TO THE EDITOR:
Graft-versus-host disease (GVHD) is a major cause of morbidity- and treatment-related mortality1 after allogeneic hematopoietic stem cell transplantation (HSCT), affecting 40% to 60% of patients.2 GVHD is associated with microbiota disruptions, which occur with prophylactic antibiotic therapy, chemotherapy, and radiation given as part of HSCT.3-5 Microbiota-directed therapies such as prebiotics (microbially accessible dietary fibers) may provide a safe approach to reduce GVHD severity.6
Galactooligosaccharide (GOS) is a well-studied prebiotic available as a dietary supplement, which reduces inflammatory GI symptoms,7 promotes intestinal barrier function,8 improves NK cell activity,9 and modulates cytokine activity.9 We conducted an interventional study in an established major histocompatibility complex–mismatched bone marrow transplantation model to assess whether GOS supplementation impacts the microbiome and improves GVHD and survival.
Starting 7 days prior to transplant (Day 7) and continuing until death or Day-100 post-HSCT (Day 100), whichever occurred first, BALB/c recipient mice (Taconic Biosciences No. BALB) received 4% of daily calorie requirements as GOS, supplemented in drinking water; control mice received regular drinking water. Starting 3 days prior to transplant (Day 3), all mice were treated with imipenem–cilastatin to mimic antibiotics’ disruptive effects on microbial community10 (supplemental Methods, available on the Blood Web site). On transplant day (Day 0), mice received 8.5 Gy total body irradiation, followed by infusion of purified bone marrow and T-cells harvested from C57BL/6J donor mice (Jackson Laboratories no. 000664).11
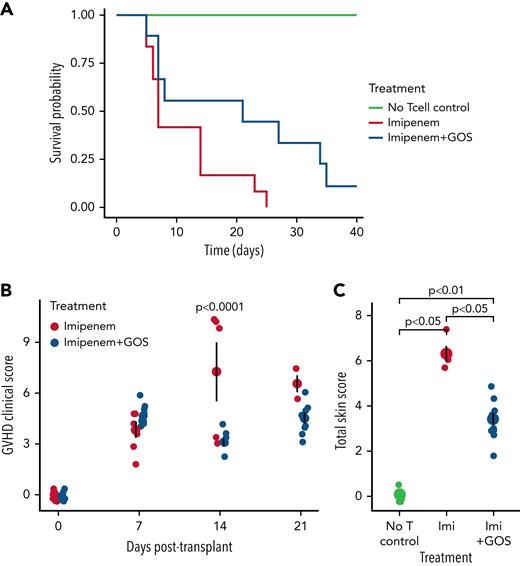
GOS-supplemented mice demonstrated improved survival compared with un-supplemented mice (P = .0087; Figure 1A). The survival improvement did not appear to be related to the potentially increased caloric intake from GOS, as body weight did not differ between the groups at any timepoint (supplemental Figure 1). Rather, GOS supplementation was linked to decreased GVHD severity, despite mice consuming GOS ad libitum (supplemental Methods), which could have contributed to individual variability in the results. First, the observed GVHD clinical score was lower in GOS mice than in control mice on Day-14 post-transplant (ANOVA P value = .0005, Tukey honestly significant difference P-value Day 14 = .0001; Figure 1B). Second, histologic examination of mice sacrificed at Day 14 showed decreased GVHD-specific pathologic changes of skin in GOS-treated mice (P = .0012, t-test; Figure 1C); no difference was observed in GVHD of gut (supplemental Figure 2) or liver (data not shown). Interestingly, GOS only improved HSCT outcomes in antibiotic-treated mice (supplemental Figure 3), which suggests prebiotics may counter antibiotics’ deleterious effects on commensal gut microbiota and clinical outcomes.5,10
Survival (A), GVHD clinical score (B), and histopathological score of skin (C) in imipenem-treated mice following HSCT, with and without GOS supplementation. GOS supplementation significantly improved survival (n = 12 mice per treatment group, n = 5 in “No Tcell control,” P = .0087, log-rank test; hazard ratio = .224, Cox regression, A). GVHD clinical score, an observational indicator of GVHD severity, was assessed weekly following transplantation for each mouse (n = 15 per group, B). Presence of GVHD and its severity was calculated out of six categories: body weight, skin, fur, posture, and activity as well as diarrhea (supplemental Table 1). This combined score was lower on Day 14 in GOS-supplemented mice (P < .0001, Tukey’s honestly significant difference post-hoc test following significant ANOVA, P = .0005). Tissue samples of skin were taken from the back of the neck and scored with a semiquantitative system for documenting GVHD damage with criteria as outlined in supplemental Table 2.25 Total skin score (a histological indicator of GVHD-mediated tissue damage in skin, graded by a blinded and independent pathologist) was assessed at Day 14 post-transplant in all surviving mice (three of 15 in GOS-free group, 10 of 15 in GOS group, C, P values calculated with Wilcoxon tests). Mean and standard error are depicted by larger points and error bars (B-C).
