Abstract
Clinical observations suggest that in chronic myelogenous leukemia (CML), the Philadelphia chromosome (Ph+) clone has a growth advantage over normal hematopoiesis. Patients with CML have high levels of neutrophil elastase, which has recently been shown to antagonize the action of granulocyte-colony-stimulating factor (G-CSF) and other growth factors. We therefore compared the effect of elastase on the growth of normal and CML progenitor cells. In 10-day suspension cultures of normal or CML CD34+ cells supplemented with G-CSF, stem cell factor (SCF), and granulocyte macrophage-colony-stimulating factor (GM-CSF), CML cells had diminished sensitivity to the growth inhibitory effect of elastase. When equal numbers of CML and normal CD34+ cells were cocultured for 10 days, there was no change in the relative proportions of normal and leukemic cells (measured by fluorescence in situ hybridization [FISH] or flow cytometry). However, when elastase was added, CML cells predominated at the end of the culture period (78% vs 22% with 1 μg/mL and 80% vs 20% with 5 μg/mL elastase). CML neutrophils substituted effectively for elastase in suppressing the proliferation of normal CD34+ cells, but this effect was abrogated by serine protease inhibitors. These results suggest that elastase overproduction by the leukemic clone can change the growth environment by digesting growth factors, thereby giving advantage to Ph+ hematopoiesis. (Blood. 2003; 102:3786-3792)
Introduction
Chronic myelogenous leukemia (CML) associated with the t(9;22) Philadelphia chromosome (Ph+) is characterized by massive leukocytosis, with all stages of granulopoiesis represented in the circulation. Although residual normal stem cells persist in low numbers, the bone marrow and blood are dominated by cells originating from a Ph+ clone typically involving erythroid, granulocytic, megakaryocytic, and B-cell, but not T-cell, lineages. As the interval from diagnosis lengthens, the proportion of normal (Ph-) cells decreases, suggesting that the leukemic process increasingly outcompetes normal hematopoiesis.1 However, normal hematopoiesis can recover after treatment with imatinib,2 interferon-alpha,3 busulfan-induced aplasia,4 and granulocyte colony stimulating factor (G-CSF) mobilization.5 In some situations, successful treatment of the Ph+ clone can result in cytopenia, as has been observed after imatinib6 and busulfan7 treatment. Irreversible marrow aplasia can occur after successful eradication of CML relapsing after allogeneic stem cell transplantation following donor lymphocyte infusion.8
These observations indicate a dynamic interaction between normal and CML hematopoiesis, tending to favor the predominance of the Ph+ clone while diminishing the normal hematopoietic stem cell pool. The question is raised whether CML actively suppresses normal hematopoiesis or simply replaces normal cells by superior proliferation.9 CML cells can survive10 and grow11,12 in the absence of growth factors, giving them a growth advantage in conditions in which growth factor availability is low.13-15 The high granulocyte counts achieved in CML also suggest that CML has reduced sensitivity to regulatory feedback mechanisms that normally maintain neutrophil counts below 10 000 mm3. We recently showed that neutrophil elastase potently inhibits granulopoiesis by the enzymatic digestion of G-CSF. We proposed that elastase and other primary granule proteases provide negative feedback on granulopoiesis by controlling the availability of G-CSF, proportionate to the relative abundance of mature granulocytes.16 Based on these observations, we hypothesize here that the high leukocyte counts achieved in CML patients are a result of G-CSF independence, allowing continued proliferation despite the production of neutrophil proteases that digest G-CSF. A corollary is that normal granulopoiesis would be suppressed by the neutrophil protease-induced reduction in G-CSF availability. The high blood levels of elastase in CML17-20 support the possibility that normal hematopoiesis could be efficiently suppressed by the enlarged Ph+ clone.
In this study we show that, compared with normal cells, CML CD34+ cells have diminished sensitivity to the growth-inhibitory effect of elastase. Furthermore, CML neutrophils substitute effectively for elastase in suppressing the proliferation of normal CD34+ cells but lose their growth advantage over normal CD34+ cells in the presence of elastase inhibitors. These findings suggest a mechanism explaining the dominance of the Ph+ clone over normal hematopoiesis.
Patients, materials, and methods
Materials
Elastase (Elastin Product Company, Owensville, MO), was resuspended in 50% glycerol/50% 0.02 M sodium acetate (NaOAc) and stored at -70°C until used. In all experiments, 50% glycerol/50% 0.02 M NaOAc solution without elastase was used as a control. Recombinant human (rh) G-CSF was obtained from Amgen (Thousand Oaks, CA); (rh) granulocyte macrophage-colony-stimulating factor (GM-CSF) and (rh) stem cell factor (SCF) were purchased from Biosource (Camarillo, CA). StemSpan serum-free medium was purchased from StemCell Technologies (Vancouver, BC, Canada). Fluorescein isothiocyanate (FITC)-conjugated anti-CD66b mouse antibody was obtained from Beckman Coulter (Miami, FL), and the FITC-conjugated anti-CD34 mouse antibody and the FITC-conjugated anti-HLA-A2 mouse antibody were obtained from Becton Dickinson Biosciences (San Jose, CA). Phenylmethylsulfonyl fluoride (PMSF) was obtained from Sigma (St Louis, MO). CD34+ cells were selected using Dynal CD34 Progenitor Cell Selection (Dynal Biotech, Lake Success, NY). Flow cytometry acquisition and analysis were performed using a FACSCalibur (Becton Dickinson, San Jose, CA). The BCR/ABL probe was obtained from Vysis (Downers Grove, IL). Apoptosis was determined using the Annexin V-phycoerythrin (PE) apoptosis detection kit I (Becton Dickinson, San Jose, CA) Transwell plates (pore size, 0.4 μm) were obtained from Corning Incorporated Costar (Corning, NY). The ACK Cell Lysing Buffer was from BioWhittaker (Pittsburgh, PA). Proliferation assays were harvested on a Tomtec (Hamden, CT). 3H-thymidine uptake was determined using a 1205 Betaplate (Wallac, Gaithersburg, MD). A Beckman Coulter EPICS Altra Flow Cytometer (Miami, FL) was used to sort CD34+ cells.
