide evidence that imatinib prevented the onset of diabetes after adoptive transfer of memory CTLs into RIP-GP mice. Therefore, our experiments may provide a pathophysiologic explanation for the observed improvement of blood-glucose concentrations in diabetic CML patients treated with imatinib.46,47 Taken together, our analyses of in vivo immune responses suggest that imatinib may reduce autoimmunity without the risk of acquiring opportunistic infections.
Prepublished online as Blood First Edition Paper, July 27, 2006; DOI 10.1182/blood-2006-04-018705.
Supported by grants from the Swiss National Science Foundation (632-66020), Oncosuisse (OCS-01312-02-2003 and OCS-01627-02-2005), and the Bernische Krebsliga. V.P. was supported by the Roche Research Foundation.
The authors declare no competing financial interests.
S.M. performed research, collected data, analyzed data, and wrote the manuscript; M.M. and V.P. performed research; and A.F.O. designed the overall research plan, reviewed the data, and wrote the manuscript.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
We thank Thomas Brunner for providing annexin V and Farzaneh Pirnia, Andrew Ziemiecki, and Marcel Merk for critical reading of the manuscript.




This feature is available to Subscribers Only
Sign In or Create an Account Close Modal