Survival (A), GVHD clinical score (B), and histopathological score of skin (C) in imipenem-treated mice following HSCT, with and without GOS supplementation. GOS supplementation significantly improved survival (n = 12 mice per treatment group, n = 5 in “No Tcell control,” P = .0087, log-rank test; hazard ratio = .224, Cox regression, A). GVHD clinical score, an observational indicator of GVHD severity, was assessed weekly following transplantation for each mouse (n = 15 per group, B). Presence of GVHD and its severity was calculated out of six categories: body weight, skin, fur, posture, and activity as well as diarrhea (supplemental Table 1). This combined score was lower on Day 14 in GOS-supplemented mice (P < .0001, Tukey’s honestly significant difference post-hoc test following significant ANOVA, P = .0005). Tissue samples of skin were taken from the back of the neck and scored with a semiquantitative system for documenting GVHD damage with criteria as outlined in supplemental Table 2.25 Total skin score (a histological indicator of GVHD-mediated tissue damage in skin, graded by a blinded and independent pathologist) was assessed at Day 14 post-transplant in all surviving mice (three of 15 in GOS-free group, 10 of 15 in GOS group, C, P values calculated with Wilcoxon tests). Mean and standard error are depicted by larger points and error bars (B-C).
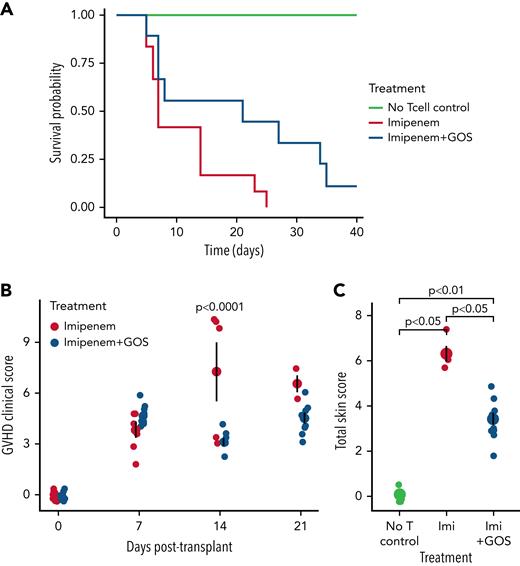
When we analyzed gut microbial community composition in recipient mice, we observed community structure changes attributable to GOS supplementation (P = .04, R2 = 0.08; Figure 2A). In GOS-treated mice, we identified two families with lower abundance (Bacteroidaceae and Bacteroidales_S24-7_group) and one (Porphyromonadaceae) with higher abundance (supplemental Figure 4A-B). Porphyromonadaceae is linked to butyrate production,12 and one member is associated with reduced GVHD severity.13 Reduced Bacteroidales_S24-7_group abundance is consistent with evidence that this group specializes in non-GOS type fiber degradation.14
Efficacy of GOS in ameliorating GVHD is dependent on the starting community composition and metabolic capacity of the gut microbiota. Principal component analysis of microbiota community composition in mice on the day prior to transplantation (A), in vitro SCFA production capacity of mouse stool prior to transplant (B-C), survival and clinical score of treatment and control BALB/c mice sourced from Jackson Laboratories (D-E), and gut microbiota among BALB/c mice sourced from Jackson Laboratories and Taconic Biosciences and their SCFA production (F-H). GOS supplementation altered community structure (n = 15 per group, P = .04, R2 = 0.080; PERMANOVA using weighted UniFrac distance, A). Mice whose diets had been supplemented with GOS harbored fecal microbiota that produced more butyrate (n = 3 replicate fermentations, P = .0082, t-test, B) and more total SCFA (n = 3 replicate fermentations, P = .024, t-test, C) than un-supplemented mice. GOS supplementation likely improved survival in Jackson mice (n = 12 per group, P = .058, log-rank test; hazard ratio = .449, Cox regression, D), but to a lesser extent than in Taconic mice (see Figure 1A). GVHD clinical score, an observational indicator of GVHD severity, was assessed weekly following transplantation for each mouse (n = 12 per group, E). Community composition was different between the two groups of mice (n = 25 mice in Jackson group, 13 in Taconic group, R2 = .18, P = .002, PERMANOVA using weighted UniFrac distance at the ASV level, F), and 23 of 42 genera were found to be differentially