Cells
Samples were obtained from patients after written informed consent. CD34+ cells were obtained from leukopheresis donations from healthy volunteers undergoing G-CSF stimulation and stem cell mobilization under National Heart, Lung, and Blood Institute (NHLBI) institutional review board-approved protocol 96-H-0049. CML blood samples and leukopheresis donations from patients with chronic-phase CML who had not received any treatment other than hydroxyurea were used as a source of neutrophils and CD34+ cells, respectively (NHLBI protocols 97-H-0099, 99-H-0046, 02-H-0111). Leukopheresis-derived mononuclear cells were obtained by Ficoll-Hypaque density separation, counted, and stored in liquid nitrogen until used. Positive selection of CD34+ cells was performed using the Dynal CD34 progenitor cell selection system. Purity of the CD34+ cells used was greater than 95%. Neutrophils from fresh peripheral blood samples were used immediately after removing peripheral blood mononuclear cells (PBMCs) by Ficoll-Hypaque and lysing red blood cells. Purity was greater than 90%.
Suspension cultures of CD34 cells
CD34+ cells (7.5 × 104) were plated in triplicate in 24-well plates in 1 mL StemSpan serum-free medium with varying additions of growth factors, with or without neutrophil serine proteases, at the concentration indicated. Plates were then incubated at 37°C, 5% CO2 for 10 days, at which time cells were counted using a ZM Coulter particle counter as follows: 200 μL each well was diluted to 20 mL with Isoton II, and cells larger than 5.5 μm were counted. Remaining cells were harvested, labeled with FITC-conjugated anti-CD66b or FITC-conjugated anti-CD34 mouse antibodies for 15 minutes at room temperature, washed, fixed in 1% paraformaldehyde, and analyzed by flow cytometry.
Proliferation assay
CD34+ cells (2 × 104) were plated in triplicate in 96-well, round-bottom plates in 200 μL StemSpan serum-free medium, supplemented with various growth factor combinations with or without neutrophil elastase at the concentration indicated in the figures. Plates were then incubated at 37°C in 5% CO2. Proliferation was assessed by 3H-thymidine incorporation (final concentration, 1 μCi/well [37 kBq/well]), added for the final 16 hours of culture. All assays were carried out in triplicate. The cultures were harvested on fiberglass paper, and incorporation of 3H-thymidine was determined by scintillation counting.
Measurement of cell death
Then 106 CML or normal CD34+ cells were plated in 12-well plates in 2 mL StemSpan serum-free medium with 25 ng/mL each of G-CSF, GM-CSF, and SCF, with or without 1 mg/mL neutrophil elastase. Plates were then incubated at 37°C, 5% CO2. Apoptosis was measured daily by flow cytometry by Annexin V-PE detection. Increased apoptosis in the presence of elastase was calculated as [% apoptosis with elastase -% apoptosis without elastase].
FISH coculture experiments
Normal (3.75 × 104) and CML CD34+ (3.75 × 104) cells were cocultured in 24-well plates in 1 mL StemSpan serum-free medium with 10 ng/mL GM-CSF, 10 ng/mL G-CSF, and 10 ng/mL SCF, with or without neutrophil serine proteases, at the concentrations indicated in the figures. Plates were then incubated at 37°C, 5% CO2 for 8 days, at which time cells were counted, harvested, treated with hypotonic buffer consisting of KCl, HEPES (N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid), ethyleneglycotetraacetic acid (EGTA), and NaOH to expose the nucleus at interphase, and fixed onto slides using methanol and acetic acid (3:1). FISH was performed with probes for BCR/ABL. Slides were denatured by immersion in formamide/20 × saline sodium citrate (SSC) solution for 5 minutes at 73°C followed by several washes in 70%, 85%, and 100% ethanol at room temperature for 1 minute each. Probes were denatured as above and applied to the cells on the slides to hybridize at 42°C overnight. After hybridization, the slides were washed in prewarmed 0.4 × SSC at 73°C for 2 minutes and 2 × SSC/0.1% Nonidet P-40 (NP-40) at room temperature for 1 minute. The slides were allowed to dry in the dark and were counterstained with DAPI-II (4,6-diamidino-2-phenylindole-II) for enumeration using a fluorescence microscope.
Flow cytometry coculture experiments
Normal and patient CD34+ cells (3.75 × 104) were cocultured in 24-well plates in 1 mL StemSpan serum-free medium with 10 ng/mL GM-CSF, 10 ng/mL G-CSF, and 10 ng/mL SCF, with or without neutrophil serine proteases, at the concentrations shown in the figures. To increase the comparability of the normal and leukemic populations, cells were first sorted by size and granularity, where indicated, using a Coulter flow cytometer. Normal CD34+ cells were from an HLA-A2 sample, whereas the patient cells had a different HLA-A type. The plates were then incubated at 37°C in 5% CO2 for 8 days, at which time cells were counted, harvested, labeled with FITC-conjugated anti-HLA-A2 for 15 minutes at room temperature, washed, fixed in 1% paraformaldehyde, and analyzed by flow cytometry. Patient or normal CD34+ cells cultured alone in the same condition were used as controls.
Transwell coculture experiments
Transwell plates (12 wells) were used as follows: the lower chamber was filled with a mixture of 105 normal and 105 patient CD34+ cells in 1.5 mL StemSpan serum-free medium with 10 ng/mL GM-CSF, 10 ng/mL G-CSF, and 10 ng/mL SCF (final concentration). The upper chamber of the transwell was filled with 0.5 mL StemSpan serum-free medium containing CML neutrophils at the concentrations indicated in the figures. Alternatively, PMSF (final concentration, 625 μM) or fetal calf serum (FCS) (final concentration, 1%) was added to the medium before the neutrophils were added. Normal CD34+ cells were from an HLA-A2 sample, whereas the patient CD34+ cells were from a different HLA-A type. Plates were then incubated at 37°C in 5% CO2 for 8 days, at which time cells were counted, harvested, labeled with FITC-conjugated anti-HLA-A2 for 15 minutes at room temperature, washed, fixed in 1% paraformaldehyde, and analyzed by flow cytometry. Patient or normal CD34+ cells cultured alone in the presence of neutrophils was used as a control.
