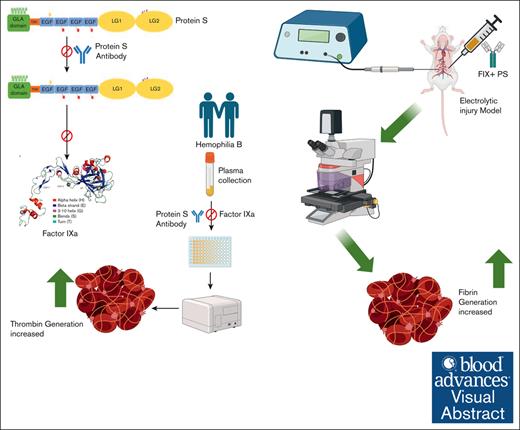
In pediatric hemophilia B (HB) plasma, thrombin generation increased with protein S (PS) antibody.
In HB mice, fibrin and platelet accumulation increased with PS antibodies.
Visual Abstract
Hemophilia B (HB) is caused by an inherited deficiency of plasma coagulation factor IX (FIX). Approximately 60% of pediatric patients with HB possess a severe form of FIX deficiency (<1% FIX activity). Treatment typically requires replacement therapy through the administration of FIX. However, exogenous FIX has a limited functional half-life, and the natural anticoagulant protein S (PS) inhibits activated FIX (FIXa). PS ultimately limits thrombin formation, which limits plasma coagulation. This regulation of FIXa activity by PS led us to test whether inhibiting PS would extend the functional half-life of FIX and thereby prolong FIX-based HB therapy. We assayed clotting times and thrombin generation to measure the efficacy of a PS antibody for increasing FIX activity in commercially obtained plasma and plasma from pediatric patients with HB. We included 11 pediatric patients who lacked additional comorbidities and coagulopathies. In vivo, we assessed thrombus formation in HB mice in the presence of the FIXa ± PS antibody. We found an accelerated rate of clotting in the presence of PS antibody. Similarly, the peak thrombin formed was significantly greater in the presence of the PS antibody, even in plasma from patients with severe HB. Furthermore, HB mice injected with PS antibody and FIX had a 4.5-fold higher accumulation of fibrin at the thrombus induction site compared with mice injected with FIX alone. Our findings imply that a PS antibody would be a valuable adjunct to increase the effectiveness of FIX replacement therapy in pediatric patients who have mild, moderate, and severe HB.
Introduction
The control of blood coagulation is an intricate process that depends on a stringently regulated balance between procoagulant and anticoagulant proteins. Any significant aberration in this balance can cause either a bleeding disorder (hemophilia) or untimely clotting (thrombosis). In the United States, about 20 000 individuals have been diagnosed with hemophilia and other related coagulopathies, and 400 infants are born each year with this group of genetic disorders.1,2
Hemophilia B (HB) is an X-linked hereditary bleeding disorder that results from a deficiency of the procoagulant protein factor IX (FIX).1,2 The severity of the HB bleeding phenotype is related to the amount of remaining functional FIX. Normal plasma levels of FIX are 50 to 150 U/dL. The FIX reference range for the pediatric population aged 1 to 12 months is 44.0 to 114.0 U/dL, and for the population aged 1 to 18 years, it is 60.0 to 116.0 U/dL (Sysmex CA-1500 Analyzer).3 Severe HB involves spontaneous, life-threatening bleeding in which the FIX levels are <1 IU/dL, usually diagnosed at birth. FIX levels of 1 to 5 IU/dL represent moderate HB, and 5 to 40 IU/dL are classified as mild HB. Moderately and mildly affected individuals may have excessive bleeding only from trauma and/or surgical procedures.
