Interleukin-17 (IL-17) is a CD4 T-cell–derived proinflammatory cytokine. We investigated the effects of locally produced IL-17 by tumors as a means to evaluate its biologic function. Although recombinant IL-17 protein or retroviral transduction ofIL-17 gene into tumors did not affect in vitro proliferation, IL-17 transfectants grew more rapidly in vivo when compared with controls. Immunostaining for Factor VIII revealed that tumors transduced with IL-17 had significantly higher vascular density when compared with controls. IL-17 indeed elicited neovascularization in rat cornea. In addition, angiogenic activity present in the conditioned media of CD4 T cells was markedly suppressed by neutralizing monoclonal antibody to IL-17. IL-17 had no direct effect on the growth of vascular endothelial cells, whereas IL-17 significantly stimulated migration. IL-17 also markedly promoted the cord formation of vascular endothelial cells. In addition, IL-17 up-regulated elaboration of a variety of proangiogenic factors by fibroblasts as well as tumor cells. These findings reveal a novel role for IL-17 as a CD4 T-cell–derived mediator of angiogenesis that stimulates vascular endothelial cell migration and cord formation and regulates production of a variety of proangiogenic factors. Furthermore, they suggest that inhibition of biologic action of IL-17 may have therapeutic benefits when applied to angiogenesis-related disorders.
Introduction
Angiogenesis, the formation of new capillaries from pre-existing blood vessels, is a fundamental process involved in physiologic and pathologic conditions, including wound healing, rheumatoid arthritis, psoriasis, and tumorigenesis.1 The physiologic regulation of angiogenesis is dependent on the proper temporal and spatial delicate balance of activators and inhibitors present within the vascular microenvironment.2 Tumor growth is an angiogenesis-dependent process that requires the sustained new vessel growth.3 Tumors or interacting stromal cells may cause imbalance to increase elaboration of angiogenic inducers or decrease production or effects of angiogenic suppressors. Identification of endogenous angiogenesis stimulators or inhibitors is an area of great interest.3
Interleukin-17 (IL-17) was originally cloned by subtractive hybridization from an activated T-cell library and initially termed cytotoxic T-lymphocyte–associated antigen (CTLA)–8.4Recently, several homologous proteins with similar or different biologic profiles have been identified.5 Human IL-17 is produced predominantly by activated CD4 T cells and has sequence homology with the open reading frame 13 of the T-lymphotropic herpesvirus saimiri.6 IL-17 has several biologic activities, including induction of IL-6, IL-8, and prostaglandin E2 (PGE2), as well as enhancement of intercellular adhesion molecule (ICAM)–1 expression in fibroblasts and keratinocytes.6-9 In addition, IL-17 induces secretion of tumor necrosis factor (TNF)–α, IL-1β, and stromelysin by macrophages.10 IL-17 receptor (IL-17R) is a type 1 transmembrane protein with an extraordinarily long intracellular domain. Signaling by IL-17R results in the activation of transcription factor nuclear factor κB, and regulates the activities of extracellular-regulated kinase 1 (ERK1), ERK2, c-Jun N-terminal kinase, and p38 mitogen–activated protein kinases.6,11,12Although the proinflammatory function and intracellular signaling pathway of IL-17 is strikingly similar to those of IL-1 and Toll receptors, IL-17R has no homology with other known receptor sequences, thus making IL-17, homologous proteins, and its viral homologue a novel cytokine family. Even though the expression of IL-17R mRNA was detected in virtually all cells and tissues tested by Northern blot analysis, whether all cells expressing IL-17R respond to IL-17 is as yet not known.6 The ubiquitous expression of the IL-17Rgene strongly suggests the possibility of other as yet unknown biologic functions for IL-17.
There are multiple means to evaluate biologic functions of cytokines and chemokines. One way to define the biologic actions of a cytokine is to express it under a strong constitutive or inducible promoter in cell lines such as tumors or in mice and observe its local and systemic effects. We constructed a retroviral vector–expressing mouse IL-17 and the NeoR selectable marker (DFG-mIL-17-IRES-Neo) and transduced tumor cells to evaluate biologic effects in situ. IL-17 secreted by transfectants markedly enhanced in vivo tumor growth and significantly increased tumor vascularity. These results suggest that IL-17 produced by tumors may promote in vivo growth via potentiation of tumor angiogenesis. Actually, IL-17 elicited neovascularization in rat cornea assay. Our findings indicate that CD4 T-cell–derived cytokine IL-17 is a novel member of the angiogenic factor family and has a prominent role in angiogenesis-related situations.
Materials and methods
Mice and reagents
Female C57BL/6 mice were purchased from Taconic Farms (Germantown, NY). Mouse IL-17 cDNA was kindly supplied by Schering-Plough (Kenilworth, NJ). Mouse IL-17 and antimouse IL-17 monoclonal antibodies (mAbs) were provided by Mr John Abrams (DNAX Research Institute, Palo Alto, CA) and also purchased from Genzyme (Cambridge, MA). Human IL-17, human basic fibroblast growth factor (bFGF), and antihuman IL-17 mAb were purchased from R&D Systems (Minneapolis, MN). Suramin and human vascular endothelial growth factor (VEGF) were purchased from KURABO (Osaka, Japan).
Cells and cell cultures
MCA205 is a weakly immunogenic murine fibrosarcoma. MC38 is a poorly immunogenic murine colon adenocarcinoma. These cells were maintained in RPMI 1640 with 10% fetal calf serum (FCS), 2 mMl-glutamine, 1 mM sodium pyruvate, 5 × 10−5M 2-mercaptoethanol, 0.1 mM nonessential amino acids (NEAA), 100 IU/mL penicillin, and 100 μg/mL streptomycin (all from Life Technologies, Grand Island, NY), which are designated as complete medium (CM). Human umbilical vein endothelial cells (HUVECs) and human microvascular endothelial cells (HMVECs) were purchased from Clonetics (San Diego, CA) and maintained in Endothelial Cell Basal Medium (EBM) with 12 μg/mL bovine brain extract, 1 μg/mL hydrocortisone, 10 ng/mL epidermal growth factor, 50 μg/mL gentamycin, 50 ng/mL amphotericin-B, and 2% or 5% FCS (all from Clonetics). Primary culture of fibroblasts was established from the lung of 7-week-old C57BL/6 mice. Peripheral blood mononuclear cells were isolated using Lymphoprep (Nycomed Pharma As, Oslo, Norway) from blood from healthy volunteers. CD4 T cells were purified using CD4 T-Cell Enrichment Column (R&D Systems) according to the manufacturer's instructions. CD4 T cells were cultured for 84 hours in RPMI 1640 with 5% FCS, 2 mMl-glutamine, 1 mM sodium pyruvate, 0.1 mM NEAA, 100 IU/mL penicillin, and 100 μg/mL streptomycin in the presence of a mixture of phorbol myristate acetate (PMA) (1 nM) and ionomycin (3 μg/mL) (both from Sigma, St Louis, MO). Conditioned media of CD4 T cells, which contained approximately 7.1 ng/mL IL-17, were concentrated 40 times using Centricon (Millipore, Bedford, MA).