Statistical analysis
Values shown are the mean ± SEM. Significant differences between groups were evaluated using the Student 2-tailed t test or the Mann-Whitney U test. Percentage inhibition was calculated as: 100 - [(mean of cell concentration (or cpm) in elastase-stimulated wells/mean of cell concentration (or cpm) in unstimulated cultures) × 100].
Results
CML cells proliferate and survive in elastase-induced growth factor deprivation
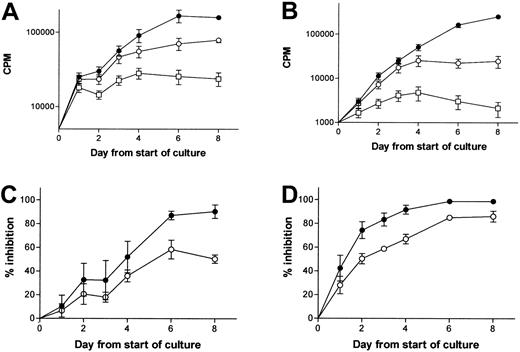
Patient CD34+ cells were cultured with 10 ng/mL G-CSF in the presence or absence of elastase. Proliferation was measured daily by 3H-thymidine uptake. As expected, elastase abrogated the proliferation induced by G-CSF, reducing proliferation back to the rate of unstimulated patient CD34+ cells. Concentrations of elastase greater than 1 μg/mL also blocked the stimulatory effect of 10 ng/mL GM-CSF. These findings confirm the responsiveness of patient CD34+ cells to G-CSF and GM-CSF and their continued ability to proliferate when growth factors are removed by elastase. In contrast, in the presence of G-CSF and elastase, or with no growth factor, normal CD34+ cells did not proliferate. When cultured in the presence of GM-CSF, G-CSF, and SCF, patient CD34+ cells resisted the apoptosis induced by the destruction of G-CSF by elastase, in contrast to normal CD34+ cells. We also noted that elastase had no direct inhibitory effect on patient CD34+ cells because patient CD34+ cells proliferated equally well in the presence or absence of 1 μg/mL elastase when stimulated by GM-CSF (which was not digested by elastase at the concentration used) (Figure 1).
Effect of elastase on normal and CML CD34+ growth. 2 × 104 CML or normal CD34+ cells/mL were cultured in 200 μL serum-free medium with or without cytokine. Daily measurement of tritiated thymidine uptake by 10 ng/mL G-CSF-stimulated CML (A) or normal (B) CD34+ cells (•, control; ○, 1 μg/mL elastase; ▪, no cytokine). Data represent mean ± SEM of triplicates and are representative of 2 different experiments. (C) Daily measurement of tritiated thymidine uptake by 10 ng/mL GM-CSF-stimulated CML CD34+ cells (•, control; ○, 1 μg/mL elastase; ▪,no cytokine; □, 5 μg/mL elastase). Data represent mean ± SEM of triplicates and are representative of 2 different experiments. (D) 106 CML or normal CD34+ cells were plated in 12-well plates in 2 mL serum-free medium with 25 ng/mL G-CSF, GM-CSF, and SCF. Plates were then incubated at 37°C in 5% CO2. Apoptosis was measured daily. Increased apoptosis in the presence of elastase was calculated as [% apoptosis in presence of elastase -% apoptosis without elastase] (•,normal CD34+ cells; ○, CML CD34+ cells).
Effect of elastase on normal and CML CD34+ growth. 2 × 104 CML or normal CD34+ cells/mL were cultured in 200 μL serum-free medium with or without cytokine. Daily measurement of tritiated thymidine uptake by 10 ng/mL G-CSF-stimulated CML (A) or normal (B) CD34+ cells (•, control; ○, 1 μg/mL elastase; ▪, no cytokine). Data represent mean ± SEM of triplicates and are representative of 2 different experiments. (C) Daily measurement of tritiated thymidine uptake by 10 ng/mL GM-CSF-stimulated CML CD34+ cells (•, control; ○, 1 μg/mL elastase; ▪,no cytokine; □, 5 μg/mL elastase). Data represent mean ± SEM of triplicates and are representative of 2 different experiments. (D) 106 CML or normal CD34+ cells were plated in 12-well plates in 2 mL serum-free medium with 25 ng/mL G-CSF, GM-CSF, and SCF. Plates were then incubated at 37°C in 5% CO2. Apoptosis was measured daily. Increased apoptosis in the presence of elastase was calculated as [% apoptosis in presence of elastase -% apoptosis without elastase] (•,normal CD34+ cells; ○, CML CD34+ cells).
Normal hematopoiesis is more sensitive than CML hematopoiesis to elastase inhibition
We then compared the inhibitory activity of elastase on normal and patient CD34+ cell proliferation stimulated by SCF, G-CSF, and GM-CSF. In experiments comparing normal and CML CD34+ cells, proliferation was reduced by elastase in both, but normal cells were more powerfully inhibited with 1 μg/mL elastase (90% ± 1% vs 57% ± 8%) and with 5 μg/mL elastase (98% ± 0.3% vs 90% ± 3%) (P < .01). This was reflected in higher cell counts in CML patients at the end of the 10-day culture period (Figures 2, 3; Table 1). CML cells had decreased sensitivity to elastase at all the concentrations tested between 100 and 5000 ng/mL. The proportions of cells in the early (CD34+), intermediate (CD34-, CD66b-), and late (CD66b+) granulocyte maturation compartments also differed. There was a higher proportion of CML cells after 10 days in the intermediate and late compartments than CD34+ cells, whereas normal cultures showed less ability to progress to mature neutrophils with a higher proportion of CD34+ cells and a lower proportion of late compartments (Figure 3; Table 1).
Normal CD34+ cell proliferation is more sensitive to elastase inhibition than CML CD34+ cells. 2 × 104 CML or normal CD34+ cells/mL were cultured in 200 μL serum-free media in the presence of 10 ng/mL G-CSF, GM-CSF, and SCF with different concentrations of elastase. Proliferation was measured daily by tritiated thymidine uptake. (A) CML CD34+ cells (•, control; ○, 1 μg/mL elastase; □, 5 μg/mL elastase). (B) Normal CD34+ cells (•, control; ○, 1 μg/mL elastase; □,5 μg/mL elastase). (C-D) Percentage inhibition induced by, respectively, 1 or 5 μg/mL elastase (•, normal CD34+ cells; ○, CML CD34+ cells). Data represent mean ± SEM of 3 experiments.
