FLT3 internal tandem duplications (FLT3/ITDs) in the juxtamembrane domain are found in approximately 25% of acute myeloid leukemia (AML) patients, ranging in size from 3 to hundreds of nucleotides. We examined whether the sizes of FLT3/ITDs were associated with clinical outcomes in 151 AML patients enrolled in Southwest Oncology Group studies: S9333 and S9500. FLT3/ITDs were identified in 32% of patients (median ITD size = 39 nucleotides; range, 15-153 nucleotides). The CR rates were 35%, 67%, and 52% for patients with large (≥ 40), small (< 40), and no ITDs, respectively (P = .19). Increasing ITD size was associated with decreasing OS (estimated 5-year OS: large = 13%, small = 26%, and no ITD = 21%, P = .072) and RFS (estimated 5-year RFS: large = 13%, small = 27%, and no ITD = 34%, P = .017). These studies suggest that ITD size may have prognostic significance.
Introduction
Approximately 25% of adult patients with acute myeloid leukemia (AML) harbor internal tandem duplications (ITDs) within the juxtamembrane (JM) domain of the FLT3 gene.1-5 FLT3/ITDs cause JM structural changes that disrupt the autoinhibitory conformation of the receptor,6 promoting constitutive activation of the receptor and downstream effectors.7-13 Although FLT3/ITDs are associated with a worse prognosis,1-5,14-18 subgroup analyses have further risk-stratified FLT3/ITD+ patients based upon mutant-wild-type allelic ratios and genetic expression patterns, suggesting that biologic and clinical differences exist between the various FLT3/ITDs.4,18,19 The sizes of FLT3/ITDs vary dramatically,14 creating a large array of different insertions within the JM domain. Previous studies have not investigated the prognostic significance of ITD size in AML1-5,15-18 ; therefore, we examined whether the sizes of FLT3/ITDs were associated with clinical outcome.
Study design
Patients and samples
Diagnostic bone marrow or peripheral blood samples from patients enrolled in Southwest Oncology Group (SWOG) studies S9333 or S9500 were obtained with approval of the Fred Hutchinson Cancer Research Center's institutional review board for the proposed studies. Informed consent was provided according to the Declaration of Helsinki. S9333 was a randomized trial comparing induction therapy with standard cytosine arabinoside (Ara-C) and daunorubicin (AD) with mitoxantrone and etoposide (ME) in AML patients aged 56 years or older.20 Because the ME regimen had an inferior survival, only the 161 subjects receiving AD induction regimens were eligible for our retrospective FLT3 analyses.20 Phase 2 study S9500 examined the efficacy of induction with AD followed by high-dose Ara-C on days 8 to 10 in 95 AML patients aged 55 years or younger.21 Neither study enrolled patients with acute promyelocytic leukemia. Although the prognostic effect of FLT3/ITDs may be most significant in patients with normal karyotypes,3 patients with chromosomal abnormalities were also included to assess whether ITD size or its effect differed according to karyotype. Diagnostic material was available for 93 and 67 patients from S9333 and S9500, respectively.
Analysis for FLT3/ITDs
Polymerase chain reaction amplification of genomic DNA and single-stranded conformation polymorphism analysis (PCR/SSCP) was used to screen for FLT3/ITDs in exons 14 and 15.5,14 Mutant allelic ratios were estimated for all FLT3/ITD+ patients using GeneScan analyses (GeneScan, Foster City, CA) and were adjusted for blast percentage,22 which was determined by examining the Ficolled biorepository specimens in all but 7 of the samples. For the other 7 samples, the blast percentage was obtained from prestudy data provided by the treating institution. ITDs were confirmed by sequencing.5
Statistical methods
Cofactors for statistical analyses of outcome included mutation status, age, sex, race, white blood cell (WBC) count, marrow and peripheral blast percentages at study entry, FAB classification, and AML onset (de novo vs secondary, known only for S9333 patients). Centrally reviewed cytogenetic data, as previously described, were also included.23 Associations between continuous variables were measured by Spearman rank order correlation coefficient (R). Comparisons between groups of patients were based on Wilcoxon rank sum test for continuous variables, and on logistic and proportional hazards regression analyses for dichotomous and time-to-event data, respectively. Outcomes analyzed included complete response (CR) and resistant disease (RD). Overall survival (OS) was measured from date of study entry until death from any cause. For patients who achieved a CR, relapse-free survival (RFS) was measured from the date that CR was achieved until relapse or death from any cause, with observation censored at the date of last contact for patients known alive without report of relapse. In analyses of treatment outcomes, ITD size was treated as a continuous variable with value 0 for FLT3/ITD- patients. Since studies S9500 and S9333 investigated somewhat different chemotherapy regimens in patients with different age ranges, all analyses of treatment outcomes were adjusted for differences between studies. Statistical significance was represented by 2-sided P values.