Currently, there is no cure for HB. There are effective treatments, but they require lifelong infusions of expensive drugs. The standard of care for HB management is either primary prophylaxis or secondary prophylaxis. Primary prophylaxis refers to the early initiation of clotting factor replacement therapy4,5 before the onset of the first joint bleed but no later than before a second joint bleed, ideally before the age of 3 years. Conversely, secondary prophylaxis denotes the introduction of clotting factor replacement therapy after 2 joint bleeds before any evidence of joint disease. However, when Roth et al6 studied plasma-derived FIX and recombinant FIX (rFIX), they identified limitations with rFIX regarding its pharmacokinetics, namely, a 25% to 30% lower in vivo recovery in children aged <15 years.7 Moreover, replacement therapy may cause viral infections from natural clotting factors, and, because of delays in treatment, replacement therapy may cause damage to joints, muscles, or other parts of the body. In addition, for severe HB, replacement therapies have been associated with the occurrence of FIX inhibitors at frequencies of 3% to 5%. A PedNet study in 20218 of an unselected cohort of previously untreated patients with severe HB showed a cumulative inhibitor incidence of 10.2% at 500 exposure days.
Thus, considering the limitations of the current replacement therapies, investigators need to devise a novel treatment approach for HB. Below, we present results that support a proof-of-concept for a new therapy for augmenting FIX-based treatment, namely, coadministration of anti-PS antibodies in a pediatric patient population.
Protein S (PS), a vitamin K–dependent protein, is a key negative regulator in the coagulation cascade.9 Deficiencies in PS cause disorders that range from a heightened risk of thrombosis to a potentially fatal purpura fulminans in newborns.10,11 PS has long been described as a cofactor for both activated protein C (APC) and tissue factor pathway inhibitor (TFPI).12-15 Chattopadhyay et al16 established a novel function of PS as an inhibitor of activated FIX (FIXa). PS inhibits FIXa in the presence or absence of FVIIIa. Inhibition of FIXa ultimately leads to inhibition of the intrinsic Xase (FIXa-FVIIIa) complex, thereby limiting thrombin generation and contributing to the prevention of thrombosis.16 Importantly, we established PS as an essential in vivo inhibitor of FIXa because disruption of the PS-FIXa interaction caused an increased rate of thrombus formation in mice.17
The novel discovery that PS inhibits FIXa16,18 has significance for the treatment of HB. As mentioned earlier, HB is usually treated by the administration of exogenous FIX.19 The burden and inconvenience of frequent intravenous FIX infusions are major barriers to patient compliance, especially in the pediatric population.20,21 Thus, a valuable enhancement to current HB therapy would be an adjunct treatment that increases the effectiveness of a given concentration of FIXa, thereby reducing the frequency of FIX injections or increasing the time interval between FIX injections.
Theoretically, the elimination or moderation of PS inhibition of FIX would extend the functional half-life of FIX. In this regard, we have identified an anti-PS antibody as a promising means to limit PS inhibition of FIX.
Our objective in this study was to evaluate the efficacy of PS antibody as an adjunct in treatment of HB. We focused on the pediatric population (aged 2 months-17 years) because, compared with adults, HB is more severe in younger individuals22 and because prophylaxis factor replacement therapy is the standard of care in the United States. We found that clotting rate and thrombin generation were increased in plasma supplemented with PS antibody. Similarly, in mice injected with PS antibody, fibrin accumulation at a thrombus induction site increased 4.5-fold. Based on these findings, we propose that this unique use of PS antibody is a novel approach to augment hemostasis and improve the quality of life for children with HB.
Materials, patients, and methods
FIX-deficient plasma was purchased from Hematologic Technologies, Inc (HTI; Essex Junction, VT) and George King Bio-Medical, Inc (Overland Park, KS). In addition, Paul Riley (Diagnostica Stago, Parsippany, NJ) provided FIX-deficient plasma. FIX-deficient plasma from HTI was prepared by immunodepletion, whereas FIX-deficient plasma from Diagnostic Stago and George King Bio-Medical was prepared from FIX-deficient individuals. We purchased PS antibodies from HTI, R&D Systems (Minneapolis, MN), and Abcam (Cambridge, MA). Antibodies can differ significantly in their inhibitory properties; thus, we assessed PS antibodies from different commercial vendors to determine whether a range existed for blocking PS function. FIX, FIXa, and corn trypsin inhibitor were obtained from HTI. Kontact reagent for the modified activated partial thromboplastin time (aPTT) assay was obtained from ThermoFisher Scientific (Waltham, MA). Thrombin generation assay reagent technothrombin thrombin generation assay reagent B (containing low amounts of tissue factor and phospholipid) and substrate were purchased from DiaPharma (West Chester, OH). The TFPI antibody was a kind gift from G. J. Broze Jr.’s laboratory. All other chemicals were American Chemical Society reagent grade.