Normal CD34+ cell proliferation is more sensitive to elastase inhibition than CML CD34+ cells. 2 × 104 CML or normal CD34+ cells/mL were cultured in 200 μL serum-free media in the presence of 10 ng/mL G-CSF, GM-CSF, and SCF with different concentrations of elastase. Proliferation was measured daily by tritiated thymidine uptake. (A) CML CD34+ cells (•, control; ○, 1 μg/mL elastase; □, 5 μg/mL elastase). (B) Normal CD34+ cells (•, control; ○, 1 μg/mL elastase; □,5 μg/mL elastase). (C-D) Percentage inhibition induced by, respectively, 1 or 5 μg/mL elastase (•, normal CD34+ cells; ○, CML CD34+ cells). Data represent mean ± SEM of 3 experiments.
Normal CD34+ cell differentiation is blocked by elastase. 7.5 × 104 normal or CML CD34+ cells/mL were cultured in 1 mL serum-free medium in the presence of 10 ng/mL G-CSF, GM-CSF, and SCF with different concentrations of elastase. After 10 days of culture nucleated cells were counted, harvested, and labeled with anti-CD66b and anti-CD34 antibodies. (A) Number of cells produced in response to increasing dose of elastase. Data represent mean ± SEM of triplicates (▪, normal CD34+ cells; □, CML CD34+ cells 1 μg/mL elastase). (B) Percentage inhibition induced by 1 or 5 μg/mL elastase. Data represent mean ± SEM of 4 different experiments (•, normal CD34+ cells; ○, CML CD34+ cells). *Indicates significant differences from normal values (*P < .01). Phenotype of normal (C) or CML (D) CD34+ cells after 10 days of culture in the presence of 0, 1, or 5 μg/mL elastase (▦, CD34 cells; □, intermediate cells; ▪, CD66b cells). Data represent mean ± SEM of 3 experiments.
Normal CD34+ cell differentiation is blocked by elastase. 7.5 × 104 normal or CML CD34+ cells/mL were cultured in 1 mL serum-free medium in the presence of 10 ng/mL G-CSF, GM-CSF, and SCF with different concentrations of elastase. After 10 days of culture nucleated cells were counted, harvested, and labeled with anti-CD66b and anti-CD34 antibodies. (A) Number of cells produced in response to increasing dose of elastase. Data represent mean ± SEM of triplicates (▪, normal CD34+ cells; □, CML CD34+ cells 1 μg/mL elastase). (B) Percentage inhibition induced by 1 or 5 μg/mL elastase. Data represent mean ± SEM of 4 different experiments (•, normal CD34+ cells; ○, CML CD34+ cells). *Indicates significant differences from normal values (*P < .01). Phenotype of normal (C) or CML (D) CD34+ cells after 10 days of culture in the presence of 0, 1, or 5 μg/mL elastase (▦, CD34 cells; □, intermediate cells; ▪, CD66b cells). Data represent mean ± SEM of 3 experiments.
Inhibition of normal and CML CD34+ cells by elastase
. | Total cell no. × 105 cells/mL . | . | . | Proliferation index . | . | . | Inhibition % . | . | CD34 after d10% . | . | . | CD34 after d10 × 104 cells/mL . | . | . | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | Elastase μg/mL . | . | . | . | . | . | . | . | . | . | . | . | . | . | |||||||||||||
. | 0 . | 1 . | 5 . | 0 . | 1 . | 5 . | 1 . | 5 . | 0 . | 1 . | 5 . | 0 . | 1 . | 5 . | |||||||||||||
| Donor | |||||||||||||||||||||||||||
| 1 | 53 | 2.9 | 0.9 | 71 | 4 | 1 | 94 | 98 | 3.2 | 12.8 | 16.3 | 17 | 3.7 | 1.5 | |||||||||||||
| 2 | 39 | 5.9 | 1.3 | 52 | 8 | 2 | 85 | 97 | 5.1 | 16.6 | 38.9 | 20 | 9.8 | 5.1 | |||||||||||||
| 3 | 47 | 5.4 | 0.9 | 63 | 7 | 1 | 89 | 98 | 5.8 | 16.1 | 25.2 | 27 | 8.7 | 2.3 | |||||||||||||
| 4 | 36 | 3.6 | 0.3 | 48 | 5 | 1 | 90 | 99 | 3.8 | 15 | 29.6 | 14 | 5.4 | 0.9 | |||||||||||||
| 5 | 22 | 2.3 | 0.3 | 30 | 3 | 1 | 90 | 99 | 1.5 | 10.9 | 23.8 | 3.3 | 2.5 | 0.7 | |||||||||||||
| 6 | 30 | 1.7 | 0.5 | 40 | 2 | 1 | 94 | 98 | ND | ND | ND | ND | ND | ND | |||||||||||||
| CML | |||||||||||||||||||||||||||
| 1 | 42 | 8.6 | 2.3 | 56 | 11 | 3 | 79 | 95 | 0.8 | 0.6 | 0.6 | 3.4 | 0.5 | 0.1 | |||||||||||||
| 2 | 57 | 11 | 4.8 | 76 | 15 | 6 | 80 | 92 | 1.8 | 3.5 | 4.7 | 10. | 3.4 | 2.3 | |||||||||||||
| 3 | 54 | 34 | 13 | 72 | 45 | 18 | 38 | 75 | 3 | 2.7 | 4.7 | 16. | 9.1 | 7.3 | |||||||||||||
| 4 | 29 | 14 | 2.4 | 38 | 19 | 3 | 50 | 92 | 3.6 | 5.3 | 9.8 | 10. | 3.4 | 2.3 | |||||||||||||
| 5 | 32 | 11 | 1.4 | 43 | 14 | 2 | 66 | 96 | 0.8 | 1.9 | 9.4 | 2.6 | 2.1 | 1.3 | |||||||||||||
| 6 | 38 | 14 | 2.9 | 50 | 18 | 4 | 64 | 92 | 3.6 | 5.1 | 7.1 | 14 | 6.9 | 2.1 | |||||||||||||
| 7 | 44 | 42 | 7.9 | 58 | 18 | 3 | 68 | 95 | ND | ND | ND | ND | ND | ND | |||||||||||||