Construction of recombinant retroviral vector carrying mouse IL-17
A retroviral vector DFG-mIL-17-IRES-Neo was constructed as follows. MFG-mIL-17 was constructed using fragments ofNco1-BsmA1 adaptor,BsmA1-EcoR1 fragment of mouse IL-17 cDNA,EcoR1-BamH1 fragment of the polymerase chain reaction products of IL-17 cDNA, and a retroviral vector termed MFG.13 IRES-Neo cassette, consisting of an EMCV IRES sequence (Novagen, Madison, WI) and cDNA of Neo obtained from pMC1neo poly(A) (Stratagene, La Jolla, CA), was subcloned into theBamH1 site of MFG-mIL-17. Transfecting DFG-mIL-17-IRES-Neo proviral construct into ecotropic packaging cell line BOSC2314 (provided by Drs Pear and Baltimore, Rockefeller University, New York, NY) generated retroviral supernatant. The titer of the retroviral supernatant used in the subsequent experiments was 1.0 to 1.2 × 106 CFU/mL. Target cells were infected with 2 mL DFG-mIL-17-IRES-Neo retroviral supernatants in the presence of polybrene (8 μg/mL) and selected in CM with 800 μg/mL G418. As a control, a retroviral vector carrying only neomycin phosphotransferase gene (G1Na, provided by GTI, Germantown, MD) was used.
Flow cytometric analysis
Surface expression of MHC class I (H-2Kb), CD56, and CD44 antigens on tumor cells was examined by a fluorescence-activated cell sorter scan (Becton Dickinson, San Jose, CA) by using the monoclonal antibodies conjugated with fluorescein isothiocyanate (FITC) or phycoerythrin (PE) purchased from PharMingen (San Diego, CA).
In vitro cell growth assay
To evaluate the effect of IL-17 on the proliferation of vascular endothelial cells, HUVECs or HMVECs were suspended in EBM with 50 μg/mL gentamycin, 50 ng/mL amphotericin-B, and 2% FCS and were plated at 1.5 × 105 cells in 10-cm culture dish coated with collagen (Becton Dickinson, Bedford, MA). After 24 hours (on day 0), medium was then replaced with EBM containing 50 μg/mL gentamycin, 50 ng/mL amphotericin-B, and 2% FCS with or without cytokines. The cell number was counted on day 5.
3-(4,5-dimethylthiazol-2-yl)-2,5-dimethyltetrazolium bromide (MTT) assay
Cells (1 × 103) were seeded into 96-well flat-bottomed plates and cultured in RPMI 1640 containing 3.5% FCS with or without 0.5 to 500 ng/mL IL-17. On day 5, cells were washed with medium, and 100 μL MTT (Sigma) solution (2.5 mg/mL in RPMI 1640 with 10% FCS) was added to each well. Plates were incubated for 90 minutes. Next, MTT solution was removed and 50 μL dimethylsulfoxide (DMSO) (Sigma) was added to each well to solubilize formazan crystals formed in viable cells. The absorbance was read at a wavelength of 590 nm on enzyme-linked immunosorbent assay (ELISA) plate reader.
In vivo evaluation of tumor growth
Mice were inoculated subcutaneously in the right flank with 1 × 105 MCA205WT, MCA205Neo, or MCA205IL-17 or with 2 × 105 MC38WT, MC38Neo, or MC38IL-17. In some experiments, mice received 500 rad of whole-body irradiation. After 24 hours, tumor cells were inoculated. Tumor volume (cubic millimeter) was calculated by using the formula ab2/2 (a = largest diameter; b = smallest diameter).15
Histologic examination and quantitation of vessel density
Tumor tissues were harvested, fixed in 10% buffered formalin, processed, embedded in paraffin, and sectioned with a microtome. Sections were stained with hematoxylin and eosin or stained for Factor VIII.
For Factor VIII staining, sections were deparaffinized in xylene, dehydrated in graded concentrations of ethanol, and then rinsed in distilled water. Sections were digested with protease XXIV (Sigma), and then washed with distilled water. Endogenous peroxidase activity was blocked by 3% aqueous hydrogen peroxide, and nonspecific proteins were blocked by using protein blocker (Shanndon Lipshaw, Pittsburgh, PA). Then, rabbit antihuman factor VIII F(ab′)2 (DAKO, Carpinteria, CA) was applied. After washing, the sections were incubated with biotinylated goat antirabbit F(ab′)2antibody (Jackson ImmunoResearch, West Grove, PA). After washing, avidin biotin complex solution (Vector, Burlingame, CA) was applied and then developed with 3-amino-9-ethylcarbazole (ScyTek, Logan, UT). Finally, the sections were weakly counterstained with hematoxylin.
Tumor sections were examined in a blinded fashion for the presence of Factor VIII immunolocalization. Factor VIII staining was evaluated by quantifying the number of stained blood vessels in 5 randomly chosen high-power fields (HPFs) (× 200) per tumor. A total of 25 HPFs were examined and counted from 5 tumors of each of the groups. Any distinct area of positive staining for Factor VIII was counted as a single, regardless of size. The results were shown as mean ± SD per square millimeter considering the area of HPF.
Rat cornea assay
The cornea assay was performed as described previously.16 Briefly, hydron pellets (Interferon Sciences, New Brunswick, NJ) containing phosphate-buffered saline (PBS) alone, 250 or 500 ng mouse IL-17, or 100 ng human bFGF were implanted into the cornea of anesthetized rats. In some experiments, hydron pellets containing 250 ng mouse IL-17 and 1 μg control antibody or antimouse IL-17 mAb were implanted into the cornea. After 7 days, the animals were killed, and cornea vessels were photographed. Eight rats were used to assess the effect on neovascularization for each group. The experiment was repeated twice.
Matrigel plug assay
Mice were injected subcutaneously at the abdominal midline with 0.5 mL Growth Factor-Reduced Matrigel Matrix (Becton Dickinson) in the presence of 8 U/mL heparin (Sigma) supplemented with conditioned media of CD4 T cells and 20 μg/mL control Ab or neutralizing antihuman IL-17 mAb. After 6 days, Matrigel plugs were removed, fixed with 4% buffered paraformaldehyde, and embedded in paraffin. Sections were stained with hematoxylin and eosin. Vessel area and the total Matrigel area were planimetrically assessed from 3 stained sections per Matrigel plug with the use of National Institutes of Health imaging. The experiment was repeated twice with 5 mice per group in each experiment.
Migration assay
Migration of HMVECs or HUVECs was evaluated by using a modified Boyden chamber assay, as described previously.17 Cells were cultured in EBM with 2% FCS for 8 hours and plated at 12 × 104 cells/cm2 onto the polycarbonate filter with 5-μm pores (KURABO) coated with 10 μg/mL fibronectin (Sigma). Cytokines in EBM with 1% FCS were applied in the lower compartments of the chamber, and cells suspended in EBM with 1% FCS were cultured for 4 hours at 37°C. The filters were fixed and stained with Diff-Quick (Harleco, Gibbstown, NJ), and the number of migrating cells was quantified by counting cells in 5 randomly selected microscopic fields (× 200) in each well. For inhibitory assay, neutralizing antihuman IL-17 mAb (20 μg/mL) was added in the lower and upper compartments of the chamber.