Study participants
The study cohort (n = 11) included 6 Caucasian and 5 African American pediatric males, median age of 10 years, with a range of 2 months to 17 years. Six participants had severe FIX deficiency, 1 had moderate deficiency, and 4 had mild deficiencies. All patients had been treated with a rFIX replacement product. Three of the 4 patients with mild deficiency received on-demand therapy with a standard half-life product (SHL). The fourth patient with mild deficiency had not begun any factor replacement therapy at the time of study enrollment. All patients with severe FIX deficiency, along with the sole patient with moderate FIX deficiency, were managed with prophylactic therapy. Of the 7 patients who received prophylaxis, 86% (n = 6) received an extended half-life product (EHL), whereas the remaining patient was treated with the SHL product. Our cohort encompassed various technologies to extend the FIX product half-life. Two patients (18%) were treated with a PEGylated factor replacement product, and 4 individuals (36%) received products with albumin fusion protein technology (Table 1). The institutional review boards of Children’s Hospital and Tulane University, New Orleans, LA, approved the enrollment process. Each parent/guardian and child provided informed consent and assent (where applicable), respectively.
Demographic characteristics of study participants
| . | Age . | Race . | Severity . | Product . | Prophylaxis . |
|---|---|---|---|---|---|
| Patient 1 | 3 y | African American | Severe | EHL | Yes |
| Patient 2 | 17 mo | African American | Severe | EHL | Yes |
| Patient 3 | 13 y | Caucasian | Mild | SHL | No |
| Patient 4 | 10 y | Caucasian | Mild | SHL | No |
| Patient 5 | 10 y | Caucasian | Mild | SHL | No |
| Patient 6 | 2 y | Caucasian | Severe | SHL | Yes |
| Patient 7 | 2 mo | Caucasian | Mild | None | N/A |
| Patient 8 | 16 y | African American | Severe | EHL | Yes |
| Patient 9 | 17 mo | African American | Severe | EHL | Yes |
| Patient 10 | 15 y | Caucasian | Severe | EHL | Yes |
| Patient 11 | 17 y | African American | Moderate | EHL | Yes |
| . | Age . | Race . | Severity . | Product . | Prophylaxis . |
|---|---|---|---|---|---|
| Patient 1 | 3 y | African American | Severe | EHL | Yes |
| Patient 2 | 17 mo | African American | Severe | EHL | Yes |
| Patient 3 | 13 y | Caucasian | Mild | SHL | No |
| Patient 4 | 10 y | Caucasian | Mild | SHL | No |
| Patient 5 | 10 y | Caucasian | Mild | SHL | No |
| Patient 6 | 2 y | Caucasian | Severe | SHL | Yes |
| Patient 7 | 2 mo | Caucasian | Mild | None | N/A |
| Patient 8 | 16 y | African American | Severe | EHL | Yes |
| Patient 9 | 17 mo | African American | Severe | EHL | Yes |
| Patient 10 | 15 y | Caucasian | Severe | EHL | Yes |
| Patient 11 | 17 y | African American | Moderate | EHL | Yes |
Plasma samples
Blood samples were collected in sodium citrate tubes. Plasma was isolated immediately via centrifugation at 5000 rpm for 15 minutes and then frozen at −80°C until analyzed.
