In this issue of Blood, Lum et al1 report excellent outcomes for pediatric and adolescent patients with Fanconi anemia (FA) who underwent allogeneic hematopoietic stem cell transplant (HSCT) in the recent era. This retrospective study of 813 transplants from 2010 to 2018 validates prior decades of pioneering work dedicated to establishing safe and effective HSCT for patients with FA.
FA is the most common inherited bone marrow failure syndrome, estimated to affect approximately 1 in 360 000 live births.2 Patients with FA can present with a variety of clinical findings, which most often include congenital anomalies, bone marrow failure, and a predisposition to hematologic malignancies and solid tumors. The underlying pathophysiology of FA involves the inability of cells to repair DNA damage caused by interstrand cross-links, leaving patients with FA exquisitely sensitive to chemotherapy and radiation, which are DNA-damaging agents and mainstays of HSCT conditioning.3 Mutations in any FA pathway gene, of which at least 23 have been identified to date, can cause FA.
The most common manifestation of FA is bone marrow failure, which occurs in the vast majority (∼90%) of patients by age 40.4 The other major hematologic abnormalities in patients with FA are myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML), the latter of which carries a relative risk ∼700-fold higher compared with the general population.5 Currently, the only curative treatment for the hematologic complications of FA is allogeneic HSCT.
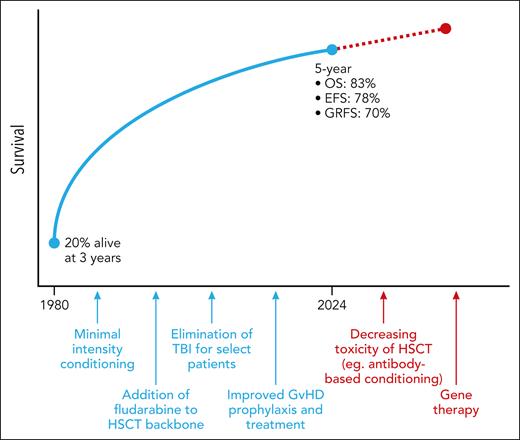
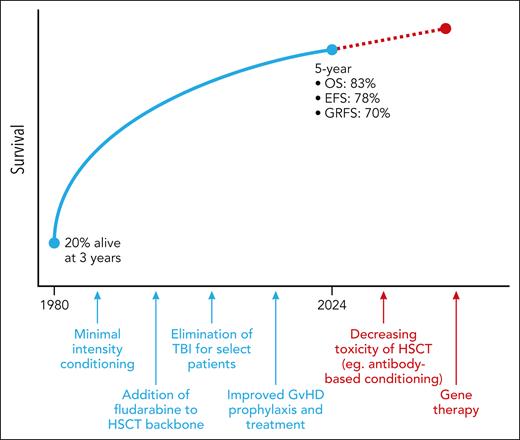
Historically, HSCT for FA has been associated with extremely high rates of toxicity, largely due to the sensitivity of FA cells to chemotherapy and radiation (both of which are commonly used in HSCT conditioning regimens). In the first published case series of HSCT for FA, Gluckman et al reported a survival rate of only 20% (1 of 5 patients) at 3 years post-HSCT.6 The major cause of death in this cohort was acute graft-versus-host disease (GVHD). Further, all patients demonstrated signs of cyclophosphamide toxicity despite receiving doses that are well tolerated in other conditions, providing early evidence of the unique sensitivity to DNA damage that is inherent to FA.
Since this first publication, HSCT outcomes for patients with FA have significantly improved (see figure). This has been driven by the adoption of major changes in the FA HSCT paradigm, including (1) the use of minimal-intensity conditioning to decrease transplant-associated toxicities; (2) inclusion of fludarabine in the HSCT backbone, which reduced rates of graft failure; (3) elimination of irradiation for select patients; and (4) improvement in both GVHD prophylaxis and treatment, which is particularly important for patients with FA as GVHD is associated with the development of late, and often fatal, secondary malignancies.7,8 As these changes have been implemented, survival rates have risen accordingly.
HSCT outcomes for patients with FA have improved dramatically over time. TBI, total body irradiation; OS, overall survival; EFS, event-free survival; GRFS, GVHD-free relapse-free survival. Professional illustration by Patrick Lane, ScEYEnce Studios.
HSCT outcomes for patients with FA have improved dramatically over time. TBI, total body irradiation; OS, overall survival; EFS, event-free survival; GRFS, GVHD-free relapse-free survival. Professional illustration by Patrick Lane, ScEYEnce Studios.
In their article, Lum et al provide the most recent data highlighting the improvement in HSCT outcomes for patients with FA. In this largest retrospective multicenter study of pediatric and adolescent (age ≤18 years) patients with FA who underwent HSCT in the most recent decade (2010-2018), 5-year outcomes are excellent and consistent with those reported in smaller cohorts including an overall survival of 83%, event-free survival of 78%, and GVHD-free relapse-free survival of 70%. Predictors of survival were comparable with those reported previously, including age at HSCT, donor type, HSCT indication, conditioning regimen, and serotherapy. The rate of secondary malignancy was low in this study (cumulative incidence of 2% at 5 years), though it is important to note that the median duration of follow-up for the entire cohort is only 3.7 years, whereas the onset of secondary malignancies typically occurs much later post-HSCT; longer follow-up of this cohort is needed to assess the later development of secondary malignancies over an extended period of time.
This study also points to opportunities for further improvement. Most notably, patients with FA who develop MDS and AML continue to suffer significantly poorer outcomes as compared with patients with FA with bone marrow failure or aplastic anemia. Both large multicenter prospective studies and smaller innovative pilot studies testing HSCT approaches and the use of pre-HSCT chemotherapy are needed. Similarly, although outcomes for patients with FA receiving HSCT from an alternative donor have improved, patients lacking an HLA-matched family or HLA-matched unrelated donor continue to experience inferior outcomes. Haploidentical donor transplantation widens the donor pool for these patients, though further work optimizing both the conditioning backbone and method of GVHD prophylaxis (ex vivo T-cell depletion vs posttransplant cyclophosphamide) is required. Finally, given the high risk of secondary malignancies associated with GVHD, aggressive GVHD prophylaxis and treatment remains an area in need of ongoing improvement. The use of alemtuzumab as serotherapy is associated with very promising outcomes for a small number of patients in this cohort, though larger studies will be needed to confirm this.
As research over the past several decades has led to significant improvements in HSCT outcomes for patients with FA, so too does current research promise to continue building toward even better outcomes over the next several decades. Transplant-related toxicities remain a major cause of morbidity and mortality for patients with FA. One approach to decrease this is through the removal of genotoxic agents, namely, total body irradiation and busulfan, from the HSCT conditioning regimen. Although this has previously not been feasible due to the risk of graft rejection, a new monoclonal antibody targeting CD117 (c-KIT) has been developed (briquilimab, formerly JSP191), which offers the possibility of human hematopoietic stem cell depletion with only minimal toxicity.9 An alternate strategy to improve outcomes for patients with FA is through the use of gene therapy (autologous HSCT with ex vivo genetic manipulation), which offers the possibility of hematopoietic cure without the risk of GVHD.10 Clinical trials testing these approaches are still ongoing, though early outcomes from both have been promising. Through these and other advances, it is all but certain that the curve of improvement for patients with FA will continue to trend up.
Conflict-of-interest disclosure: A.M.S. declares no competing financial interests.