Endothelial cord formation
In vitro cord formation was performed according to the manufacturer's instruction (KURABO).18 Briefly, HUVECs and human dermal fibroblasts were admixed and seeded into 24-well plates in medium with or without cytokine. Cells were incubated for up to 11 days, and the medium was replaced with fresh every 3 days. HUVECs were stained for Factor VIII. The analyses were made by computerized image analysis of the number of pixels occupied by the tubules in a total of 15 random areas from these separate wells.
Cytokine and nitric oxide (NO) assays
Fibroblasts or tumor cells (1 × 105/mL) were cultured in RPMI 1640 containing 3.5% FCS with or without IL-17 for 48 hours. Cell-free supernatants were collected and stored at −70°C. Concentrations of angiogenic factors were measured by ELISA kits (R&D Systems). To assess the production of NO, the concentration of nitrite was measured with the use of the Griess reaction, as described previously.19
Reverse transcription–polymerase chain reaction (RT-PCR)
The primer sequences of the oligonucleotides used for PCR were as follows: β-actin, sense: 5′-TTCTACAATGAGCTGCGTGTG-3′, antisense: 5′-CACTGTGTTGGCATAGAGGTC-3′; IL-17, sense: 5′-ATGAGTCCAGGGAGAGCTTCAT-3′, antisense: 5′-TTAGGCTGC CTGGCGGACAATC-3′; acidic fibroblast growth factor (aFGF), sense: 5′-GCAGCCC TGACCGAGAGGTTC-3′, antisense: 5′-CTTACAGCTCCCGTTCTTCTT-3′; bFGF, sense: 5′-ACACGTCAAACTACAACTCCA-3′, antisense: 5′-TCAGCTCTTAGCAGAC ATTGG-3′; transforming growth factor (TGF)–β, sense: 5′-CTAATGGTGGACCGCAA CAAC-3′, antisense: 5′-CGGTTCATGTCATGGATGGTG-3′. Total RNA was extracted by using RNAzolTMB (TEL-TEST, Friendswood, TX) according to manufacturer's instruction. Total RNA (4 μg) was applied for the synthesis of cDNA with SuperScriptTM RNaseH-Reverse Transcriptase (Life Technologies). PCR was performed in a DNA Thermal Cycler (Perkin-Elmer Cetus, Norwalk, CT) using Taq polymerase (Boehringer Mannheim, Indianapolis, IN).
Statistical analysis
Statistical analysis was performed using an unpaired 2-tailed Student t test with a confirmation by parametric andF tests. Difference was considered significant with aP value less than .05.
Results
Establishment and characterization of IL-17–producing tumor cell lines
We generated a retroviral vector DFG-mIL-17-IRES-Neo as described in “Materials and methods” and used it to transduce the fibrosarcoma MCA205 as well as the colon adenocarcinoma MC38. No expression of either IL-17 mRNA or protein could be detected in MCA205 and MC38 before transfection (data not shown). After G418 selection, expression of IL-17 mRNA by stable transfectants was determined by RT-PCR (data not shown). MCA205IL-17 or MC38IL-17 secretes 153 or 43 ng/1 × 106 cells per 48 hours, respectively, determined by ELISA. The biologic activity of expressed protein in the supernatants of MCA205IL-17 or MC38IL-17 was confirmed by its ability to induce IL-6 production (Table 1). Flow cytometric analysis demonstrated that the surface ICAM-1 expression of IL-17 transfectants was slightly enhanced when compared with parental cells (data not shown), which was consistent with a previous report.6 The expression of H-2Kb and CD44 was not altered (data not shown).
IL-6 production is markedly increased in IL-17–transfected tumor cell lines
| Cell lines . | IL-6 (pg/mL) . |
|---|---|
| MCA205WT | < 7.8 |
| MCA205Neo | < 7.8 |
| MCA205IL-17 | 238 |
| MCA205IL-17 + mAb | 11 |
| MC38WT | < 7.8 |
| MC38Neo | < 7.8 |
| MC38IL-17 | 146 |
| MC38IL-17 + mAb | < 7.8 |
| Cell lines . | IL-6 (pg/mL) . |
|---|---|
| MCA205WT | < 7.8 |
| MCA205Neo | < 7.8 |
| MCA205IL-17 | 238 |
| MCA205IL-17 + mAb | 11 |
| MC38WT | < 7.8 |
| MC38Neo | < 7.8 |
| MC38IL-17 | 146 |
| MC38IL-17 + mAb | < 7.8 |
Cells (1 × 105/mL) were cultured in CM with or without 20 μg/mL antimouse IL-17 monoclonal antibody. Cell-free supernatants were collected after 48 hours and assayed for the production of IL-6 by ELISA. Tumor cells transduced withIL-17 gene produced more IL-6 in response to IL-17 produced by themselves.
IL-17 significantly promotes in vivo tumor growth
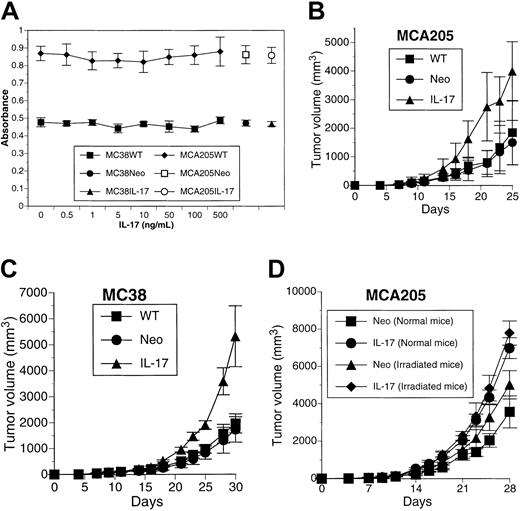
IL-17 has no direct effect on proliferation of either MCA205 or MC38 cells determined by MTT assay (Figure1A). Significant changes were not observed in in vitro growth of IL-17 transfectants when compared with that of parental cells or neomycin phosphotransferase gene transfectants (Figure 1A). When MCA205WT, MCA205Neo, or MCA205IL-17 cells were implanted subcutaneously into syngeneic mice, they all formed solid tumors. However, MCA205IL-17 developed tumors with an increased growth rate compared with MCA205WT or MCA205Neo (MCA205IL-17 versus MCA205WT, P < .008; MCA205IL-17 versus MCA205Neo,P < .0004, on day 25) (Figure 1B). A similar increase in in vivo tumor growth was observed when MC38IL-17 was implanted subcutaneously into the syngeneic animals compared with MC38WT or MC38Neo (MC38IL-17 versus MC38WT, P < .0002; MC38IL-17 versus MC38Neo, P < .0002, on day 30) (Figure1C).
IL-17 promotes in vivo tumor growth in a nonirradiation-dependent fashion.
