To the Editor:
Characterization of β-thalassemia mutations in different populations has shown 180 mutant alleles.1 So far, 25 mutations have been reported among Indians, 5 of which comprise more than 80% of the mutant alleles.2-4
We report two interesting Indian families showing a novel β+-thalassemia mutation and a rare transcriptional mutation. They had come to us for second trimester prenatal diagnosis by globin biosynthesis.
The β-thalassemia mutations were characterized by denaturing gradient gel electrophoresis (DGGE) analysis.5 Both mutations were detected in fragment B of the β-globin gene spanning from the upstream −64 nucleotide to IVS-I-nt61 containing the promoter boxes and exon-1. DNA Sequencing was performed by the dideoxy method using Sequenase version 2.0 to identify the mutation.6
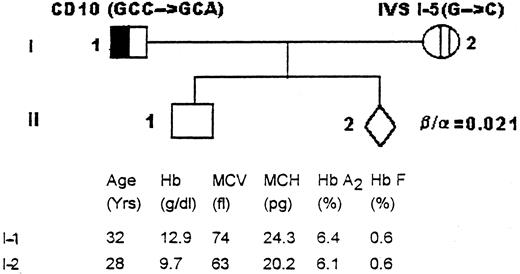
Family I was from Madhya Pradesh in central India. Both parents (I-1) and (I-2) had classical β-thalassemia trait (Fig 1). Their 3-year-old son (II-1) with severe homozygous β-thalassemia had been diagnosed at 8 months of age and had been transfused every month. This child was not available for investigation. Fetal diagnosis at 18 weeks of gestation in the next pregnancy showed that the β/α biosynthetic ratio was 0.021, indicating that the fetus had homozygous β+-thalassemia (normal β/α ratio, >0.03).
Family I. The hematologic profile and the β-thalassemia mutations characterized in the parents (I-1 and I-2).
Family I. The hematologic profile and the β-thalassemia mutations characterized in the parents (I-1 and I-2).
DGGE analysis showed that the mother (I-2) had the IVS I-5 G → C mutation, whereas the father (I-1) had an anomalous DGGE pattern in fragment B.
Sequencing of this region using the forward primer showed a novel mutation at codon 10 GCC → GCA on the coding strand. Both the normal codon (GCC) and the mutant codon (GCA) code for alanine. This C → A substitution creates the sequence CAGTTA in the mutated region. A catalogue of the sequences found at functional splice sites has identified the 5′ consensus sequence C/A AGG TG/AA. The C → A change in codon 10 produces a sequence that has homology to 5 of 6 nucleotides of the normal splice site at the exon 1-intron-I boundary. It has been reported earlier that this homologous sequence in the exon causes alternative splicing at the site giving a β+-phenotype.2 7
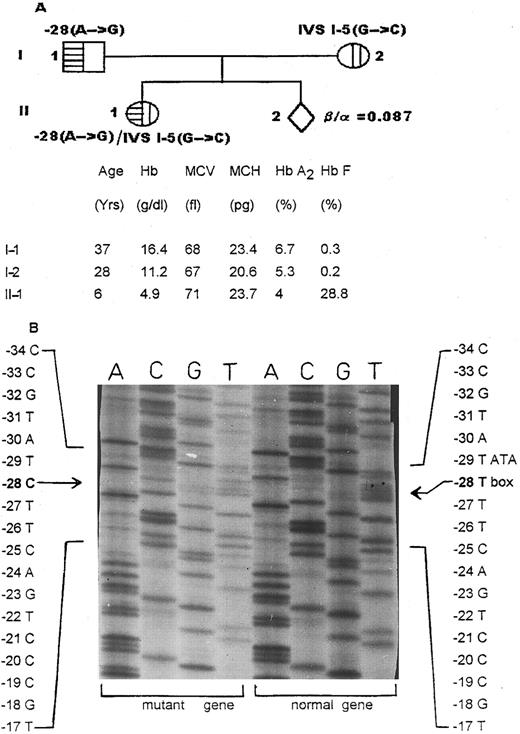
Family II was from Karnataka in south India. Both parents (I-1 and I-2) had classical β-thalassemia trait (Fig 2A). Their 6-year-old daughter had a homozygous β-thalassemic picture but had never been transfused. Globin biosynthesis in the next pregnancy showed that the 18-week-old fetus was normal.
Family II. (A) The hematologic profile and the β-thalassemia mutations characterized in the parents (I-1 and I-2) and their 6-year-old daughter (II-1). (B) Part of a sequencing gel showing the rare T → C mutation on the noncoding strand at position (−28) of the upstream ATA box in the father (I-1).
Family II. (A) The hematologic profile and the β-thalassemia mutations characterized in the parents (I-1 and I-2) and their 6-year-old daughter (II-1). (B) Part of a sequencing gel showing the rare T → C mutation on the noncoding strand at position (−28) of the upstream ATA box in the father (I-1).
DGGE analysis showed the IVS I-5 G → C mutation in the mother (I-2), whereas the father (I-1) had another anomalous DGGE pattern in fragment B. The DGGE pattern in the homozygous child (II-1) was different from that seen in the parents.
Sequencing of this β-globin gene region using the forward primer did not show any mutation. Sequencing with the reverse primer showed a T → C change on the noncoding strand (Fig 2B). This mutation was found in the daughter (II-1) as well, who also showed the presence of the IVS-I-5 G → C mutation. This A → G change in the upstream ATA box at position (−28) is a transcriptional mutant reported among Chinese.8 Nevertheless, it has not been reported among Indians.
These two new rare mutations could be added to the 25 different β-thalassemic mutations that have been reported among Indians so far.
ACKNOWLEDGEMENT
We thank S.R. Shirsat for preparation of the manuscript.