| 8 | 51 | 42 | 7.9 | 69 | 56 | 11 | 18 | 85 | ND | ND | ND | ND | ND | ND | |||||||||||||
. | Total cell no. × 105 cells/mL . | . | . | Proliferation index . | . | . | Inhibition % . | . | CD34 after d10% . | . | . | CD34 after d10 × 104 cells/mL . | . | . | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | Elastase μg/mL . | . | . | . | . | . | . | . | . | . | . | . | . | . | |||||||||||||
. | 0 . | 1 . | 5 . | 0 . | 1 . | 5 . | 1 . | 5 . | 0 . | 1 . | 5 . | 0 . | 1 . | 5 . | |||||||||||||
| Donor | |||||||||||||||||||||||||||
| 1 | 53 | 2.9 | 0.9 | 71 | 4 | 1 | 94 | 98 | 3.2 | 12.8 | 16.3 | 17 | 3.7 | 1.5 | |||||||||||||
| 2 | 39 | 5.9 | 1.3 | 52 | 8 | 2 | 85 | 97 | 5.1 | 16.6 | 38.9 | 20 | 9.8 | 5.1 | |||||||||||||
| 3 | 47 | 5.4 | 0.9 | 63 | 7 | 1 | 89 | 98 | 5.8 | 16.1 | 25.2 | 27 | 8.7 | 2.3 | |||||||||||||
| 4 | 36 | 3.6 | 0.3 | 48 | 5 | 1 | 90 | 99 | 3.8 | 15 | 29.6 | 14 | 5.4 | 0.9 | |||||||||||||
| 5 | 22 | 2.3 | 0.3 | 30 | 3 | 1 | 90 | 99 | 1.5 | 10.9 | 23.8 | 3.3 | 2.5 | 0.7 | |||||||||||||
| 6 | 30 | 1.7 | 0.5 | 40 | 2 | 1 | 94 | 98 | ND | ND | ND | ND | ND | ND | |||||||||||||
| CML | |||||||||||||||||||||||||||
| 1 | 42 | 8.6 | 2.3 | 56 | 11 | 3 | 79 | 95 | 0.8 | 0.6 | 0.6 | 3.4 | 0.5 | 0.1 | |||||||||||||
| 2 | 57 | 11 | 4.8 | 76 | 15 | 6 | 80 | 92 | 1.8 | 3.5 | 4.7 | 10. | 3.4 | 2.3 | |||||||||||||
| 3 | 54 | 34 | 13 | 72 | 45 | 18 | 38 | 75 | 3 | 2.7 | 4.7 | 16. | 9.1 | 7.3 | |||||||||||||
| 4 | 29 | 14 | 2.4 | 38 | 19 | 3 | 50 | 92 | 3.6 | 5.3 | 9.8 | 10. | 3.4 | 2.3 | |||||||||||||
| 5 | 32 | 11 | 1.4 | 43 | 14 | 2 | 66 | 96 | 0.8 | 1.9 | 9.4 | 2.6 | 2.1 | 1.3 | |||||||||||||
| 6 | 38 | 14 | 2.9 | 50 | 18 | 4 | 64 | 92 | 3.6 | 5.1 | 7.1 | 14 | 6.9 | 2.1 | |||||||||||||
| 7 | 44 | 42 | 7.9 | 58 | 18 | 3 | 68 | 95 | ND | ND | ND | ND | ND | ND | |||||||||||||
| 8 | 51 | 42 | 7.9 | 69 | 56 | 11 | 18 | 85 | ND | ND | ND | ND | ND | ND | |||||||||||||
ND indicates not determined.
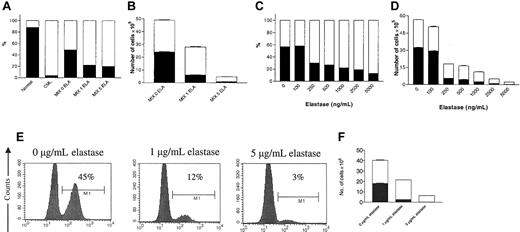
Elastase confers a growth advantage to CML CD34+ cells in coculture with normal CD34+ cells
Equal numbers of normal and patient CD34+ cells were cocultured in GM-CSF, G-CSF, and SCF with 0, 1, or 5 μg/mL elastase for 8 days. Cells were then counted and analyzed for the expression of the BCR/ABL fusion gene using FISH. In the absence of elastase there was no significant change in the proportion of normal and CML cells (49% vs 51%), but when elastase was added, the ratio of normal to CML cells decreased to 22%:78% with 1 μg/mL elastase and 20%:80% with 5 μg/mL elastase. The total number of viable nucleated cells at the end of the culture decreased with increasing doses of elastase (1 μg and 5 μg/mL) in both populations, but more in normal cell cultures—from 24 × 105 cells/mL to 6 × 105 and 1 × 105 for normal cells and from 25 × 105 to 22 × 105 and 4 × 105 for CML cells (Figure 4A-B).
Elastase gives a growth advantage to CML cells in cocultures of normal and CML CD34+ cells. Mixtures of 4 × 104 normal (HLA-A2+) and CML (HLA-A2-) CD34+ cells/mL were cultured in 1 mL serum-free medium in the presence of 10 ng/mL G-CSF, GM-CSF, and SCF with different concentrations of elastase. After 8 days of culture, nucleated cells were counted, harvested, and analyzed for BCR/ABL or HLA-A2 expression. CML and normal cells cultured separately under the same conditions were used as controls. (A) Percentage and (B) absolute number of BCR/ABL-positive and -negative cells in the presence of 0, 1, or 5 μg/mL elastase (▪, BCR/ABL- cells; □, BCR/ABL+ cells). Data are representative of 2 independent experiments. (C) Percentage and (D) absolute number of HLA-A2-positive (normal) and -negative (CML) cells in the presence of increasing doses of elastase (▪, HLA-A2+ cells; □, HLA-A2-). Data represent mean ± SEM of triplicates. CML (HLA-A2-) and normal (HLA-A2+) CD34+ cells were sorted by size and granularity and cultured in the same conditions mentioned. After 8 days of culture, nucleated cells were counted, harvested, and analyzed for HLA-A2 expression by FACS. (E) Percentage and (F) absolute number of HLA-A2+ (normal) and HLA-A2- (CML) cells in the presence of 0, 1, or 5 μg/mL elastase (▪, HLA-A2+ cells; □; HLA-A2-). Data represent mean ± SEM of triplicates) and are representative of 2 independent experiments.