Modified aPTT assay
To measure the effect of PS on the intrinsic pathway of blood coagulation, an aPTT assay was used with normal pooled plasma and increasing PS antibody concentrations. We added Kontact reagent and incubated for 1 minute before CaCl2 (25 mmol/L) initiated coagulation. PS antibody (450 nM) was added to FIX-deficient plasma (40 μL) and incubated at 37°C for 15 minutes, then increasing concentrations of FIXa (0.025-0.5 nM; HTI) were added and incubated for 1 minute. A SoftMax plate reader monitored clotting at 450 nm (Molecular Devices, Sunnyvale, CA).16,18
Thrombin generation assay
Thrombin generation was measured in FIX-deficient plasma in the presence of added FIX with 450 nM PS antibody, 30 nM TFPI antibody, and 450 nM PS + 30 nM TFPI antibody along with controls (saline without antibody).16,18,23 To monitor the effect of PS antibody on thrombin generation specifically by the FIXa pathway,24,25 corn trypsin inhibitor was added to suppress contribution from the contact activation pathway. Commercial or patient plasma was mixed with 450 nM PS antibody (ab 133730, Abcam) and 4 μL corn trypsin inhibitor (6 μg mL−1 final concentration), followed by incubation at 37°C for 15 minutes in a 96-well microplate. A physiological concentration of 90 nM FIX (HTI) was used. Buffer (50 mM Tris, 175 mM NaCl, 0.1% PEG-8000, pH 7.4) was added to each well to a total volume of 120 μL. The reaction was activated with 10 μL Technothrombin reagent B (DiaPharma) that contained a low concentration (0.4 pmol/L) of tissue factor and 8 μmol/L phospholipid.25,26 The reaction was initiated with the fluorogenic substrate Z-Gly-Gly-Arg-AMC (DiaPharma). Fluorescence was measured on a SpectraMax plate reader with emission and excitation wavelengths of 360 and 460 nm. Both the modified aPTT and thrombin generation assays were performed with 40 μL of plasma for a total reaction volume of 120 μL.
Thrombus formation in HB mice infused with FIX ± PS antibody
Under pentobarbital anesthesia (50 mg/kg, i.p., with supplement as needed), young HB male mice were divided into 3 groups (n = 4 per group) based on pretreatment intravenous injections before thrombus induction: (1) 50 IU/kg of rFIX (BeneFIX, Pfizer) followed by a saline injection; (2) the same FIX solution, followed by an injection of PS antibody (1.3 ug/kg 5 minutes later); and (3) 2 control series with saline only. We used a mouse monoclonal antibody from R&D (MAB4976), which has no reactivity with humans in an ELISA assay. All injections were in a 50 μL volume via the left saphenous vein. Ten minutes later, Alexa Fluor-647–labeled anti-fibrin (59D8 monoclonal antibody27) and rhodamine 6G (2 mg/kg) were injected into the right jugular vein. The right femoral vein was then exposed by skin incision and retraction, and an electrolytic injury was induced on the vein surface with 1.5 volts of anodal current delivered for 30 seconds, as described previously.28 The site of injury was subsequently imaged for 60 minutes with 532 and 650 nm laser light and emission capture, as described previously.28 Offline quantitation of fluorescence was performed with ImageJ software. Variance analysis evaluated in vivo thrombosis data, with post hoc between-group comparisons performed using the Tukey HSD test (P = .05). All experiments with mice were approved by the institutional review boards.
Statistical analysis
We used appropriate statistics for multigroup analysis, and the data were appropriate for parametric analysis. We used a log-linear model for testing the effect of FIXa dose (concentration in nM) and the effect of adding PS antibody on clotting time (in seconds) in the control HB plasma. Upon visual exploration of the data, we found a linear relationship between the clotting time and the log-dose. However, we used log(0.001 + dose) as a predictor to avoid problems with 0 dose. The final model included the response variable clotting time and the predictor variables log(0.001 + dose) and presence/absence of PS antibody. We used one-way analysis of variance for the control samples’ data.
Results
Effect of PS antibody on clotting time
We used a partial thromboplastin time (aPTT) assay to measure clotting time in commercial and patient FIX-deficient plasma, supplemented with FIXa. The addition of PS antibody to FIXa-supplemented commercial plasma decreased clotting time (Figure 1A for HTI plasma and Figure 1B for Diagnostic Stago plasma). Similarly, PS antibody decreased the clotting times of the patient plasmas (Figure 2). The type of factor replacement that the patients received did not have any significant effect on the results. Findings were reproducible regardless of whether patients received on-demand therapy with an SHL product or prophylactic treatment with either SHL or EHL products (Table 2).