(A) Recombinant IL-17 protein or transduction of the IL-17gene has no direct effect on in vitro growth of tumor cells determined by MTT assay. Each value represents mean ± SD (n = 5). (B-C) Mice were inoculated with MCA205WT, MCA205Neo, or MCA205IL-17 or with MC38WT, MC38Neo, or MC38IL-17. Each value represents mean tumor volume (cubic millimeter) ± SD for 6 or 7 mice per group. The result is a representative of 3 independent experiments. (D) Mice were irradiated with 500 rad whole-body irradiation before inoculation with MCA205Neo or MCA205IL-17. Each value represents mean tumor volume (cubic millimeter) ± SD for 5 mice per group. The result is a representative of 2 independent experiments.
IL-17 promotes in vivo tumor growth in a nonirradiation-dependent fashion.
(A) Recombinant IL-17 protein or transduction of the IL-17gene has no direct effect on in vitro growth of tumor cells determined by MTT assay. Each value represents mean ± SD (n = 5). (B-C) Mice were inoculated with MCA205WT, MCA205Neo, or MCA205IL-17 or with MC38WT, MC38Neo, or MC38IL-17. Each value represents mean tumor volume (cubic millimeter) ± SD for 6 or 7 mice per group. The result is a representative of 3 independent experiments. (D) Mice were irradiated with 500 rad whole-body irradiation before inoculation with MCA205Neo or MCA205IL-17. Each value represents mean tumor volume (cubic millimeter) ± SD for 5 mice per group. The result is a representative of 2 independent experiments.
We next examined whether immunologic mechanisms were involved in the increased in vivo growth of IL-17 transfectants. To address this issue, groups of mice were irradiated and challenged with MCA205Neo or MCA205IL-17 cells. Even in irradiated mice, MCA205IL-17 grew more rapidly when compared with control (MCA205IL-17 versus MCA205Neo,P < .002 [normal mice]; MCA205IL-17 versus MCA205Neo,P < .01 [irradiated mice] on day 28) (Figure 1D). Therefore, irradiation-sensitive cells, presumably immune cells including macrophages, do not primarily mediate the increased in vivo tumor growth induced by locally produced IL-17.
IL-17 markedly enhanced tumor microvascularity
To investigate the mechanisms by which the in vivo growth of tumors transduced with IL-17 was markedly enhanced, we first performed histologic examination of tumor tissues.
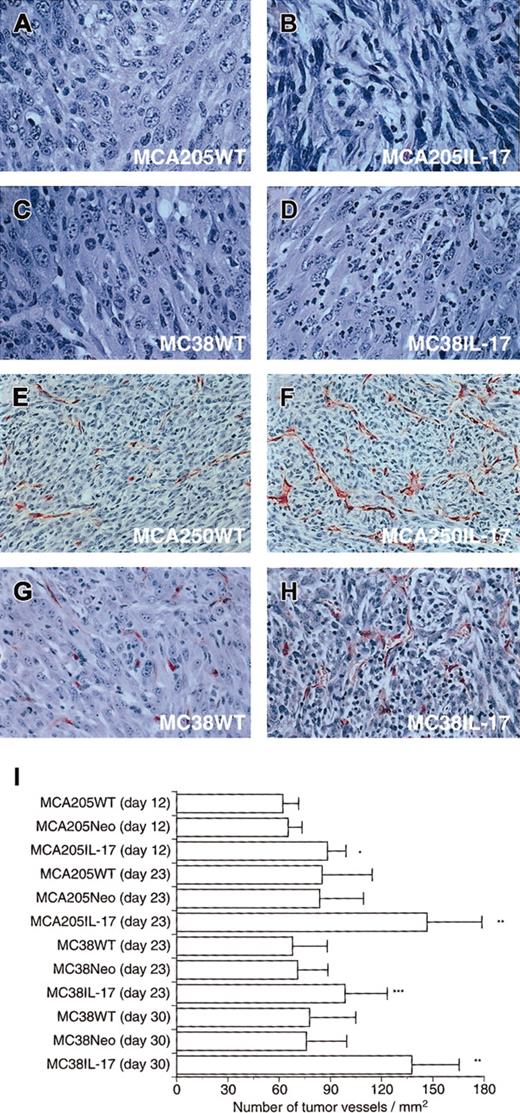
Hematoxylin-eosin–stained sections demonstrated that MCA205IL-17 tumors were more infiltrated with neutrophils and were edematous compared with controls (Figure 2A-B). Similar results were found in MC38IL-17 tumors (Figure 2C-D).
Immunostaining for Factor VIII showed that tumor tissues of IL-17 transfectants were more markedly vascularized than parental and Neo-transfected tumors (Figure 2E-H and data not shown). To compare the vascular density, we determined the mean number of blood vessels of Factor VIII–stained sections obtained from 5 independent IL-17 transfectants, Neo transfectants, or parental tumors (Figure 2I). The mean number of microvessels in sections of MCA205IL-17 tumors was significantly higher than that of controls on days 12 and 23 (P < .001). Similar results were obtained in MC38 tumors on days 23 and 30 (P < .01).
Histologic examination of subcutaneous tumor tissues.
Tumor tissues were harvested, fixed in 10% buffered formalin, processed, embedded in paraffin, and sectioned with a microtome. Sections were stained with hematoxylin and eosin or with Factor VIII. (A) Hematoxylin-eosin–stained section from MCA205WT (original magnification × 150). (B) Hematoxylin-eosin–stained section from MCA205IL-17 (original magnification × 150). (C) Hematoxylin-eosin–stained section from MC38WT (original magnification × 150). (D) Hematoxylin-eosin–stained section from MC38IL-17 (original magnification × 150). Note: IL-17 transfectants were markedly infiltrated with neutrophils and were edematous. (E) Factor VIII–stained section from MCA205WT (original magnification × 80). (F) Factor VIII–stained section from MCA205IL-17 (original magnification × 80). (G) Factor VIII–stained section from MC38WT (original magnification × 100). (H) Factor VIII–stained section from MC38IL-17 (original magnification × 100). Note: Immunostaining for the presence of Factor VIII showed that tumors transduced with IL-17 were more markedly vascularized when compared with parental tumors. (I) Factor VIII–stained sections were evaluated by quantifying the number of stained blood vessels in 5 randomly chosen HPFs (× 200) per tumor. Bars represent mean number of vessels ± SD per square millimeter considering the area of HPF (n = 25). The result is a representative of 2 independent experiments. *P < .001; **P < .0001; ***P < .01.
Histologic examination of subcutaneous tumor tissues.