abundant between groups (q < 0.05, Benjamini–Hochberg-corrected Wilcoxon rank-sum test implemented with ALDEx2, G). All genera are plotted, with phylum designated by color and with significantly different genera designated by labels (∗Ruminococcaceae shortened to “R.” and Lachnospiraceae shortened to “L.” in subplot G). Benjamini–Hochberg-corrected q-value cutoff of 0.05 is shown as a horizontal dotted line. A positive effect size estimate indicates higher relative abundance in Taconic mice vs Jackson mice (G).The ability of gut microbiota to produce SCFA from GOS in vitro was assessed in both groups, with the experiment repeated with three different batches of mice, with two fermentations run per iteration as technical replicates. Taconic mice produced more total SCFA than did Jackson mice (n = 3 biological replicates [technical duplicates per experiment were averaged for statistical tests], P = .034, paired t-test, H). Mean and standard error are depicted by larger points and error bars (B,C,E,H).
Efficacy of GOS in ameliorating GVHD is dependent on the starting community composition and metabolic capacity of the gut microbiota. Principal component analysis of microbiota community composition in mice on the day prior to transplantation (A), in vitro SCFA production capacity of mouse stool prior to transplant (B-C), survival and clinical score of treatment and control BALB/c mice sourced from Jackson Laboratories (D-E), and gut microbiota among BALB/c mice sourced from Jackson Laboratories and Taconic Biosciences and their SCFA production (F-H). GOS supplementation altered community structure (n = 15 per group, P = .04, R2 = 0.080; PERMANOVA using weighted UniFrac distance, A). Mice whose diets had been supplemented with GOS harbored fecal microbiota that produced more butyrate (n = 3 replicate fermentations, P = .0082, t-test, B) and more total SCFA (n = 3 replicate fermentations, P = .024, t-test, C) than un-supplemented mice. GOS supplementation likely improved survival in Jackson mice (n = 12 per group, P = .058, log-rank test; hazard ratio = .449, Cox regression, D), but to a lesser extent than in Taconic mice (see Figure 1A). GVHD clinical score, an observational indicator of GVHD severity, was assessed weekly following transplantation for each mouse (n = 12 per group, E). Community composition was different between the two groups of mice (n = 25 mice in Jackson group, 13 in Taconic group, R2 = .18, P = .002, PERMANOVA using weighted UniFrac distance at the ASV level, F), and 23 of 42 genera were found to be differentially abundant between groups (q < 0.05, Benjamini–Hochberg-corrected Wilcoxon rank-sum test implemented with ALDEx2, G). All genera are plotted, with phylum designated by color and with significantly different genera designated by labels (∗Ruminococcaceae shortened to “R.” and Lachnospiraceae shortened to “L.” in subplot G). Benjamini–Hochberg-corrected q-value cutoff of 0.05 is shown as a horizontal dotted line. A positive effect size estimate indicates higher relative abundance in Taconic mice vs Jackson mice (G).The ability of gut microbiota to produce SCFA from GOS in vitro was assessed in both groups, with the experiment repeated with three different batches of mice, with two fermentations run per iteration as technical replicates. Taconic mice produced more total SCFA than did Jackson mice (n = 3 biological replicates [technical duplicates per experiment were averaged for statistical tests], P = .034, paired t-test, H). Mean and standard error are depicted by larger points and error bars (B,C,E,H).