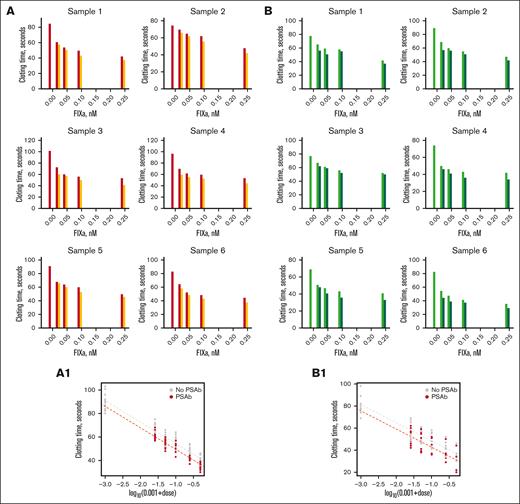
Clotting times of commercial FIX deficient plasmas measured by a partial thromboplastin time assay. Partial thromboplastin time (aPTT) assay to determine clotting time of FIX-deficient commercial plasma (A: HTI plasma, B: Stago plasma) with different concentrations of FIXa in the presence and absence of PS antibody. (A) Clotting time of different samples of HTI FIX-deficient plasma. The plasma was incubated with different concentrations of FIXa, and clotting times were measured in the absence (red bar) and presence (orange bar) of PS antibody (450 nM). (B) The same experiment with Stago plasma. Light green and dark green bars represent the clotting time without and with PS antibody, respectively. (A1,B1) The bottom panels in panels A and B show data presented in each panel in a single scatterplot of log(0.001 + dose) vs clotting time for all samples. The dashed lines represent the fitting by the log-linear model. The y-axis scales vary between graphs for greater visibility of results.
Clotting times of commercial FIX deficient plasmas measured by a partial thromboplastin time assay. Partial thromboplastin time (aPTT) assay to determine clotting time of FIX-deficient commercial plasma (A: HTI plasma, B: Stago plasma) with different concentrations of FIXa in the presence and absence of PS antibody. (A) Clotting time of different samples of HTI FIX-deficient plasma. The plasma was incubated with different concentrations of FIXa, and clotting times were measured in the absence (red bar) and presence (orange bar) of PS antibody (450 nM). (B) The same experiment with Stago plasma. Light green and dark green bars represent the clotting time without and with PS antibody, respectively. (A1,B1) The bottom panels in panels A and B show data presented in each panel in a single scatterplot of log(0.001 + dose) vs clotting time for all samples. The dashed lines represent the fitting by the log-linear model. The y-axis scales vary between graphs for greater visibility of results.
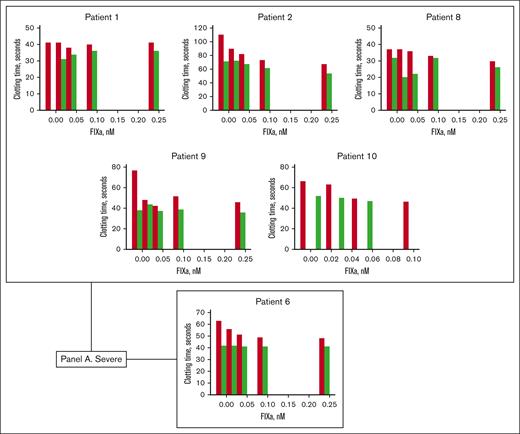
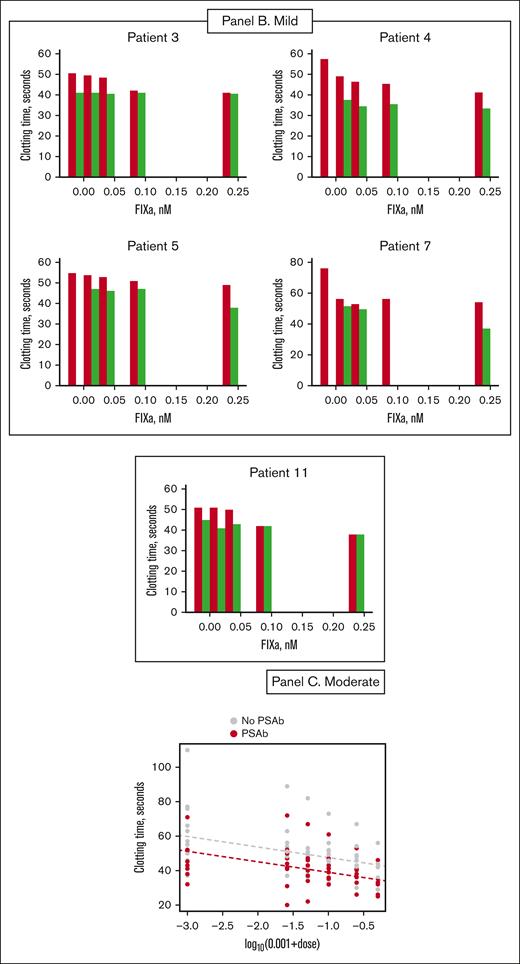