Tumor tissues were harvested, fixed in 10% buffered formalin, processed, embedded in paraffin, and sectioned with a microtome. Sections were stained with hematoxylin and eosin or with Factor VIII. (A) Hematoxylin-eosin–stained section from MCA205WT (original magnification × 150). (B) Hematoxylin-eosin–stained section from MCA205IL-17 (original magnification × 150). (C) Hematoxylin-eosin–stained section from MC38WT (original magnification × 150). (D) Hematoxylin-eosin–stained section from MC38IL-17 (original magnification × 150). Note: IL-17 transfectants were markedly infiltrated with neutrophils and were edematous. (E) Factor VIII–stained section from MCA205WT (original magnification × 80). (F) Factor VIII–stained section from MCA205IL-17 (original magnification × 80). (G) Factor VIII–stained section from MC38WT (original magnification × 100). (H) Factor VIII–stained section from MC38IL-17 (original magnification × 100). Note: Immunostaining for the presence of Factor VIII showed that tumors transduced with IL-17 were more markedly vascularized when compared with parental tumors. (I) Factor VIII–stained sections were evaluated by quantifying the number of stained blood vessels in 5 randomly chosen HPFs (× 200) per tumor. Bars represent mean number of vessels ± SD per square millimeter considering the area of HPF (n = 25). The result is a representative of 2 independent experiments. *P < .001; **P < .0001; ***P < .01.
IL-17 induces neovascularization in rat cornea
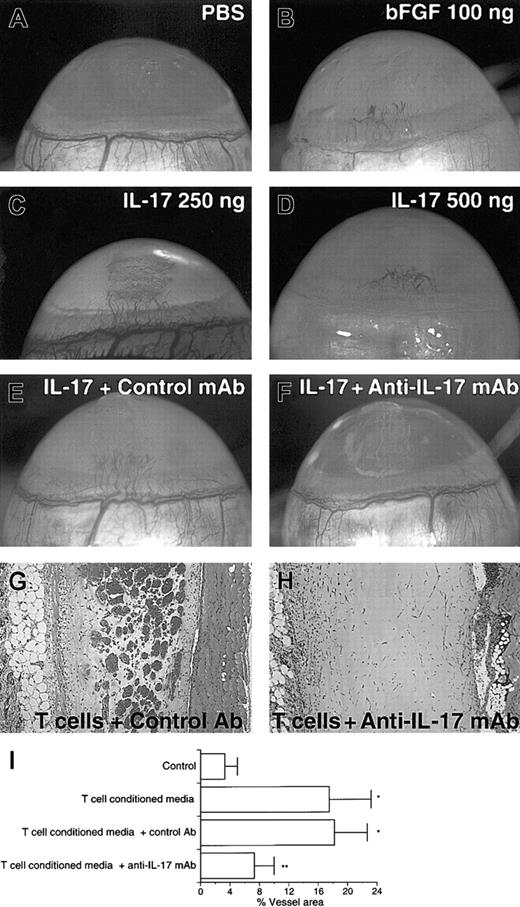
To confirm that IL-17 is angiogenic in vivo, we first examined the formation of new capillary blood vessels in rat cornea assay. Implants containing 100 ng human bFGF as positive control induced angiogenic response (8 positives of 8 corneas tested) (Figure3B). Although control implants containing PBS gave no angiogenic response (Figure 3A) (no positives of 8 corneas tested), implants containing 250 or 500 ng mouse IL-17 induced significant angiogenic response (8 positives of 8 corneas tested for each dose) (Figure 3C-D). No signs of accompanying inflammatory reaction were observed as shown by the persistence of cornea transparency for the duration of the experiment. The addition of neutralizing antimouse IL-17 mAb to the implants completely abolished the IL-17–induced vascularization but not control antibody (Figure 3E-F).
IL-17 elicits neovascularization in rat cornea assay, and the addition of anti–IL-17 mAb into Matrigel suppresses the neovessel formation induced by CD4 T cells in mouse Matrigel assay.
(A) No angiogenic response to PBS was observed. (B-D) Vigorous angiogenic response to mouse IL-17 or human bFGF was observed. (E) IL-17–induced vessel formation (250 ng) was not abolished by control mAb. (F) IL-17–induced vessel formation (250 ng) was completely abolished by neutralizing anti–IL-17 mAb. (G) Matrigel plug containing CD4 T-cell–conditioned media plus 20 μg/mL control Ab. (H) Matrigel plug containing CD4 T-cell–conditioned media plus 20 μg/mL anti–IL-17 mAb. (I) Conditioned media from CD4 T cells were shown to induce neovascularization (T cells versus control;P < .05). The addition of neutralizing anti–IL-17 mAb to Matrigel resulted in marked reduction in neovessel formation elicited by CD4 T-cell–conditioned media (T cells or T cells + control Ab versus T cells + anti–IL-17 mAb; *P < .05). Results are expressed as percentage ± SD of the vessel area to the total Matrigel area assessed from 3 stained sections per Matrigel plug. Each individual experimental group included 5 mice. The result is a representative of 2 independent experiments.
IL-17 elicits neovascularization in rat cornea assay, and the addition of anti–IL-17 mAb into Matrigel suppresses the neovessel formation induced by CD4 T cells in mouse Matrigel assay.
(A) No angiogenic response to PBS was observed. (B-D) Vigorous angiogenic response to mouse IL-17 or human bFGF was observed. (E) IL-17–induced vessel formation (250 ng) was not abolished by control mAb. (F) IL-17–induced vessel formation (250 ng) was completely abolished by neutralizing anti–IL-17 mAb. (G) Matrigel plug containing CD4 T-cell–conditioned media plus 20 μg/mL control Ab. (H) Matrigel plug containing CD4 T-cell–conditioned media plus 20 μg/mL anti–IL-17 mAb. (I) Conditioned media from CD4 T cells were shown to induce neovascularization (T cells versus control;P < .05). The addition of neutralizing anti–IL-17 mAb to Matrigel resulted in marked reduction in neovessel formation elicited by CD4 T-cell–conditioned media (T cells or T cells + control Ab versus T cells + anti–IL-17 mAb; *P < .05). Results are expressed as percentage ± SD of the vessel area to the total Matrigel area assessed from 3 stained sections per Matrigel plug. Each individual experimental group included 5 mice. The result is a representative of 2 independent experiments.
Moreover, we attempted to elucidate the angiogenic activity of IL-17 secreted by CD4 T cells. Concentrated conditioned media from CD4 T cells stimulated by PMA and ionomycin were shown to induce significant neovascularization (Figure 3G). The addition of neutralizing antihuman IL-17 mAb to Matrigel resulted in marked reduction in neovessel formation (Figure 3H). These results demonstrate that most of the angiogenic activity present in the conditioned media of CD4 T cells activated in vitro was neutralizable with anti-IL-17 mAb.
Effects of IL-17 on vascular endothelial cells
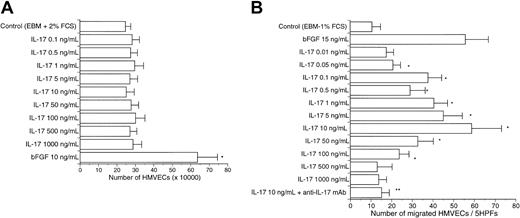
We examined the effects of IL-17 on in vitro angiogenesis-related functions of vascular endothelial cells. In contrast to bFGF, a wide range of doses of human IL-17 (0.1-500 ng/mL) had no direct effect on the proliferation of HMVECs and HUVECs (Figure4A and data not shown). However, as shown in Figure 4B, we observed a dose-dependent chemotactic response of HMVECs toward IL-17. In addition, IL-17 also induced chemotactic response of HUVECs (data not shown). The maximal chemotactic response for HMVECs occurred around 10 ng/mL IL-17. The specificity of this chemotactic response was determined by using neutralizing mAb (20 μg/mL) to human IL-17. IL-17–induced migration of HMVECs (10 ng/mL) was similar to that induced by 15 ng/mL bFGF.
