Abstract
Metabolic reprogramming is a hallmark of cancer. MYC oncoproteins control many aspects of this response, by inducing the expression of genes involved in mitochondrial biogenesis, glycolysis, glutaminolysis and amino acid transport. This coordinated response allows cancer cells to meet the demands for macromolecules and energy necessary to sustain the anabolic state.
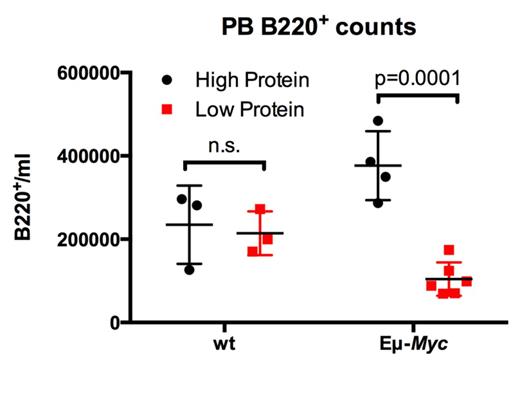
Normal cells adapt to nutrient-limiting conditions, such as amino acid (AA) starvation, by activating the autophagy-lysosomal pathway that is necessary for the maintenance of amino acid pools and for providing other building blocks (e.g., ATP) that are needed for cell survival. Surprisingly, our ex vivo and in vivo studies of premalignant and neoplastic MYC-expressing B cells of Eμ-Myc transgenic mice, and of human MYC-driven B cell lymphoma (e.g., Burkitt lymphoma), revealed that MYC suppresses the catabolic autophagy-lysosomal pathway, and that, accordingly, Myc-expressing premalignant and neoplastic B cells are exquisitely sensitive to AA starvation. For example, analyses of the effects of low (6%) versus high (20%) protein diets revealed that limiting AA pools in vivo selectively reduces the numbers of circulating premalignant Eμ-Myc B220+ B cells without affecting B cell numbers in wild type littermate mice. Thus, MYC-driven tumor cells are unable to sufficiently adapt to a state of nutrient deprivation (Figure 1).
Expression analyses revealed that this MYC suppresses the autophagy-lysosomal system by transcriptionally repressing genes that encode regulators and components of this pathway, and that this response is a hallmark of human malignancies with MYC involvement. Further, suppressing these genes has functional consequences, where MYC provokes marked reductions in autophagic flux that lead to marked increases in the levels of cargo such as p62/Sequestrin that are normally degraded by this pathway.
A master regulator of autophagy and lysosomal biogenesis is TFEB that, like MYC, functions as a basic helix-loop-helix leucine zipper transcription factor and shares a similar DNA recognition sequence. Our studies suggest that MYC blocks TFEB function at three levels. First, MYC can directly repress TFEB transcription. Second, MYC can directly repress TFEB transcription targets by competing with TFEB for binding to the promoter-regulatory regions of autophagy-lysosome gene targets. Third, MYC-expressing B cells have activated mTORC1, which phosphorylates TFEB and blocks its nuclear localization. Notably, forced reactivation of the autophagy-lysosomal pathway via inducible expression of a of a constitutively active (mTORC1-resistant and nuclear) form of TFEB (TFEBS211A) disables the malignant state, where TFEBS211A triggers cell cycle arrest and senescence of both mouse and human MYC-driven lymphomas ex vivo, and compromises tumorigenic potential in vivo. Thus, TFEB acts as tumor suppressor for MYC-driven malignancies.
We hypothesized that MYC-driven tumor cells compensate for the reductions in the autophagy pathway and maintain AA homeostasis by activating compensatory mechanisms, including AA transport and the proteasome. In support of this notion, the expression of AA transporters and components of the proteasome, and AA transport and proteasome activity, are markedly augmented in premalignant and neoplastic MYC-expressing B cells. Accordingly, MYC-expressing B cells are exquisitely sensitive to treatment with proteasome inhibitors.
Collectively, these findings suggest that MYC drives the anabolic state by suppressing the catabolic autophagy-lysosomal pathway, and that to maintain AA pools MYC-driven cancer cells up-regulate AA transport and the proteasome. This scenario provides attractive opportunities for combination therapies that should disable MYC-driven malignancies, including protein-restricted diets and proteasome and TORC1.
Premalignant Eμ-Myc and wild type (WT) littermates were treated for one week with low (6%) and high (20%) protein diets and B220+ cell numbers in peripheral blood were assessed.
Premalignant Eμ-Myc and wild type (WT) littermates were treated for one week with low (6%) and high (20%) protein diets and B220+ cell numbers in peripheral blood were assessed.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.