Elastase gives a growth advantage to CML cells in cocultures of normal and CML CD34+ cells. Mixtures of 4 × 104 normal (HLA-A2+) and CML (HLA-A2-) CD34+ cells/mL were cultured in 1 mL serum-free medium in the presence of 10 ng/mL G-CSF, GM-CSF, and SCF with different concentrations of elastase. After 8 days of culture, nucleated cells were counted, harvested, and analyzed for BCR/ABL or HLA-A2 expression. CML and normal cells cultured separately under the same conditions were used as controls. (A) Percentage and (B) absolute number of BCR/ABL-positive and -negative cells in the presence of 0, 1, or 5 μg/mL elastase (▪, BCR/ABL- cells; □, BCR/ABL+ cells). Data are representative of 2 independent experiments. (C) Percentage and (D) absolute number of HLA-A2-positive (normal) and -negative (CML) cells in the presence of increasing doses of elastase (▪, HLA-A2+ cells; □, HLA-A2-). Data represent mean ± SEM of triplicates. CML (HLA-A2-) and normal (HLA-A2+) CD34+ cells were sorted by size and granularity and cultured in the same conditions mentioned. After 8 days of culture, nucleated cells were counted, harvested, and analyzed for HLA-A2 expression by FACS. (E) Percentage and (F) absolute number of HLA-A2+ (normal) and HLA-A2- (CML) cells in the presence of 0, 1, or 5 μg/mL elastase (▪, HLA-A2+ cells; □; HLA-A2-). Data represent mean ± SEM of triplicates) and are representative of 2 independent experiments.
In another experiment CD34+ cells from a normal HLA-A2+ sample were cocultured with those from a non-HLA-A2 (HLA-A2-) CML sample. After 8 days of culture the cells were counted, and the percentage of HLA-A2+ cells in the culture was determined by flow cytometry using murine anti-HLA-A2. As controls, normal and patient CD34+ cells were cultured separately with or without elastase. No effect of elastase was observed on the HLA-A2 expression of normal or CML cells (data not shown). As with the previous experiment, elastase inhibited normal and patient CD34+ cells but favored the ratio of CML cells over normal cells in a dose-dependent manner (Figure 4C-D).
The Clarkson group21 showed that CML CD34+ cells are more mature than normal CD34+ cells and may react differently to growth and to apoptotic stimuli. Therefore, to increase comparability between leukemic and normal CD34 cells, we sorted CML (HLA-A2-) and normal (HLA-A2+) CD34+ cells by their size and granularity and cocultured similarly gated populations in GM-CSF, G-CSF, and SCF with 0, 1, or 5 μg/mL elastase for 8 days. As observed in Figure 4E-F, the selected CML cells still had an advantage over the normal selected cells when cultured in the presence of elastase.
Neutrophils provide a growth advantage to CML cells that is reversed by serine protease inhibition
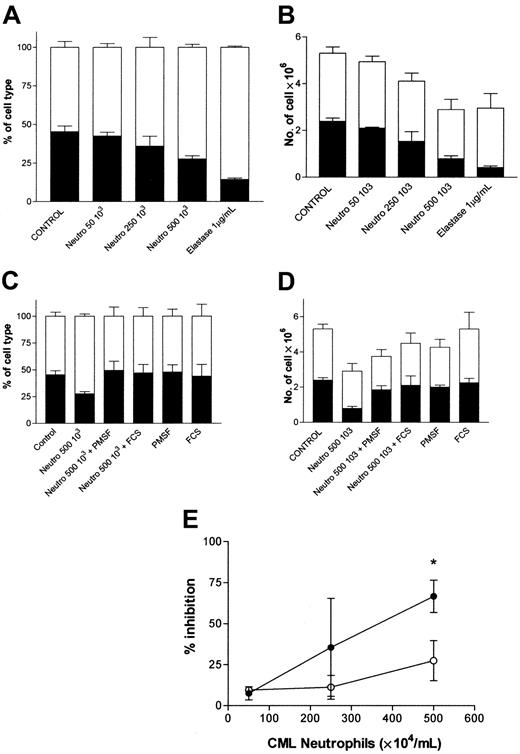
Because elastase is mainly expressed by neutrophils and their precursors, we investigated the possibility of providing a growth advantage to CML cells by culturing normal and leukemic CD34+ cell mixtures with CML neutrophils. In 3 experiments normal (HLA-A2+) and patient (HLA-A2-) CD34+ cells were cultured with GM-CSF, G-CSF, and SCF. Increasing numbers of CML neutrophils (5 × 104/mL, 25 × 104/mL, or 50 × 104/mL) were added in a transwell allowing only soluble factor exchanges with the cultured CD34+ cells. To block serine protease activity, PMSF or FCS was added to the culture medium before exposure to neutrophils. Elastase was used as a positive control. After 8 days of culture, cells were counted and analyzed for HLA-A2 expression. Figure 5 shows that CML neutrophils inhibited normal and CML CD34+ cells, comparable to the effect of elastase. However, normal cells were significantly more sensitive to this inhibition than CML cells. In the presence of 50 × 104 CML neutrophils/mL, we observed, respectively, 67% ± 6% inhibition of normal cells versus 27% ± 12% for CML cells (P < .05). This advantage was reversed by the serine protease inhibitor PMSF and by 1% FCS. Similar effects were seen with normal neutrophils (data not shown).