Dose-dependent migration of HMVECs following stimulation with IL-17.
(A) HMVECs were allowed to grow over 5 days in the presence of increasing concentrations of IL-17. Bars represent mean cell number ± SD (n = 3). The result is a representative of 2 independent experiments (10 ng/mL bFGF versus 0-1000 ng/mL IL-17; *P < .05). (B) A wide range of doses of IL-17 stimulated the migration of HMVECs. BFGF (15 ng/mL) also stimulated the migration of HMVECs. Bars represent the mean number of migrated cells ± SD per 5 HPFs (× 200) (n = 5). The result is a representative of 3 independent experiments (control versus 0.05-100 ng/mL IL-17; *P < .05; 10 ng/mL IL-17 versus 10 ng/mL IL-17 + anti-IL-17 mAb; **P < .001).
Dose-dependent migration of HMVECs following stimulation with IL-17.
(A) HMVECs were allowed to grow over 5 days in the presence of increasing concentrations of IL-17. Bars represent mean cell number ± SD (n = 3). The result is a representative of 2 independent experiments (10 ng/mL bFGF versus 0-1000 ng/mL IL-17; *P < .05). (B) A wide range of doses of IL-17 stimulated the migration of HMVECs. BFGF (15 ng/mL) also stimulated the migration of HMVECs. Bars represent the mean number of migrated cells ± SD per 5 HPFs (× 200) (n = 5). The result is a representative of 3 independent experiments (control versus 0.05-100 ng/mL IL-17; *P < .05; 10 ng/mL IL-17 versus 10 ng/mL IL-17 + anti-IL-17 mAb; **P < .001).
IL-17 markedly promotes endothelial cord formation in vitro
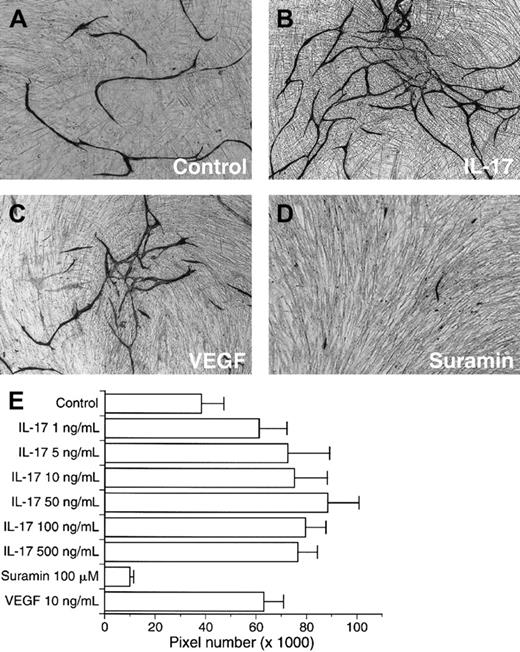
We further evaluated the effects of IL-17 on vascular endothelial cell cord formation. Control culture developed small amounts of vessel formation (Figure 5A). VEGF (10 ng/mL) as positive control increased vessel-like structure (Figure 5C). The addition of a wide range of doses of human IL-17 at the start of assay and with each medium change resulted in a marked enhanced development of structures resembling a microvasculature bed (Figure 5B). IL-17–increased microvessel was estimated at more than 60% of the control value (Figure 5E). Around 50 ng/mL IL-17 developed most abundant microvessel-like structures in this assay system.
IL-17 promotes endothelial cord formation.
(A) Factor VIII–stained microvessels developed by medium alone. (B) Factor VIII–stained microvessels developed by 50 ng/mL IL-17. (C) Factor VIII–stained microvessels developed by 10 ng/mL VEGF. (D) Factor VIII–stained microvessels developed by 100 μM suramin. (E) IL-17 (1-500 ng/mL) markedly enhanced the development of microvessel-like structures. VEGF (10 ng/mL) also promoted the microvessel formation. Suramin significantly inhibited the development of microvasculature. Bars represent the mean number of pixels ± SD (n = 6). The result is a representative of 2 independent experiments.
IL-17 promotes endothelial cord formation.
(A) Factor VIII–stained microvessels developed by medium alone. (B) Factor VIII–stained microvessels developed by 50 ng/mL IL-17. (C) Factor VIII–stained microvessels developed by 10 ng/mL VEGF. (D) Factor VIII–stained microvessels developed by 100 μM suramin. (E) IL-17 (1-500 ng/mL) markedly enhanced the development of microvessel-like structures. VEGF (10 ng/mL) also promoted the microvessel formation. Suramin significantly inhibited the development of microvasculature. Bars represent the mean number of pixels ± SD (n = 6). The result is a representative of 2 independent experiments.
IL-17 markedly up-regulates production of a variety of proangiogenic factors by fibroblasts as well as tumor cells
To examine another potential mechanisms by which IL-17 mediates angiogenesis, we tested whether IL-17 might induce fibroblasts and tumor cells to produce angiogenic growth factors and angiogenic cytokines that had been reported to promote tumor angiogenesis. IL-17 significantly increased elaboration of multiple angiogenic factors, including PGE1,20PGE2,21 VEGF,22keratinocyte-derived chemokine (KC),23 macrophage inflammatory protein (MIP)–2,23 and/or NO24(Tables2-3). The addition of neutralizing antimouse IL-6 mAb or indomethacin to the culture did not abolish IL-17–induced VEGF production (data not shown), suggesting that this effect was direct. In addition, MCA205IL-17 cells produced more VEGF, KC, and PGE2 in response to IL-17 secreted by themselves than the parental cells and MCA205Neo cells (Table 4). Neutralization of IL-17 inhibited the increased elaboration of these proangiogenic factors (Table 4). In contrast, induction of aFGF,25bFGF,25 and TGF-β26 production was not observed by semiquantitative RT-PCR (data not shown). These results indicate that IL-17 promotes angiogenesis in part via induction of a variety of proangiogenic factors by fibroblasts and tumors.