We next examined if GOS altered microbiota metabolic capacity. Bacterial metabolites, including short chain fatty acids (SCFAs) such as butyrate, are associated with GVHD protection in patients,15 and butyrate supplementation ameliorated GVHD in mice.16 To evaluate effects of GOS on fecal microbiota SCFA production, we used an in vitro fermentation model17 (supplemental Methods). GOS-supplemented mice exhibited modified gut microbial metabolic capacity by transplant day (Day 0), characterized by increased butyrate production (P = .0082; Figure 2B) and total SCFA production (P = .024; Figure 2C). Increased butyrate production persisted to Day 3 post-transplant in the GOS-supplemented group compared with the un-supplemented group (P = .014; supplemental Figure 5A). Total SCFA production was lower in the GOS-supplemented group than in the un-supplemented group at Day 3 (P = .002; supplemental Figure 5B), but both butyrate and total SCFA production in GOS-supplemented mice remained elevated relative to Day 0 (P < .001, P = .001, respectively; t-tests; supplemental Figure 5C-D). The elevated levels of total SCFA production in un-supplemented mice may reflect rapid expansion in microbiome metabolism following release from antibiotic pressure18 on transplant day (Day 0). The protective effects of prebiotic treatment prior to transplant may have limited such expansion in the GOS-supplemented group. These findings suggest that prebiotic supplementation enhanced the ability of the antibiotic-impacted murine microbiota to produce beneficial SCFAs from GOS, which may then have contributed to protection from transplant-associated mortality.16
Given that GOS-mediated microbiota changes were associated with improved treatment outcomes, we wondered whether differences in baseline gut microbiota community composition might influence GOS therapeutic efficacy. Using genetically identical mice from a different vendor, Jackson Laboratories, we found that GOS-supplemented Jackson BALB/c recipient mice also showed improved survival (P = .058), with GOS supplementation providing a hazard ratio of 0.449 compared with un-supplemented mice (Figure 2D). However, this effect was not as strong in Jackson mice when compared with Taconic mice (hazard ratio 0.224). Further, the observed GVHD clinical score was not lower in GOS-supplemented Jackson mice compared with un-supplemented Jackson mice (Figure 2E).
We then probed the microbiota composition of the two genetically identical sets of mice. As expected, at baseline, stool microbiota of Taconic and Jackson mice were distinct (R2 = 0.18, P = .002; Figure 2F). We identified 84 of 319 ASVs as specifically differentially abundant between the sets of mice (q < 0.05), and 23 of 42 genera (q < 0.05; Figure 2G, supplemental Methods). Of these 23 genera, Bacteroides, Parabacteroides, and Prevotellaceae_UCG-001 were the most differentially abundant and uniformly increased in Taconic mice relative to Jackson, and each has a potential role in GOS utilization or gut health maintenance.19-22 Interestingly, Porphyromonadaceae members, which were enriched in GOS-treated Taconic mice (supplemental Figure 5B), were also enriched in mice treated with imipenem alone (supplemental Figure 5B) and were more abundant in untreated Taconic than Jackson mice at baseline (Figure 2G). Together, these observations suggest that Porphyromonadaceae may be required at higher abundances in order to mediate the positive outcomes observed with GOS supplementation.
We then compared the in vitro fecal microbiota metabolic activity from Jackson and Taconic mice collected prior to GOS supplementation. We found that stool microbiota from Taconic mice (which showed a greater therapeutic response to GOS) produced more total SCFA from GOS fermentation than did those from Jackson mice (P = .034, paired t-test; Figure 2H, supplemental Methods). Together, these data support the hypothesis that GOS supplementation is differentially efficacious across mice harboring distinct microbiota.
The observation that prebiotic efficacy depends on baseline microbiota community composition suggests that prebiotic therapies could be optimally adapted to individual patients. Studies have found that individuals non-responsive to one prebiotic may be more responsive to a chemically distinct prebiotic.17,23,24 Alternatively, patients could be stratified as prebiotic responders and non-responders according to habitual dietary fiber consumption24 or fecal SCFA concentration measurement.24 Future studies may pave the way for personalized prebiotics, or combinations of prebiotics and/or probiotics, to improve microbiota health and HCT outcomes.
To our knowledge, this is among the first published instances of prebiotics improving HSCT outcome in a murine model and supports previous work showing the importance of the SCFA butyrate in mitigating GVHD.16 A phase-1 dose escalation study of GOS in HSCT patients is currently underway (ClinicalTrials ID NCT04373057). If therapeutic benefits of prebiotic treatment in HSCT are seen in patients, prebiotics may become a powerful adjunctive therapy for preventing GVHD.