CML neutrophils confer growth advantage to CML cells. Mixtures of 105 normal (HLA-A2) and CML (HLA-A2-) CD34+ cells were cultured in 2 mL serum-free medium supplemented with 10 ng/mL G-CSF, GM-CSF, and SCF in the presence of varying concentrations of CML neutrophils. After 8 days of culture, nucleated cells were counted, harvested, and analyzed for the percentage expression of HLA-A2. (A) Percentage and (B) absolute number of HLA-A2-positive (normal) and HLA-A2- (CML) cells in the presence of increasing numbers of CML neutrophils (▪, HLA-A2+ cells; □, HLA-A2- cells). (C) Percentage and (D) absolute number of HLA-A2+ (normal) and -negative (CML) cells in the presence of CML neutrophils and protease inhibitor (▪, HLA-A2+ cells; □, HLA-A2-). (E) Percentage inhibition induced by CML neutrophils. Data represent mean ± SEM of 3 experiments (•, normal CD34+ cells; ○, CML CD34+ cells). *Indicates significant differences from normal values (*P < .05).
CML neutrophils confer growth advantage to CML cells. Mixtures of 105 normal (HLA-A2) and CML (HLA-A2-) CD34+ cells were cultured in 2 mL serum-free medium supplemented with 10 ng/mL G-CSF, GM-CSF, and SCF in the presence of varying concentrations of CML neutrophils. After 8 days of culture, nucleated cells were counted, harvested, and analyzed for the percentage expression of HLA-A2. (A) Percentage and (B) absolute number of HLA-A2-positive (normal) and HLA-A2- (CML) cells in the presence of increasing numbers of CML neutrophils (▪, HLA-A2+ cells; □, HLA-A2- cells). (C) Percentage and (D) absolute number of HLA-A2+ (normal) and -negative (CML) cells in the presence of CML neutrophils and protease inhibitor (▪, HLA-A2+ cells; □, HLA-A2-). (E) Percentage inhibition induced by CML neutrophils. Data represent mean ± SEM of 3 experiments (•, normal CD34+ cells; ○, CML CD34+ cells). *Indicates significant differences from normal values (*P < .05).
Discussion
Evidence from in vitro experiments suggests that the CML clone has a growth advantage over residual normal hematopoiesis because it survives and proliferates in conditions of growth factor deprivation.11,12,15,22 How CML could establish such a favorable low growth factor environment to promote its clonal dominance is unclear. In this study we suggest a role for neutrophil elastase. Neutrophil elastase is a serine protease that avidly digests G-CSF, and we recently proposed that neutrophil elastase contributes to the regulation of normal granulopoiesis by negative feedback on G-CSF.15 Interestingly, in CML-CP, abnormally high concentrations of elastase occur in serum.17-20 As shown by Morishita et al,20 the concentration of elastase in CML at the time of diagnosis is increased more than 10-fold—from 67 ± 44 ng/mL in normal samples to 743 ± 415 ng/mL in CML samples—and can exceed 3000 ng/mL. They also found that elastase levels correlated with the leukocyte count: when the count is reduced by hydroxyurea treatment, the serum level of elastase also decreases. In support of a special role for elastase in CML is the observation that G-CSF is low or undetectable in CML-CP and is inversely correlated with the leukocyte count, suggesting that the production of large amounts of elastase from CML neutrophils might contribute to the clonal dominance of CML by reducing the availability of growth factors for normal hematopoiesis.13 However, in patients in the accelerated phase or blast crisis of CML, G-CSF levels approach normal despite elevated leukocyte counts.23 It is likely that the effects of elastase in patients in accelerated phase or blast crisis are abrogated by increased G-CSF production24 and a decrease in elastase production caused by the failure of neutrophil maturation as the disease progresses.25-28 Therefore, we restricted our study to patients in CML-CP.
Here we provide further evidence that elastase production by CML neutrophils preferentially suppresses normal hematopoiesis. First we found that though G-CSF-dependent proliferation of CML was abrogated by elastase, leukemic CD34+ cells, unlike normal cells, could proliferate equally in the presence of elastase or without any added growth factor. Similar effects were found with GM-CSF at higher elastase concentrations. CML CD34+ cells, unlike normal CD34+ cells, were resistant to the apoptosis induced by elastase through G-CSF destruction. When cultures of normal and CML CD34+ cells were compared, normal CD34+ cells were found to be more sensitive to inhibition by elastase than CML cells over a range of enzyme concentrations, and this was associated with a relative failure of maturation to neutrophils in the healthy volunteers. In coculture experiments, elastase inhibited the proliferation of normal and CML cells, but the proportion of CML versus normal cells was higher at the end of the culture period.
The in vivo action of increased elastase on the bone marrow microenvironment of CML patients could explain several features of CML pathophysiology: because CML CD34+ cells have been shown to be more mature than normal CD34+ cells,21 their ability to grow better in the presence of elastase could be attributed to a more advanced stage of maturation. We therefore obtained CML and normal CD34+ cells comparable in size and granularity by flow sorting. We found that CML CD34+ cells retained their advantage over comparable populations of normal CD34+ cells when cultured in the presence of elastase. Our findings are in agreement with recent observations that after 4 days of culture in GM-CSF and G-CSF, the proliferation of normal CD34+ cells is more affected than that of CML CD34+ cells if the cells are subsequently cultured in a low or an inadequate growth factor environment.15 These investigators also showed that the proliferative advantage of CML cells in growth factor deprivation is directly dependent on the BCR/ABL oncogene because it was possible to reverse the growth advantage with a specific tyrosine kinase inhibitor.15
Given that neutrophil elastase is essentially expressed by neutrophils and their precursors, the effect of elastase derived from CML neutrophils was tested in cocultures of normal and patient CD34+ cells. CML neutrophils more strongly inhibited the proliferation of normal rather than patient CD34+ cells. The leukemic growth advantage was blocked by adding the antiserine protease agent PMSF or by FCS (as a source of the elastase inhibitor α-1 antitrypsin), suggesting that elastase or other primary granule enzymes mediated the inhibitory effect.