IL-17 markedly augments production of proangiogenic factors of tumors
| Angiogenic factors . | MCA205 . | MCA205 + IL-17* . | MCA205 + IL-17 + mAb . | MC38 . | MC38 + IL-17 . | MC38 + IL-17 + mAb . |
|---|---|---|---|---|---|---|
| VEGF | 5.87 ± 0.25 | 9.88 ± 0.67 | 5.75 ± 0.37 | 2.26 ± 0.13 | 2.7 ± 0.24 | 2.25 ± 0.18 |
| KC | 1.6 ± 0.13 | 29.26 ± 1.76 | 1.57 ± 0.17 | 0.36 ± 0.07 | 6.1 ± 0.5* | 0.38 ± 0.1 |
| MIP-2 | 10.0 ± 0.4 | 65.0 ± 4.2 | 11.0 ± 0.7 | 11.0 ± 0.8 | 78.0 ± 3.3* | 12.1 ± 0.7 |
| PGE1 | 27.5 ± 2.04 | 67.0 ± 4.49 | 27.7 ± 1.77 | 2.2 ± 0.12 | 4.5 ± 0.3* | 2.3 ± 0.15 |
| PGE2 | 9.0 ± 1.22 | 23.1 ± 3.67 | 10.1 ± 1.7 | 3.0 ± 0.41 | 5.8 ± 0.49* | 2.78 ± 0.57 |
| NO | 0.05 ± 0.01 | 0.09 ± 0.01 | 0.06 ± 0.01 | 0.95 ± 0.04 | 2.98 ± 0.58* | 0.97 ± 0.05 |
| Angiogenic factors . | MCA205 . | MCA205 + IL-17* . | MCA205 + IL-17 + mAb . | MC38 . | MC38 + IL-17 . | MC38 + IL-17 + mAb . |
|---|---|---|---|---|---|---|
| VEGF | 5.87 ± 0.25 | 9.88 ± 0.67 | 5.75 ± 0.37 | 2.26 ± 0.13 | 2.7 ± 0.24 | 2.25 ± 0.18 |
| KC | 1.6 ± 0.13 | 29.26 ± 1.76 | 1.57 ± 0.17 | 0.36 ± 0.07 | 6.1 ± 0.5* | 0.38 ± 0.1 |
| MIP-2 | 10.0 ± 0.4 | 65.0 ± 4.2 | 11.0 ± 0.7 | 11.0 ± 0.8 | 78.0 ± 3.3* | 12.1 ± 0.7 |
| PGE1 | 27.5 ± 2.04 | 67.0 ± 4.49 | 27.7 ± 1.77 | 2.2 ± 0.12 | 4.5 ± 0.3* | 2.3 ± 0.15 |
| PGE2 | 9.0 ± 1.22 | 23.1 ± 3.67 | 10.1 ± 1.7 | 3.0 ± 0.41 | 5.8 ± 0.49* | 2.78 ± 0.57 |
| NO | 0.05 ± 0.01 | 0.09 ± 0.01 | 0.06 ± 0.01 | 0.95 ± 0.04 | 2.98 ± 0.58* | 0.97 ± 0.05 |
Cells (1 × 105/mL) were cultured for 48 hours with or without 50 ng/mL IL-17 or 50 ng/mL IL-17 plus 20 μg/mL neutralizing antimouse IL-17 mAb. Cell-free supernatants were collected and assayed for the concentrations of VEGF, KC, MIP-2, PGE1, PGE2, and NO (ng/mL for VEGF, KC, PGE1, and PGE2; pg/mL for MIP-2; μM for NO). Data were expressed as mean ± SD (n = 3 per data point).
P < .05.
IL-17 markedly augments production of proangiogenic factors of fibroblast
| Angiogenic factors . | Fibroblast . | Fibroblast + IL-173-150 . | Fibroblast + IL-17 + mAb . |
|---|---|---|---|
| VEGF | 2.82 ± 0.18 | 4.69 ± 0.33 | 2.77 ± 0.23 |
| KC | 1.3 ± 0.05 | 26.5 ± 2.15 | 1.41 ± 0.12 |
| MIP-2 | 36.0 ± 1.3 | 52.0 ± 2.2 | 37.7 ± 1.5 |
| PGE1 | 0.8 ± 0.35 | 4.8 ± 0.65 | 0.87 ± 0.33 |
| PGE2 | 39.5 ± 2.45 | 81.1 ± 7.35 | 38.7 ± 2.86 |
| NO | 8.1 ± 0.95 | 13.3 ± 1.0 | 8.21 ± 1.1 |
| Angiogenic factors . | Fibroblast . | Fibroblast + IL-173-150 . | Fibroblast + IL-17 + mAb . |
|---|---|---|---|
| VEGF | 2.82 ± 0.18 | 4.69 ± 0.33 | 2.77 ± 0.23 |
| KC | 1.3 ± 0.05 | 26.5 ± 2.15 | 1.41 ± 0.12 |
| MIP-2 | 36.0 ± 1.3 | 52.0 ± 2.2 | 37.7 ± 1.5 |
| PGE1 | 0.8 ± 0.35 | 4.8 ± 0.65 | 0.87 ± 0.33 |
| PGE2 | 39.5 ± 2.45 | 81.1 ± 7.35 | 38.7 ± 2.86 |
| NO | 8.1 ± 0.95 | 13.3 ± 1.0 | 8.21 ± 1.1 |
Cells (1 × 105/mL) were cultured for 48 hours with or without 50 ng/mL IL-17 or 50 ng/mL IL-17 plus 20 μg/mL neutralizing antimouse IL-17 mAb. Cell-free supernatants were collected and assayed for the concentrations of VEGF, KC, MIP-2, PGE1, PGE2, and NO (ng/mL for VEGF, KC, PGE1, and PGE2; pg/mL for MIP-2; μM for NO). Data were expressed as mean ± SD (n = 3 per data point).
P < .05.
Transduction with IL-17 into tumor cells markedly augments production of proangiogenic factors of tumor
| Angiogenic factors . | MCA205 . | MCA205Neo . | MCA205IL-174-150 . | MCA205IL-17 + mAb . |
|---|---|---|---|---|
| VEGF | 4.12 ± 0.31 | 4.17 ± 0.25 | 6.87 ± 0.57 | 4.25 ± 0.37 |
| KC | 1.25 ± 0.12 | 1.31 ± 0.17 | 25.27 ± 2.15 | 1.41 ± 0.17 |
| PGE2 | 15.5 ± 1.1 | 14.8 ± 1.3 | 47.5 ± 3.2 | 16.1 ± 1.5 |
| Angiogenic factors . | MCA205 . | MCA205Neo . | MCA205IL-174-150 . | MCA205IL-17 + mAb . |
|---|---|---|---|---|
| VEGF | 4.12 ± 0.31 | 4.17 ± 0.25 | 6.87 ± 0.57 | 4.25 ± 0.37 |
| KC | 1.25 ± 0.12 | 1.31 ± 0.17 | 25.27 ± 2.15 | 1.41 ± 0.17 |
| PGE2 | 15.5 ± 1.1 | 14.8 ± 1.3 | 47.5 ± 3.2 | 16.1 ± 1.5 |
Cells (1 × 105/mL) were cultured for 48 hours in CM with or without 20 μg/mL neutralizing antimouse IL-17 mAb. Cell-free supernatants were collected and assayed for the concentrations of VEGF, KC, and PGE2 (ng/mL) by ELISA. MCA205 cells transduced with IL-17 produced more VEGF, KC, and PGE2 in response to IL-17 produced by themselves. Data were expressed as mean ± SD (n = 3 per data point).
P < .05.