Clotting times of patient plasmas (patients 1-11) measured by a partial thromboplastin time assay. (A) Severe hemophilia B; (B) mild hemophilia B; (C) moderate hemophilia B. Different concentrations of FIXa were added to patient plasmas (1-11), and the clotting times were measured in the absence (red bar) and presence (green bar) of PS antibody. Different panels were created based on the severity of FIX deficiency. Patient samples 1, 2, 8, 9, and 10 are from patients with severe HB who were administered EHL. Patient 6 had severe HB and was treated with SHL. Patient samples 3, 4, 5, and 7 were from patients with mild HB treated with SHL, and patient 11 had moderate hemophilia, treated with an EHL product.
Clotting times of patient plasmas (patients 1-11) measured by a partial thromboplastin time assay. (A) Severe hemophilia B; (B) mild hemophilia B; (C) moderate hemophilia B. Different concentrations of FIXa were added to patient plasmas (1-11), and the clotting times were measured in the absence (red bar) and presence (green bar) of PS antibody. Different panels were created based on the severity of FIX deficiency. Patient samples 1, 2, 8, 9, and 10 are from patients with severe HB who were administered EHL. Patient 6 had severe HB and was treated with SHL. Patient samples 3, 4, 5, and 7 were from patients with mild HB treated with SHL, and patient 11 had moderate hemophilia, treated with an EHL product.
Thrombin generation with addition of 450 nM PS antibody according to the severity of the disease
| Patient . | Control, nM . | +PS Ab, nM . | Total increase, nM . |
|---|---|---|---|
| Group A (severe, on EHL product) | |||
| 1 | 349 | 670 | 321 |
| 2 | 243 | 397 | 154 |
| 8 | 324 | 390 | 66 |
| 9 | 342 | 425 | 83 |
| 10 | 284 | 474 | 190 |
| Average | 308.4 | 471.2 | 162.8 |
| Group B (severe, on SHL product) | |||
| 6 | 226 | 385 | 159 |
| Group C (moderate) | |||
| 11∗ | 492 | 507 | 15 |
| Group D (mild) | |||
| 3 | 367 | 470 | 103 |
| 4 | 402 | 454 | 52 |
| 5 | 339 | 422 | 83 |
| 7 | 517 | 665 | 148 |
| Average | 406 | 503 | 97 |
| Patient . | Control, nM . | +PS Ab, nM . | Total increase, nM . |
|---|---|---|---|
| Group A (severe, on EHL product) | |||
| 1 | 349 | 670 | 321 |
| 2 | 243 | 397 | 154 |
| 8 | 324 | 390 | 66 |
| 9 | 342 | 425 | 83 |
| 10 | 284 | 474 | 190 |
| Average | 308.4 | 471.2 | 162.8 |
| Group B (severe, on SHL product) | |||
| 6 | 226 | 385 | 159 |
| Group C (moderate) | |||
| 11∗ | 492 | 507 | 15 |
| Group D (mild) | |||
| 3 | 367 | 470 | 103 |
| 4 | 402 | 454 | 52 |
| 5 | 339 | 422 | 83 |
| 7 | 517 | 665 | 148 |
| Average | 406 | 503 | 97 |
The single patient with moderate FIX deficiency had an unusually high baseline peak thrombin level. The addition of PS antibody produced a 15-nM increase in thrombin generation.
