Introduction: Hematopoietic cell transplantation (HCT)-related factors, such as total body irradiation (TBI) used for conditioning, graft-versus-host disease (GvHD), and prolonged exposure to calcineurin-inhibitors, can result in high risk for subsequent skin cancers in long-term survivors. Previous studies examining skin cancers after HCT have largely focused on patients transplanted in earlier eras (<2000), which may not be as informative for providers caring for survivors treated with contemporary conditioning (e.g. less myeloablative) approaches or GvHD treatment regimens, and do not account for newer stem cell sources (e.g. cord, haploidentical) with their evolving immunosuppression risk. Additionally, due to registry reporting, past studies have largely focused on melanoma, which underestimates the burden due to more common skin cancers such as squamous cell carcinoma (SCC) and basal cell carcinoma (BCC). The current study describes the incidence and risk factors of melanoma and non-melanoma skin cancer in a large contemporary cohort of HCT survivors.
Methods: 2338 consecutive patients who underwent a first HCT between 2005 and 2014 at City of Hope (COH) and survived ≥1 year, were included in the study. Patients with a history of skin cancer prior to HCT were excluded from the cohort. All skin cancers were validated using pathology reports, and physician notes (20%) whenever the former was not available. Skin cancers included SCC, BCC, melanoma, atypical fibroxanthoma, and merkel cell carcinoma. Patients were followed from HCT to death, second HCT, last known alive date, or December 31, 2018, whichever occurred first. Cumulative incidence of skin cancer was estimated taking into consideration competing risk of death. Fine-Gray sub-distributional hazard regression was used to calculate hazard ratio (HR) estimates, adjusted for relevant covariates.
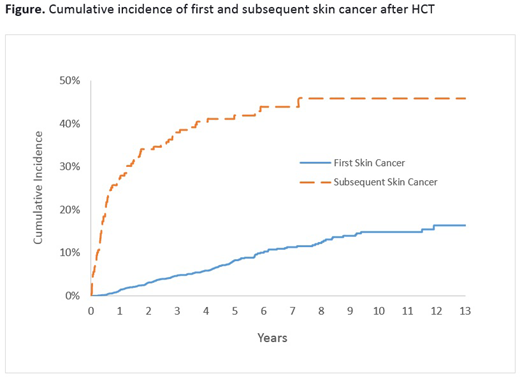
Results: Median age at HCT was 51.8y (range: 0.7-78.7); 57.6% were male; 56.5% were non-Hispanic white (NHW); 4.4% had a history of Pre-HCT cancer (antecedent to primary diagnosis); 47.7% underwent allogeneic HCT; 22.0% received myeloablative TBI (≥1200 cGy)-based conditioning. Among survivors of allogeneic HCT, 57.2% developed ≥moderate chronic GvHD (84% involving the skin); the most common immunosuppressive agents used for the management of ≥moderate chronic GvHD were tacrolimus (84.6%), followed by sirolimus (76.5%), and cyclosporine (28.4%). Burden of skin cancer over time: 179 survivors developed a total of 450 skin cancers after HCT; median time from HCT to first skin cancer was 2.8 years (range: 0.2-13.6). SCC was the most common subtype (59.1%), followed by BCC (31.3%), melanoma (5.1%), and other (4.4%); 43.4% of patients with SCC had invasive or high grade disease at presentation. The cumulative incidence of de novo skin cancer after HCT was 8.3% at 5 years, and 14.8% at 10 years (figure). Eighty-nine patients with skin cancer had a second skin cancer at a median of 0.5 years (range: 0.2 to 7.2) from the first; 55.1% were of a different histology. The cumulative incidence of a second skin cancer was 41.9% at 5-years (figure). Risk factors: Multivariate analysis revealed male sex (HR=1.7, p=0.0008), NHW race/ethnicity (HR=9.0, p<0.0001), older age HCT (≥50years at HCT, HR=2.4, p<0.0001), allogeneic HCT (HR=2.2, p<0.0001), and a history non-skin cancer prior to HCT (HR=1.7, p=0.04) to be significant and independent predictors of post-HCT skin cancer risk. Among allogeneic HCT patients, neither severity of chronic GvHD, location (e.g. skin), nor duration of post-HCT immunosuppression were associated with risk of de novo skin cancer, irrespective of skin cancer histology.
Conclusions: This study confirms the higher risk of skin cancer by sex, race/ethnicity, and age at HCT, and highlights the greater than two-fold risk of skin cancer among allogeneic HCT survivors compared to autologous patients, a finding that is not entirely explained by GvHD and its treatment. Further, we identified a previously unreported association between pre-HCT (non-skin) cancer and post-HCT cancer risk, and describe the very high burden of multiple histologically distinct skin cancers over time. Taken together, these data set form the basis for implementation of updated risk-based screening and prevention practices in survivors at highest risk of skin cancer after HCT.
Abdulla:Johnson & Johnson: Research Funding; Elorac: Research Funding; Trillium: Research Funding; Stemline: Research Funding; MiRagen: Research Funding; Bionz: Research Funding; Mallinckrodt: Research Funding; Mallinckrodt: Consultancy; Mallinckrodt: Speakers Bureau. Nakamura:Merck: Membership on an entity's Board of Directors or advisory committees; Celgene: Other: support for an academic seminar in a university in Japan; Alexion: Other: support to a lecture at a Japan Society of Transfusion/Cellular Therapy meeting ; Kirin Kyowa: Other: support for an academic seminar in a university in Japan.
Author notes
Asterisk with author names denotes non-ASH members.