Discussion
Here we report for the first time that IL-17 is a novel CD4 T-cell–derived angiogenic factor that plays a potential role in T-cell–mediated neovascularization. Neither recombinant IL-17 protein nor IL-17 gene transduction influenced in vitro tumor growth. However, tumors transduced with IL-17 grew more rapidly than controls in immunocompetent and irradiated mice. Vascular elements of tumor tissues in IL-17 transfectants were significantly increased when compared with those of controls. Because neovascularization is a critical process for the sustained growth of solid tumors, our findings suggest that IL-17 may accelerate in vivo tumor growth via acting as an angiogenic factor.
On the basis of these observations, we hypothesized that IL-17 may be a CD4 T-cell–derived stimulator of angiogenesis. To assess the validity of this hypothesis and to elucidate its contribution to the observed enhanced vascularity in situ, we first evaluated the ability of IL-17 to mediate neovessel formation in vivo. Amounts as small as 250 ng IL-17 induced significant corneal angiogenic response with no evidence of nonspecific inflammation. The IL-17 dependency of in vivo angiogenic processes was confirmed by the inhibitory effect of anti–IL-17 mAb. Moreover, there have been reports demonstrating that CD4 T cells induce angiogenesis via elaboration of angiogenic lymphokines.27 28 Therefore, we determined whether IL-17 contributed to the angiogenic activity liberated by human T cells activated in vitro. Importantly, neutralization of IL-17 in the T-cell–conditioned media resulted in significant diminution of the angiogenic response in Matrigel plug assay. These studies indicate that IL-17 is a novel CD4 T-cell–derived mediator of angiogenesis and suggest a role for CD4 T cells as cellular effectors of angiogenesis mediated in part by IL-17.
To explore the mechanisms by which IL-17 mediates neovascularization, we next evaluated the influence of IL-17 on in vitro angiogenesis-related functions of vascular endothelial cells. IL-17 had no direct effect on the in vitro proliferation of vascular endothelial cells. However, a wide range of doses of IL-17 (0.05-100 ng/mL) significantly stimulated the migration. Moreover, in in vitro cord formation assay, IL-17 markedly promoted the development of microvessel-like structures. Therefore, IL-17 promotes angiogenesis via stimulation of vascular endothelial cell migration and cord formation. Growth factors and cytokines that stimulate angiogenesis in vivo but fail to demonstrate mitogenic activity for vascular endothelial cells in vitro have been called indirect angiogenic factors. Therefore, these characteristics of IL-17 appear to resemble those of the class of indirect angiogenic stimulators, including TGF-β and platelet-derived growth factor B subunit homodimer (PDGF BB), that mediate angiogenesis in vivo but do not stimulate proliferation of vascular endothelial cells in vitro.29
Fibroblasts are a source of proangiogenic factors, including VEGF, during development, hypoxia, tumor growth, and inflammation, and they markedly promote inflammatory and tumor angiogenesis.30,31 We found that IL-17 is able to markedly up-regulate VEGF, KC, MIP-2, prostaglandins, and NO production by fibroblasts. IL-17, therefore, stimulates production of proangiogenic factors in fibroblasts and promotes fibroblast-induced neovessel formation in inflammation and tumors. Although previous studies indicated the involvement of IL-17 in pathologic situations such as rheumatoid arthritis,32-34 the role of IL-17 in pathologic angiogenesis has not been evaluated yet. Our findings suggest that IL-17 functions as a CD4 T-cell–derived angiogenic mediator, now implicated in the pathogenesis of inflammatory angiogenesis. In addition, these findings suggest that some of the angiogenic activities of IL-17 may reflect the indirect effects on vascular endothelial cells mediated by an array of proangiogenic factors produced by fibroblasts.
Several investigations about the effects of IL-17 on in vivo tumor growth using other tumor model systems have been reported. Hirahara et al35 and Benchetrit et al36 demonstrated that IL-17 inhibits tumor growth in a T-cell–dependent manner. In contrast, Tartour et al37 reported that IL-17 promotes human cervical tumor growth via IL-6–dependent mechanism. Therefore, it appears that IL-17 may be a pleiotropic cytokine with possible tumor-promoting or tumor-suppressing effects. However, although human cervical tumors are generally poorly immunogenic, murine Meth-A fibrosarcoma, P815, and J558L leukemic cells are immunogenic. In addition, MCA205 and MC38 used in our experiments are weakly and poorly immunogenic tumors, respectively. Thus, the effects of IL-17 on in vivo tumor growth may depend largely on the immunogenicity and cell type of tumors.
Macrophages have been thought to play a key role in physiologic and pathologic angiogenesis.37 Secretory products such as TNF-α, IL-8, and VEGF released by activated macrophages have the capacity to influence each phase of the angiogenic process, including modifying the local extracellular matrix, induction of endothelial cells to migrate or proliferate, and inhibition of vascular growth with formation of differentiated capillaries.37 In contrast, even though T cells are usually found at the same sites as macrophages in angiogenic processes, including tumors, few reports have analyzed the role of T cells in mediating angiogenesis. We show here that IL-17 secreted by CD4 T cells promotes neovascularization. These findings provide insight into the role of infiltrating CD4 T cells to regulate angiogenesis via elaboration of IL-17.
Although the immune system has been suggested to be an important negative regulator of tumor growth, several experimental models suggest that immune responses could on occasion promote tumor growth.38 In particular, there have been reports demonstrating the deleterious effects of CD4 T cells on tumor immunity in several treatment models.39-41 We show here that the CD4 T-cell–derived cytokine IL-17 has significant tumor-promoting effects via potentiation of tumor angiogenesis. CD4 T cells infiltrating into tumor tissues may stimulate tumor cells and stromal fibroblasts, in conjunction with tumor-associated macrophages, to elaborate a variety of proangiogenic factors, including VEGF. VEGF also influences the immune system via inhibition of the development of dendritic cells42 and subsequently promotes tumor angiogenesis and growth. This could explain in part the reported paradoxical role of CD4 T cells in tumor immunity.
In conclusion, our findings illustrate a novel biologic function of CD4 T-cell–derived cytokine IL-17. IL-17 promotes angiogenesis via not only stimulation of vascular endothelial cell migration but also induced elaboration of a variety of proangiogenic factors that lead to the imbalance between angiogenesis activators and inhibitors present within the vascular microenvironment. Investigation of the physiologic regulation of IL-17 production by CD4 T cells may thus be useful for the treatment in clinical settings characterized by persistent neovascularization. Its production by herpesvirus saimiri also suggests that vascular events, including angiogenesis, are important for virus pathogenesis and persistence.
We thank Dr Tatsuro Ishibashi and Dr Michihiko Kuwano for general assistance. We are also grateful to Susan F. Schoonover, Loraine R. McKenzie, and Jennifer Lonellie for their excellent technical assistance.
Prepublished online as Blood First Edition Paper, October 31, 2002; DOI 10.1182/blood-2002-05-1461.
Supported in part by National Institutes of Health grant POICA59371 (H.T. and M.T.L.).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Muneo Numasaki, Department of Geriatric and Respiratory Medicine, Tohoku University School of Medicine, 1-1 Seiryo-machi, Aoba-ku, Sendai 980-8574, Japan; e-mail:numasaki77@aol.com.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal