BACKGROUND: DLBCL is the most common and a potentially curable non-Hodgkin lymphoma. Multiple previous studies have shown that minority populations have worse outcomes compared to Caucasians (Tao L, Blood 2014; Griffiths R, BMC Cancer 2010; Koroukian et al, Cancer, 2010, Shenoy PJ Cancer 2010). Moreover, it has been reported that uninsured and Medicaid insured patients with DLBCL have inferior survival compared to privately insured patients (Han X, Cancer 2014; Koroukian et al, Cancer 2010;). It has also been well established that minorities are underrepresented in clinical trials (Gerrero S, Sci Rep 2018; Kwiatkowski K, Cancer 2013). We present the baseline characteristics, treatment paradigms and outcomes of Caucasian (C) and non-Caucasian (NC) patients with de novo DLBCL treated at a single academic hybrid cancer center.
METHODS: We collected demographic, disease, insurance coverage, treatment characteristics, and treatment outcomes for patients with de novo DLBCL who presented between January 2016 and January 2019 at Levine Cancer Institute, Charlotte, North Carolina. Patient race, C or NC were self-reported. Insurance was categorized as Government (Medicaid or Medicare), Private, or Uninsured. We used the Revised International Prognostic Index (R-IPI) to risk stratify patients. Double-hit lymphomas (DHL) were defined as having the MYC translocation with either BCL2 or BCL6 translocation on fluorescent in situ hybridization. Treatments included standard chemoimmunotherapies, stem cell transplantation and clinical trials including chimeric antigen receptor therapies (CART). Outcomes of overall survival (OS) and progression free survival (PFS) were calculated using the Kaplan Meier method and compared with log rank test. Demographic data was compared using Fisher's Exact tests.
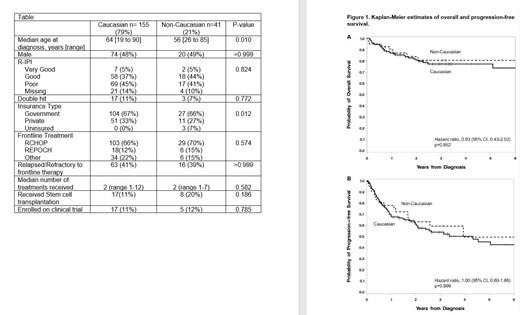
RESULTS: One hundred and ninety-six consecutive patients with de novo DLBCL were included in the analysis [155 (79%) = C, 41 (21%) = NC] (Table). The NC group was predominantly African American (71%) followed by Hispanic (15%). Prognostic scores (R-IPI) and the incidence of DHL were similar between C and NC. The median age at diagnosis in the NC group was lower than in C. There were significant differences in insurance coverage between the 2 groups (p=0.012). The C group did not have any uninsured patients and had more patients with private insurance (33%) compared to the NC group (7% uninsured and 27% with private insurance). The most common frontline treatment was RCHOP (C=66%, NC=70%) followed by dose adjusted REPOCH (C=12%, NC=15%). Median follow up was 31.6 months. There was no difference in OS and PFS between the 2 groups (Figure 1). OS at 2 years from date of diagnosis was 81% for C and 84% for NC, p=0.852. Two-year PFS from time of diagnosis were similar for both groups: 61% for C and 63% for NC, p=0.999.
Similar numbers of patients in both groups developed relapsed or refractory (R/R) disease after frontline therapy. Median number of treatments was 2 for both groups, p=0.582. For patients who developed R/R DLBCL, the 2-year OS was 60% for C and 63% for NC, p=0.590. Similar proportions underwent stem cell transplantation: 11% for C and 20% for NC, p= 0.186. Clinical trial enrollment was comparable: 11% for C and 12% for NC, p=0.785.
CONCLUSION: Unlike previous population-based studies that have shown racial disparities with superior outcomes for Caucasians and for patients with private insurance, our single center experience demonstrates similar survival outcomes between Caucasians and non-Caucasians diagnosed with de novo DLBCL, despite differences in insurance coverage favoring Caucasians. In the R/R setting, similar proportions of both groups underwent stem cell transplantation and enrolled on clinical trials. The likely explanation is that our safety net cancer center, with extensive nurse navigator support and access to standard treatments, stem cell transplants and cutting-edge clinical trials may abrogate the inferior outcomes in minority populations that have been previously reported.
Symanowski:Immatics: Consultancy; Eli Lilly: Consultancy; Carsgen Therapeutics: Consultancy; Boston Biomedical: Consultancy. Park:Rafael Pharma: Membership on an entity's Board of Directors or advisory committees; BMS: Consultancy, Research Funding; Gilead: Speakers Bureau; Teva: Consultancy, Research Funding; G1 Therapeutics: Consultancy; Seattle Genetics: Research Funding, Speakers Bureau. Avalos:Juno: Membership on an entity's Board of Directors or advisory committees; Best Practice-Br Med J: Patents & Royalties: receives royalties from a coauthored article on evaluation of neutropenia. Jacobs:Genentech: Speakers Bureau; AstraZeneca: Speakers Bureau; TG Therapeutics: Honoraria, Research Funding; AbbVie: Consultancy, Speakers Bureau; JUNO: Consultancy; Gilead: Consultancy; Pharmacyclics LLC, an AbbVie Company: Research Funding, Speakers Bureau. Ghosh:TG Therapeutics: Consultancy, Honoraria, Research Funding; SGN: Consultancy, Honoraria, Research Funding, Speakers Bureau; Bristol-Myers Squibb: Honoraria, Speakers Bureau; Gilead: Consultancy, Honoraria, Speakers Bureau; Janssen: Consultancy, Honoraria, Research Funding, Speakers Bureau; Pharmacyclics LLC, an AbbVie Company: Consultancy, Honoraria, Research Funding, Speakers Bureau; Celgene: Consultancy, Research Funding; Genentech: Research Funding; AstraZeneca: Honoraria, Speakers Bureau; Forty Seven Inc: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract