Abstract
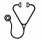
Introduction: Within clinical trials in CLL, response assessment is based on the NCI 1996 guidelines or its update, the iwCLL 2008 guidelines, respectively. Whereas detailed response criteria with clear cut-off values are provided for the assessment of most parameters, a few parameters such as splenomegaly are not defined quantitatively in the guidelines. In addition, the detection of MRD, which was recommended for clinical trials but not formally included in the definition of response, is gaining increasing importance. Both MRD negativity (with a threshold of <10-4 leukemic cells per leukocytes) and the occurrence of a complete response (CR) predict long progression free survival (PFS). In order to investigate the value of MRD with respect to clinical response, the MRD status was explored in patients (pts) with CR and partial remission (PR) in two phase III trials of the GCLLSG. Furthermore, we evaluated the relevance of residual splenomegaly, lymphadenopathy or bone marrow involvement in MRD negative (-) pts with clinical PR.
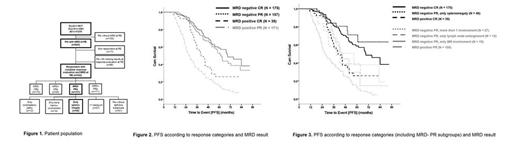
Patients and Methods: 542 pts from two prospective phase III trials of the GCLLSG (CLL8 trial: fludarabine and cyclophosphamide without (FC) or with rituximab (FCR); CLL10 trial: FCR vs bendamustine and rituximab (BR)) were included in the analysis (Figure 1). The comprehensive dataset included MRD results from peripheral blood at final restaging (RE) (2 months after the end of last treatment cycle), bone marrow (BM), clinical and radiological assessment for organomegaly and lymphadenopathy. Clinical response was defined according to the iwCLL 2008 guidelines. Splenomegaly was determined by physical and radiological examination. Moreover, different cut-off values defining splenomegaly by CT or ultrasound (longest diameter >12 cm and >14 cm) were investigated. PFS was analyzed using Kaplan-Meier methodology, and survival curves were compared using two-sided log-rank tests. Additionally hazard ratios (HR) and 95% confidence intervals (CI) were calculated. Significance was set at a p value<0.05. No adjustments for multiple testing were performed.
Results: 542 pts were included in the analysis. Their median age was 61 years, median CIRS score 2 (range 0-6), median follow up time 45.9 months (mo) (range 5.5-96.1). 121 pts (22.3%) received FC, 283 (52.2%) FCR and 138 (25.5%) BR. 13.3% of pts had Binet stage A, 52.3 % stage B and 34.4% stage C disease. Among 514 pts with IGHV results, 63.0% had an unmutated status. In 524 pts FISH was available, del(17p) was only found in 1.3% (exclusion criterion in CLL10 trial), del(11q) in 25.0%, 12+ in 10.1%, normal in 25.4%, and del(13q) in 38.2% of the pts.
MRD negativity was achieved in 81.8% (175/214) of pts with CR and in 47.9% (157/328) of the pts with PR, respectively. There was a statistically significant difference in PFS between MRD- CRs and MRD positive (+) CRs (69.2 mo vs 40.4 mo; HR 0.445, 95% CI=0.282-0.703, p=0.001). Additionally, there was a statistically significant difference between MRD- PRs and MRD+ CRs (61.7 mo vs 40.4 mo; HR 0.537, 95% CI=0.340-0.847, p=0.008). No statistically significant difference between MRD- CRs and MRD- PRs was detected (69.2 mo vs. 61.7 mo; HR 0.822, 95% CI=0.572-1.182, p=0.29) (Figure 2).
Of the 157 pts presenting with an MRD- PR, 106 pts were evaluable for remaining CLL involvement: 48 pts (45.3%) had a splenomegaly, 12 (11.3%) lymphadenopathy, 19 (17.9%) bone marrow involvement as the sole abnormality. Only 25.5% (27) of the pts had more than one abnormality. There was no statistically significant difference in PFS between MRD- PRs with single splenomegaly and MRD- CRs (not reached (NR) vs 69.2 mo; HR 0.737, 95% CI=0.387-1.404, p=0.4). Moreover, patients with MRD- PRs and single splenomegaly had a statistically significant longer PFS than MRD+ CR pts (NR vs 40.4 mo; HR 0.348, 95% CI=0.172-0.701, p=0.003). (Figure 3) The difference was independent of the cut-off values used for splenomegaly (12 cm or 14 cm) (p=0.001 and p=0.03).
Conclusion: MRD negativity determined in the peripheral blood after end of treatment is a potent predictor of treatment efficacy regardless of the clinical response assessment. The persistence of splenomegaly as sole abnormality post treatment in MRD- patients has no negative influence on PFS. More data are needed to prove the relevance of residual BM involvement and lymphadenopathy in MRD- PR pts. These results support the use of MRD for response evaluation.
Boettcher:Roche: Honoraria, Research Funding, Travel grant Other. Ritgen:Roche: Honoraria, Research Funding, Travel grant Other. Cramer:Mundipharma: Travel grant, Travel grant Other; Roche: Travel grant Other. Maurer:Mundipharma: Travel grant Other. Doehner:Roche: Research Funding. Stilgenbauer:Roche: Consultancy, Honoraria, Research Funding. Kneba:Mundipharma: Consultancy, Research Funding; Roche: Consultancy, Research Funding. Fischer:Roche: Travel grant Other. Hallek:Mundipharma: Consultancy, Research Funding; Roche: Consultancy, Research Funding. Eichhorst:Roche: Membership on an entity's Board of Directors or advisory committees, Research Funding, Travel grant Other; Mundipharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Travel grant, Travel grant Other; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Gilead: Consultancy. Off Label Use: The Combination of Bendamustine and Rituximab is not approved for frontline chemoimmunotherapy of CLL.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract