Background: Ibrutinib has become a widely used drug in the management of B-cell lymphoproliferative neoplasms (BLPNs). Past or active HBV infection is an exclusion criteria for the clinical trials of ibrutinib and for patients (pts) treated outside clinical trials there are contradictory data or suggestions concerning hepatitis B virus (HBV) prophylaxis. In this study, we evaluated the real-life data on the outcome of HBV infection status of pts with BLPNs receiving ibrutinib.
Methods: Demographic features, previous treatments (including rituximab (RTX)) and follow-up durations, HBV infection and reactivation status, prophylaxis information, serum transaminase and total bilirubin levels of the pts were noted retrospectively. Past HBV infection was defined as concurrent detectable HBV core IgG antibody (Ab) (anti-HBc IgG) and undetectable HBV surface Ag (HbsAg) ± HBV surface Ab (anti-HBs). Active HBV infection was defined as HbsAg and/or HBV core IgM Ab (anti-HBc IgM) positivity. Quantitative HBV-DNA PCR results are also recorded, when available.
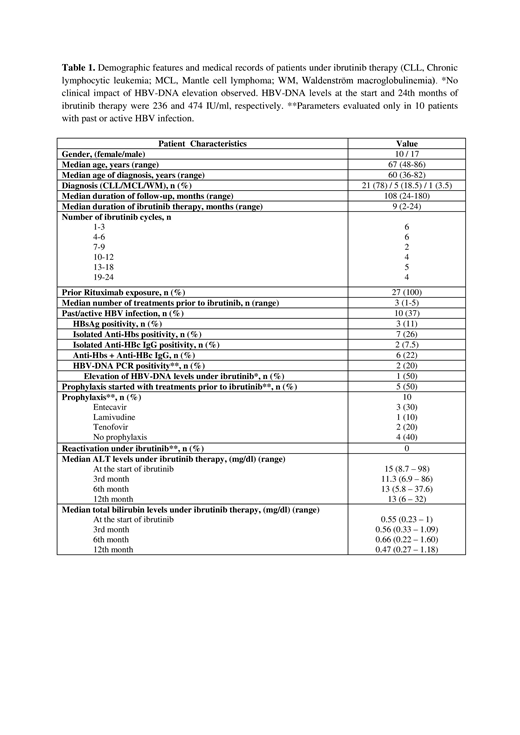
Results: Twenty-seven consecutive pts were included, and pts' characteristics are displayed in Table 1. Median durations of ibrutinib therapy and entire follow-up were 9 and 108 months, respectively. All pts had received RTX prior to ibrutinib. Among the 27 patients, 8 had past HBV infection, while 2 had active HBV infection. HBV prophylaxis or treatment was started before ibrutinib in 5 pts, and simultaneously with ibrutinib in one. The remaining 4 pts with past HBV infection were not given any prophylaxis. Among these 4 cases with anti-HBc IgG positivity, three were also positive for anti-HBs. Currently, these 4 pts, including the one with a negative anti-HBs status, are at the 2nd, 4th, 12th, and 16th cycles of ibrutinib. To date, no signs of HBV reactivation was detected in our cohort.
Discussion & Conclusion: All pts in our cohort had received RTX containing regimens prior to ibrutinib. In our analysis, 5 pts were under HBV prophylaxis or treatment when ibrutinib was started. Two of these had active HBV infection. Simultaneous HBV prophylaxis was started with ibrutinib for one pt and the remaining 4 pts with past HBV infection (anti-HBs + anti-HBc IgG positivity) did not receive any prophylaxis, but they were planned to be followed by monitoring the anti-HBs titers. In a study, 21 BLPN pts (10 treated with RTX previously) with occult HBV infection with a median follow-up of 9.5 months treated with ibrutinib without anti-HBV prophylaxis, and only two HBV reactivations were reported (Hammond et al. Blood. 2018;131(17):1987-1989). In another study, no HBV reactivation was detected in 7 CLL pts with past HBV infection, with a median follow-up duration of 25 months under ibrutinib, without any anti-viral prophylaxis (Tedeschi et al. Leuk Lymphoma. 2017; 58(12):2966-2968). The increase in the number of pts treated with ibrutinib, is followed by the awareness about the infectious complications including HBV reactivation. The consecutive use of RTX and ibrutinib has increased the risk of HBV-related complications. This issue has become more important in areas with a higher prevalence of HBV. Although the number of pts is limited and follow-up periods are relatively short, our results may indicate that pts with occult HBV infection treated with ibrutinib for BLPNs, might only be closely followed for HBV reactivation, rather than routine anti-HBV prophylaxis. Since pts with previous RTX exposure are likely to lose anti-HBs antibodies, ibrutinib-treated pts with both anti-HBs and anti-HBc IgG positivity should be closely monitored for possible risk of HBV reactivation. Anti-viral prophylaxis under ibrutinib therapy might still be an option for the selected cases.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.