Acknowledgments
The authors thank Heather Durand and Sharon Jiang for assistance with this study.
This work was supported by a grant from the Damon Runyon Cancer Research Foundation (to A.D.S. and L.A.D.). L.A.D. acknowledges support from NIH grant DK116187.
Authorship
Contribution: Z.C.H., L.A.D., and A.D.S.: conceptualization; Z.C.H., C.L., A.B., B.C.N., Y.J., M.C., D.M.C., B.J.C., L.A.D., and A.D.S.: methodology; Z.C.H., C.L., A.B., M.C.K., and D.M.C.: investigation; Z.C.H.: visualization; Z.C.H., L.A.D., and A.D.S.: funding acquisition; L.A.D. and A.D.S.: project administration; L.A.D., A.D.S., B.J.C., and N.J.C.: supervision; Z.C.H., H.T., B.C.N., L.A.D., and A.D.S.: writing – original draft; and Z.C.H., L.A.D., A.D.S., H.T., B.C.N., C.L., D.M.C., B.J.C., N.J.C., and M.C.K.: writing – review and editing.
Conflict-of-interest disclosure: A.D.S. has received research supplies from Royal DSM and Clasado for studies not described in this manuscript. L.A.D. was a member of the Kaleido Biosciences Strategic Advisory Board and retains equity in the company. The remaining authors declare no competing financial interests.
Correspondence: Lawrence A. David, Molecular Genetics & Microbiology, Duke Microbiome Center, Duke University, Box 103053, Durham, NC 27710; e-mail: lawrence.david@duke.edu; and Anthony D. Sung, Hematologic Malignancies & Cellular Therapy, Duke Blood Cancer Center, 2400 Pratt St, Suite 5000, Durham, NC 27710; e-mail: anthony.sung@duke.edu.
References
Author notes
L.A.D. and A.D.S. are joint senior authors.
The full-text version of this letter contains a data supplement.
There is a Blood Commentary on this article in this issue.

![Efficacy of GOS in ameliorating GVHD is dependent on the starting community composition and metabolic capacity of the gut microbiota. Principal component analysis of microbiota community composition in mice on the day prior to transplantation (A), in vitro SCFA production capacity of mouse stool prior to transplant (B-C), survival and clinical score of treatment and control BALB/c mice sourced from Jackson Laboratories (D-E), and gut microbiota among BALB/c mice sourced from Jackson Laboratories and Taconic Biosciences and their SCFA production (F-H). GOS supplementation altered community structure (n = 15 per group, P = .04, R2 = 0.080; PERMANOVA using weighted UniFrac distance, A). Mice whose diets had been supplemented with GOS harbored fecal microbiota that produced more butyrate (n = 3 replicate fermentations, P = .0082, t-test, B) and more total SCFA (n = 3 replicate fermentations, P = .024, t-test, C) than un-supplemented mice. GOS supplementation likely improved survival in Jackson mice (n = 12 per group, P = .058, log-rank test; hazard ratio = .449, Cox regression, D), but to a lesser extent than in Taconic mice (see Figure 1A). GVHD clinical score, an observational indicator of GVHD severity, was assessed weekly following transplantation for each mouse (n = 12 per group, E). Community composition was different between the two groups of mice (n = 25 mice in Jackson group, 13 in Taconic group, R2 = .18, P = .002, PERMANOVA using weighted UniFrac distance at the ASV level, F), and 23 of 42 genera were found to be differentially abundant between groups (q < 0.05, Benjamini–Hochberg-corrected Wilcoxon rank-sum test implemented with ALDEx2, G). All genera are plotted, with phylum designated by color and with significantly different genera designated by labels (∗Ruminococcaceae shortened to “R.” and Lachnospiraceae shortened to “L.” in subplot G). Benjamini–Hochberg-corrected q-value cutoff of 0.05 is shown as a horizontal dotted line. A positive effect size estimate indicates higher relative abundance in Taconic mice vs Jackson mice (G).The ability of gut microbiota to produce SCFA from GOS in vitro was assessed in both groups, with the experiment repeated with three different batches of mice, with two fermentations run per iteration as technical replicates. Taconic mice produced more total SCFA than did Jackson mice (n = 3 biological replicates [technical duplicates per experiment were averaged for statistical tests], P = .034, paired t-test, H). Mean and standard error are depicted by larger points and error bars (B,C,E,H).