No effect of elastase was observed on CML or normal CD34+ cells when GM-CSF was used as the growth factor. Thus, elastase exerted its growth-modulating effects solely by digesting G-CSF. Our experiments do not, however, address the mechanism whereby CML cells survive and proliferate when deprived of growth factor. This property is inevitably linked to the BCR/ABL oncogene,10,15,29 and several molecular mechanisms have been proposed to explain the growth and survival of CML cells in low growth factor concentrations.15,24,29-32 Nevertheless, the in vivo action of increased elastase within the bone marrow of CML patients may explain several features of CML pathophysiology:
First, our findings may explain the tendency of the Ph+ clone to dominate the marrow: hematopoiesis is the result of a balance between positive effects of growth factors (G-CSF, GM-CSF, SCF, and interleukin-3 [IL-3]) and negative factors (transforming growth factor-β [TGF-β], tumor necrosis factor-α [TNF-α], macrophage inflammatory protein 1α [MIP-1α] and monocyte chemoattractant protein-1 [MCP-1]).22,33 A less well-defined negative regulator, the leukemia-associated inhibitor (LAI) isolated from conditioned media of myeloid leukemic cells, inhibits normal but not leukemic granulopoiesis.34 Recently, LAI was identified as pro-proteinase 3, a member of the neutrophil serine protease family that includes elastase, azurocidin, and cathepsin G, found in primary (azurophilic) granules of granulocytes.35 In mice, neutrophil elastase, cathepsin G, proteinase 3, and matrix metalloproteinase-9 cleave the SCF receptor (CD117) on hematopoietic progenitors after G-CSF administration.36 Leukocytosis in CML results in the overproduction of granule proteins, including elastase, which could, therefore, negatively affect normal hematopoietic regulation by growth factor inactivation and SCF-receptor digestion. Because CML cells survive and proliferate without growth factors, the leukemic clone would have a growth advantage. This advantage would be abrogated if neutropenia were induced by intensive therapy. After myeloablative treatment and in the absence of neutrophils, normal and CML progenitors should be equally responsive to the high concentrations of growth factor released. Indeed, transient recovery of normal Ph-negative hematopoiesis is frequently observed after autologous stem cell transplantation for CML9,37 or after busulfan-induced aplasia.7
Second, the observation that CML retains some susceptibility to a regulatory action of elastase may explain the phenomenon observed in CML of cyclical neutrophilia,38,39 with an inverse cycling of myeloblasts in DNA synthesis.40 Invoking the same mechanism that we have described for normal hematopoiesis,16 high elastase levels in CML may provide negative feedback, switching off growth factor function until neutrophil production, and hence elastase production, drops. However, because of the intrinsic ability of CML to proliferate without growth factors, the nadir of the cycle is well above the upper limit of the normal range.
Third, it has recently been shown that in transforming CML, the G-CSF receptor is progressively down-regulated by increased production of the BCR-ABL fusion protein.27,28 The low G-CSF environment associated with elastase overproduction in CML may therefore favor the emergence of blastic-phase cells by positive selection of G-CSF-independent subclones.
Fourth, mobilizing CD34+ cells from the marrow to the periphery after G-CSF injection appears to be related to digestion of the adhesion molecules CXCR4, SDF-1, and vascular cell adhesion molecule-1 (VCAM-1) by matrix metalloproteinase-9 (MMP-9) and neutrophil serine proteases, including elastase.41-43 Sustained high concentrations of these proteases18,44,45 in CML could be responsible for the characteristic spilling of juvenile marrow cells into the blood in CML.
In conclusion, we show here that normal and CML hematopoietic progenitors proliferate differently in the presence of neutrophil elastase. These findings may explain some of the characteristic features of CML behavior. However, it is also clear that neutrophil elastase is only one of a family of biologically potent serine proteases exerting similar effects on hematopoiesis. In vitro and vivo studies to examine other proteases and to further substantiate their role in granulopoietic regulation in normal and leukemic states are indicated.
Prepublished online as Blood First Edition Paper, July 31, 2003; DOI 10.1182/blood-2003-03-0861.
Supported by grants from MEDIC Foundation.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Philippe Martiat, Dr Philippe Lewalle, Dr Redouane Rouas, Dr Mathias Grube, and Dr Minocher Battiwalla for their help.

![Figure 1. Effect of elastase on normal and CML CD34+ growth. 2 × 104 CML or normal CD34+ cells/mL were cultured in 200 μL serum-free medium with or without cytokine. Daily measurement of tritiated thymidine uptake by 10 ng/mL G-CSF-stimulated CML (A) or normal (B) CD34+ cells (•, control; ○, 1 μg/mL elastase; ▪, no cytokine). Data represent mean ± SEM of triplicates and are representative of 2 different experiments. (C) Daily measurement of tritiated thymidine uptake by 10 ng/mL GM-CSF-stimulated CML CD34+ cells (•, control; ○, 1 μg/mL elastase; ▪,no cytokine; □, 5 μg/mL elastase). Data represent mean ± SEM of triplicates and are representative of 2 different experiments. (D) 106 CML or normal CD34+ cells were plated in 12-well plates in 2 mL serum-free medium with 25 ng/mL G-CSF, GM-CSF, and SCF. Plates were then incubated at 37°C in 5% CO2. Apoptosis was measured daily. Increased apoptosis in the presence of elastase was calculated as [% apoptosis in presence of elastase -% apoptosis without elastase] (•,normal CD34+ cells; ○, CML CD34+ cells).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/102/10/10.1182_blood-2003-03-0861/6/m_h82235244001.jpeg?Expires=1769944315&Signature=jLq-zqHYLrxLQbpWqBfEtxACBps2pWL~MOnny5HnS6C8fqg~5sD0zNQK0mMo1rO6zeo5SrIa~dvu39GVVSZgnLfFPXLvgFE0zBA05S4g1JBUjROBIiWeeFh7CENTHRQewJlDLOS0ALkrHfwDmTz0mOiyoLxjEbi99Sqd8s3PHe1mdR68~5Wl~KviE0gE2fUeaGhHWRaR-hAp95SBKZoCwS67VStXK5G8iCDa4pEptl0346iZwTGq5oSus9y-xZ~S1YNj~8yLnqe3-v3w8mg~ghSZpaha7kOaGH92pcKGDzoLgDq8Lw8uoVWcE2~GbMH45miU9rC8JSqrG6P9py33sg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal