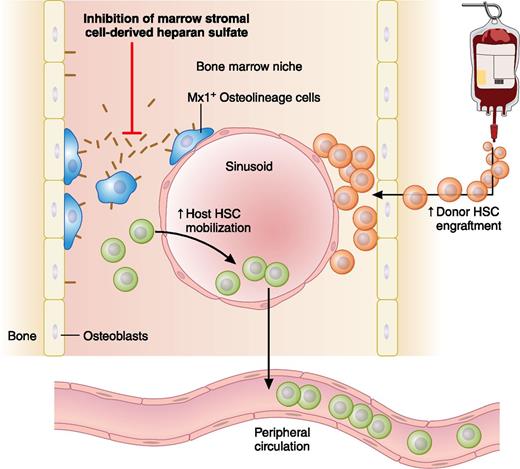
In this issue of Blood, Saez et al demonstrate that inhibition of heparan sulfate proteoglycan production by bone marrow osteolineage stromal cells results in hematopoietic stem cell egress from the bone marrow niche into the peripheral circulation.1
Inhibiting Mx1+ osteolineage cell production of heparan sulfate proteoglycans disrupts HSC interactions within the bone marrow niche, leading to potential clinical applications that include enhancing donor stem cell mobilization in response to granulocyte colony-stimulating factor (G-CSF) and enabling donor stem cells to engraft in the host bone marrow niche without cytotoxic conditioning. Professional illustration by Patrick Lane, ScEYEnce Studios.
Inhibiting Mx1+ osteolineage cell production of heparan sulfate proteoglycans disrupts HSC interactions within the bone marrow niche, leading to potential clinical applications that include enhancing donor stem cell mobilization in response to granulocyte colony-stimulating factor (G-CSF) and enabling donor stem cells to engraft in the host bone marrow niche without cytotoxic conditioning. Professional illustration by Patrick Lane, ScEYEnce Studios.
Promising new stem cell–based therapies have curative potential for many children and adults with nonmalignant blood cell disorders. Such strategies include autologous transplantation of hematopoietic stem cells (HSCs) genetically “corrected” by gene editing or transfer technologies2 and allogeneic familial haploidentical stem cell transplantation.3 Unfortunately, these approaches currently share 2 common limitations. First, both therapies require mobilization of high doses of donor CD34+ cells for optimal efficacy. Second, both also require considerable cytotoxic conditioning to achieve durable engraftment, resulting in significant toxicity risk or exclusion of patients with preexisting comorbidities.
In their intriguing translational report, Saez et al1 tackle both issues by showing that it is possible to both enhance host HSC mobilization and facilitate donor hematopoietic engraftment by targeting a single molecular pathway: bone marrow stromal-cell production of heparan sulfate proteoglycans (HSPGs) (see figure). They first demonstrate this effect using an elegant chimeric mouse model, in which bone marrow hematopoietic cells are phenotypically normal but in which bone marrow stromal cells cease to produce HSPGs after deletion of floxed alleles for the glycosyltransferase gene Ext1. This Ext1 deletion is driven by Cre-recombinase expressed only in stromal cells capable of Mx1 promoter induction by poly(I):poly(C). This group’s previous work demonstrated that such Mx1+ stromal cells are osteolineage progenitors within the HSC niche that fulfill many nosologic criteria of mesenchymal stem cells.4 Using this model, Saez et al demonstrate that stromal-cell HSPG deficiency markedly reduces bone marrow HSC and progenitor populations, with reciprocal increases in functional HSC mobilization to peripheral blood and spleen.1
Translating these exciting results into a clinically applicable strategy, Saez et al then demonstrate that blocking HSPG function by reversible, competitive inhibition with heparin enhances G-CSF–induced HSC peripheral blood mobilization in wild-type mice. Compared with HSC mobilized with G-CSF alone, HSC mobilized by G-CSF plus heparin displayed a remarkably distinct transcriptome, correlating with faster neutrophil/platelet recovery times and increased durability of donor engraftment posttransplantation. Concluding their study with another provocative finding, Saez et al demonstrate that the HSC egress from bone marrow induced by Mx1+ osteolineage cell Ext1 deficiency clears sufficient “niche space” to allow sustained, cell dose–dependent donor HSC engraftment without chemotherapy or radiation.
HSPGs are a ubiquitous family of membrane-bound or secreted glycoproteins with the shared feature of possessing at least 1 heparan sulfate chain. Ext1 encodes part of the glycosyltransferase heparan sulfate copolymerase required for early stages of heparan sulfate biosynthesis5 ; thus deletion of Ext1 renders cells irreversibly deficient in HSPG production. In contrast, heparan sulfate mimetics (including heparin) mediate reversible loss of HSPG function by competitive inhibition of ligand-binding sites. HSPGs facilitate stem cell niche maintenance in several ways.5 First, HSPGs are critical components of extracellular matrix scaffolds needed for niche-cell migration. They also bind chemokines, growth factors, and other morphogens, enabling formation of gradients that control directionality of cell chemotaxis and differentiation within the niche. Finally, HSPGs serve as ligands for adhesion molecules including integrins and selectins,6 and thus HSPGs may mediate direct adhesion interactions within the bone marrow niche that govern HSC distribution and function.7
The work of Saez et al will undoubtedly inspire future investigations into identifying the specific stromal cell–derived HSPGs and their functions that, under homeostatic conditions, prevent bone marrow HSC egress caused by Ext1 deficiency or heparin treatment. The observation that VCAM-1 protein expression is decreased on Mx1+ osteolineage cells, and that VCAM-1 inhibition also enhances G-CSF–induced HSC mobilization in wild-type but not Ext1-deficient mice,1 suggests that HSPG inhibition may induce HSC mobilization by blocking adhesion between VCAM-1–expressing Mx1+ osteolineage cells and HSCs expressing VLA-4 integrin.8 However, this hypothesis is somewhat counter to recent evidence suggesting that mature and immature osteolineage cells mainly serve indirect roles within the HSC niche during homeostasis, with most direct HSC interactions mediated by perivascular and endothelial cells.7 Alternatively, inhibition of soluble HSPGs normally released by Mx1+ osteolineage cells might disrupt chemokine and growth-factor gradients in the niche, although the identity of such HSPGs is not known. Blockade of adhesion molecule–associated HSPGs on systemic Mx1+ vascular endothelial cells could also contribute to increased HSC mobilization yields by preventing extravasation and return of circulating HSCs back to the marrow space.6
From a clinical perspective, it is evident how mobilization strategies using heparan sulfate inhibition could soon help overcome stem cell dose barriers to successful application of allogeneic haploidentical transplantation and autologous stem cell/gene therapy for nonmalignant hematologic diseases. In fact, a brief report from a decade ago indicated that heparin use as an anticoagulant during leukopheresis increased CD34+ cell yields.9 Saez et al now provide a clear mechanistic explanation for this phenomenon.
In contrast, future studies will need to determine clinical applicability of the observation that Mx1+ stromal-cell Ext1 deficiency enables sustained donor HSC engraftment. In particular, whether reversible competitive HSPG inhibition using heparin or other mimetics would achieve the degree of engraftment seen with transgenic HSPG deficiency needs to be determined. Interestingly, the syngeneic transplant model of Saez et al is analogous to the transplantation of nearly syngeneic autologous HSCs used in gene therapy approaches. In fact, using reversible HSPG inhibition with minimally toxic heparin sulfate mimetics to facilitate engraftment might be ideal for this setting, because it would allow repeated cell infusions without significant conditioning toxicity in the event of suboptimal long-term gene correction from initial infusion.2 HSPG inhibition alone may, however, have limited applicability to haploidentical transplantation, where allogenicity is the major engraftment barrier, likely necessitating the addition of cytotoxic agents for immune suppression.3 Studies examining how the “niche-clearing” efficacy of HSPG inhibition is influenced by the disruption of stromal-cell populations and homeostatic growth factor/chemokine gradients caused by cytotoxic conditioning10 would help clarify the utility of this strategy in the allogeneic transplantation setting.
Conflict-of-interest disclosure: The author declares no competing financial interests.