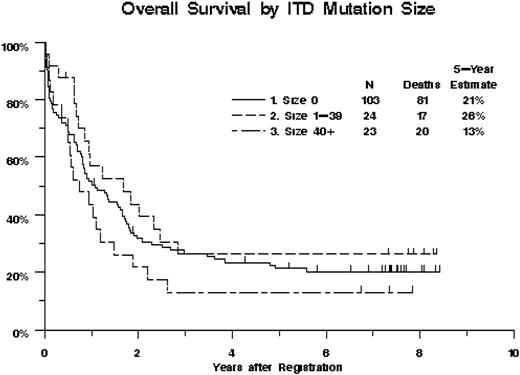
Overall survival of 151 AML patients, by FLT3/ITD mutation status and ITD size. Kaplan-Meier estimates are shown. Tick marks indicate censored observations. Overall survival decreased with increasing FLT3/ITD size (P = .072, treating ITD size as a continuous variable with size = 0 for ITD- patients).
Overall survival of 151 AML patients, by FLT3/ITD mutation status and ITD size. Kaplan-Meier estimates are shown. Tick marks indicate censored observations. Overall survival decreased with increasing FLT3/ITD size (P = .072, treating ITD size as a continuous variable with size = 0 for ITD- patients).
Results and discussion
Cryopreserved diagnostic samples were available from 160 (63%) of the 256 AML patients enrolled on study S9500 or the AD arm of S9333. The included patients had significantly (P < .001) higher WBC count (median of 31.9 × 109/L versus 4.1 × 109/L), peripheral blast percentage (46% vs 8%), and marrow blast percentage (73% vs 58%) than the 96 patients without available material. The 2 groups did not differ significantly with respect to CR rate (P = .55), RD rate (P = .51), or OS (P = .44), although the included patients had somewhat better RFS (P = .088).
Diagnostic samples of 151 patients (94%) were successfully screened for FLT3/ITDs using PCR/SSCP analyses.14 FLT3/ITDs were found in 48 patients (32%; 95% confidence interval [CI], 24%-40%). ITDs were associated with higher WBC count (median of 40.5 × 109/L versus 28.4 × 109/L; P = .008) and higher marrow blast percentage (median of 79% versus 70%; P = .003), and were more common in patients with normal karyotypes (frequency, 43% vs 8%; P < .001). The CR (P = .41) and RD (P = .97) rates were not significantly different between patients with or without FLT3/ITDs. OS was somewhat poorer for FLT3/ITD+ patients (estimated hazard ratio [HR], 1.35; CI, 0.90-2.02; P = .16) compared with FLT3/ITD- patients, as was RFS (HR = 1.70; CI, 0.93-3.08; P = .092). These results are consistent with previous studies, which have demonstrated that FLT3/ITDs are a poor prognostic factor, especially for RFS.1-4,15,17
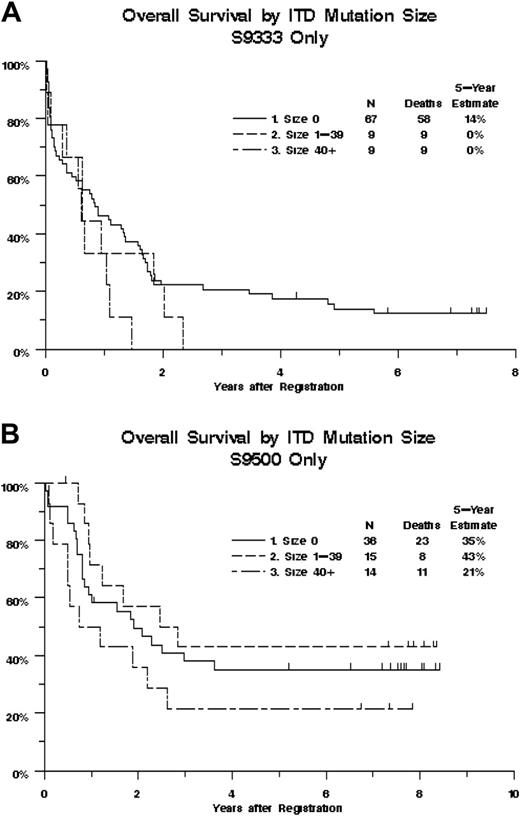
We next examined whether ITD size had an impact on clinical outcomes. ITD size could be determined by sequencing the available material for 47 of 48 FLT3/ITD+ patients. Five (10.6%) of these 47 had evidence of 2 ITDs by GeneScan. In all 5 cases, the larger ITD was chosen for the size analyses because the larger ITD displayed the higher allelic ratio. The size of the ITDs ranged from 15 to 153 nucleotides (median, 39 nucleotides). FLT3/ITD size was not significantly correlated with age (R = -0.06, P = .70), WBC count (R = 0.17, P = .24), marrow (R = -0.06, P = .70) or peripheral (R = 0.19, P = .21) blast counts, or DNA ITD/WT ratio (R = -0.10, P = .49; Table S1 and Figure S1, available on the Blood website; see the Supplemental Materials link at the top of the online article). Treatment outcomes were poorest for patients with large ITDs. The CR rates were 35%, 67%, and 52% for patients with large (≥ 40), small (< 40), and no ITDs, respectively (P = .19, treating ITD size as a continuous variable). Increasing ITD size was associated with decreasing OS (Figure 1, P = .072), with estimated probabilities of 5-year survival equal to 13%, 26%, and 21% for patients with large, small, and