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/140/21/10.1182_blood.2021015178/5/m_blood_bld-2021-015178-gr2.jpeg?Expires=1769097184&Signature=ZDh3xkVPYe3IHahjl-GCKmxDPYLtU6ksSem9wJsX5NYOj0c-wbZiCTNC8dTHfTLC6dgz~qDlMa20JlkT0ZPdfkgiukLOj5Sbmi~nmvDho1CP3STDSXdRB0voNpwdB6TA~ByKDkTIj-DrA9zHohemdAJTNevwgrn~7MS7zVGmX6Gl8z8bcE5krYvOkag~-B5QpUr~4bi2a1K3b-5heODXnLFVaYdyRfrUwrAp9KPssYuIIT3mwDxVgiHednDVetpyYC-56TDssSmj6pIUI~YD5aniwqRNONcjqSNhXPhFnoOalLD517MXhUwo-Z3IsnhxM~X~nWwn3OZox27yMhWyxA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Efficacy of GOS in ameliorating GVHD is dependent on the starting community composition and metabolic capacity of the gut microbiota. Principal component analysis of microbiota community composition in mice on the day prior to transplantation (A), in vitro SCFA production capacity of mouse stool prior to transplant (B-C), survival and clinical score of treatment and control BALB/c mice sourced from Jackson Laboratories (D-E), and gut microbiota among BALB/c mice sourced from Jackson Laboratories and Taconic Biosciences and their SCFA production (F-H). GOS supplementation altered community structure (n = 15 per group, P = .04, R2 = 0.080; PERMANOVA using weighted UniFrac distance, A). Mice whose diets had been supplemented with GOS harbored fecal microbiota that produced more butyrate (n = 3 replicate fermentations, P = .0082, t-test, B) and more total SCFA (n = 3 replicate fermentations, P = .024, t-test, C) than un-supplemented mice. GOS supplementation likely improved survival in Jackson mice (n = 12 per group, P = .058, log-rank test; hazard ratio = .449, Cox regression, D), but to a lesser extent than in Taconic mice (see Figure 1A). GVHD clinical score, an observational indicator of GVHD severity, was assessed weekly following transplantation for each mouse (n = 12 per group, E). Community composition was different between the two groups of mice (n = 25 mice in Jackson group, 13 in Taconic group, R2 = .18, P = .002, PERMANOVA using weighted UniFrac distance at the ASV level, F), and 23 of 42 genera were found to be differentially abundant between groups (q < 0.05, Benjamini–Hochberg-corrected Wilcoxon rank-sum test implemented with ALDEx2, G). All genera are plotted, with phylum designated by color and with significantly different genera designated by labels (∗Ruminococcaceae shortened to “R.” and Lachnospiraceae shortened to “L.” in subplot G). Benjamini–Hochberg-corrected q-value cutoff of 0.05 is shown as a horizontal dotted line. A positive effect size estimate indicates higher relative abundance in Taconic mice vs Jackson mice (G).The ability of gut microbiota to produce SCFA from GOS in vitro was assessed in both groups, with the experiment repeated with three different batches of mice, with two fermentations run per iteration as technical replicates. Taconic mice produced more total SCFA than did Jackson mice (n = 3 biological replicates [technical duplicates per experiment were averaged for statistical tests], P = .034, paired t-test, H). Mean and standard error are depicted by larger points and error bars (B,C,E,H).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/140/21/10.1182_blood.2021015178/5/m_blood_bld-2021-015178-gr2.jpeg?Expires=1769097185&Signature=s5T9CiEbz4uGbW9xMsRNOs-VxNmZO5pd-OpLUEkodsXw4G11X9E92qip-mQMB2MG9zpuDCtLy-qEGjJ18tA8DvG8nGfHwnrTDDs9UCT285X~lJvE6xXzOUzZrgZ~GdkeJ2fw6ciQLRgQpv4PYP4ymdd~fYzV4xSzGd9A7Fzi1hRrOog~uZ1SZQo0yv45qQUtVHoBD1p1psHXjnTLGf1Q0m-alYqkmG1GTS4~kLnB5Kz7HRYuoSsdJQQqortGAXaPp3UqKyh-~jFU2PmhkWOBw2L1WkDy9DMFcHYrJqnY3UAXAS~7B7IOd7PaVUR5EffYkfggiba25Mjo9SMiR93Zyw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)