no ITDs, respectively. Likewise, RFS tended to decrease with increasing ITD size (P = .017), with estimated probabilities of 5-year RFS equal to 13%, 27%, and 34%, for patients with large, small, and no ITDs, respectively. The effect of ITD size on OS was somewhat stronger in patients from study S9333 (aged 56-84 years), with the mortality hazard increasing an estimated 1.3% for each additional nucleotide, compared with 0.3% for patients from study S9500 (aged 18-55 years); however, the difference in trends between the 2 studies was not statistically significant (P = .17, Figure 2).
Overall survival of 151 AML patients, by study, FLT3/ITD mutation status, and ITD size. Kaplan-Meier estimates are shown. Tick marks indicate censored observations. (A) Study SWOG-9333 (patients aged 56-84 years). (B) Study SWOG-9500 (patients aged 18-55 years). The effect of ITD size on overall survival did not differ significantly between the 2 studies (P = .17).
Overall survival of 151 AML patients, by study, FLT3/ITD mutation status, and ITD size. Kaplan-Meier estimates are shown. Tick marks indicate censored observations. (A) Study SWOG-9333 (patients aged 56-84 years). (B) Study SWOG-9500 (patients aged 18-55 years). The effect of ITD size on overall survival did not differ significantly between the 2 studies (P = .17).
Since a high ITD-wild-type (ITD/WT) ratio has been found to be associated with worse clinical outcomes,2-4,17,18,24 multivariable analyses were performed to determine whether the effect of ITD size on treatment outcomes was independent of ITD/WT ratio. ITD/WT ratio, which ranged from 0.05 to 30.7 (median, 0.836), was not significantly associated with treatment outcomes, although patients with higher ratios had somewhat poorer OS (P = .13) and RFS (P = .15). Moreover, the effects of ITD size on CR, OS, and RFS were independent of any effect that ITD/WT ratio may have on clinical outcomes (Table S2; Figure S2). Although these data suggest that ITD size has prognostic effects independent of ITD/WT ratio, there are currently no “standards” for determining ITD/WT ratios or adjusting this ratio for blast percentage. This makes interpreting the results of ITD/WT ratios difficult and generalizing these results to other studies potentially problematic.
Studies have discovered different types of mutations (point mutations, deletions, ITDs) in the JM of FLT3,7-11 and although the crystal structure of FLT3 suggests that ITDs negate the intrinsic autoinhibitory activity of the JM domain,6 it is not clear how the highly divergent types and sizes of ITDs affect the receptor's function. One could hypothesize that small ITDs may preserve some of the intrinsic autoinhibitory function of the JM domain, whereas large ITDs completely disrupt the autoinhibitory activity of the JM. Alternatively, the size of the ITDs may play a role in determining what downstream effectors are capable of docking with the receptor. In either case, these data suggest that the large ITDs are associated with a worse overall prognosis, and other studies are needed to confirm these findings.
Prepublished online as Blood First Edition Paper, December 20, 2005; DOI 10.1182/blood-2005-08-3453.
Supported by National Institutes of Health grants (nos. K23 CA92405, CA18029, CA32102, and CA114563).
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.