For substantiation, we performed the clotting assay with several commercial PS antibodies (see “Materials, patients, and methods” for detailed antibody description); the results were alike. Because different antibodies produced similar results, we used only antibodies from Hematologic Technologies for the experiments shown in Figure 1. The results were statistically significant based on a log-linear model with clot time as the response and log(0.001 + dose) as the predictor (Figure 1A1,B1), whereas adjusting for the effect of the presence or absence of PS antibody. The effects of both the dose and PS antibody were statistically significant (P < 10−5) for dose and for PS antibody (P = .001).
In Figure 2, the patient clotting assay data are organized according to the severity of HB (5 severely deficient patients who received an EHL product are grouped together, and 1 severe patient who received an SHL product is placed below in a separate panel). Therefore, the severe patient data are arranged in Figure 2A, the mild patient data are displayed in Figure 2B, and the moderate patient data are in Figure 2C. In the presence of varying concentrations of FIXa, clotting times were consistently shortened with the addition of PS antibody. We observed an average decrease of 10.5 seconds in plasma from severe patients who received EHL products and a shortening of 12 seconds in plasma from the severe patient treated with an SHL product. The average decrease in clotting times for moderate and mild patient plasmas was 4.6 and 10 seconds, respectively.
Effect of PS antibody on thrombin generation
We performed thrombin generation assays to independently verify the effect of PS antibody in the clotting assays; thrombin generation is the most physiological measure of coagulation, hence a better measure of blood coagulation ex vivo. Like the aPTT assay, thrombin generation showed positive effects with PS antibody. Using various commercial plasmas, we found that thrombin generation increased with the addition of PS antibody (Figure 3A). Each data set was an average of 3 independent reactions.
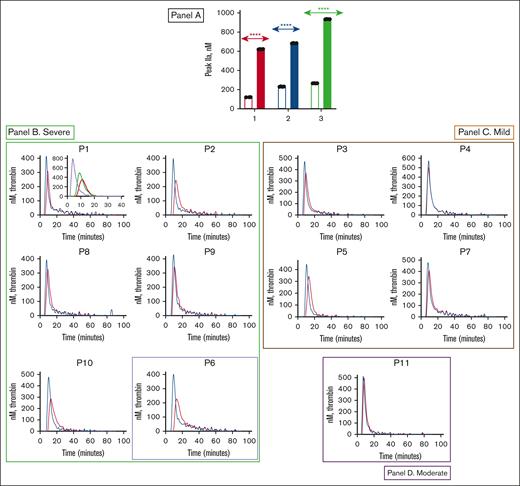
Effect of PS antibody on plasma thrombin generation. (A) Effect of PS antibody on plasma thrombin generation in 3 FIX-deficient commercial plasmas (George King, Stago, and HTI plasmas) represented as red (1), blue (2), and green (3) bars, respectively. The plasmas were incubated with 90 nM of FIX without (empty bar in each category) and with (filled bar in each category) a 450-nM PS antibody. Thrombin generation was measured in the presence of corn trypsin inhibitor (1 μM) and reagent B containing 0.4-pM tissue factor. The thrombin generation was initiated with a mixture of CaCl2 and the fluorogenic substrate Z-Gly-Gly-Arg-AMC. The bars represent the peak thrombin that formed. Although many patients had recently received prophylactic FIX infusions, peak thrombin still increased in the presence of PS antibody. Student uncoupled t test was performed (P < .0001) to determine the level of significance as described.34 (B-D) Effect of PS antibody on thrombin generation in patient plasmas (patients 1-11). Results are grouped according to severity of hemophilia, with panels B-D representing severe, mild, and moderate hemophilia, respectively. Patient 6, the single severe patient managed with an SHL product, is denoted by highlighting with a purple box. Patient plasmas were incubated with (blue curve) and without (red curve) PS antibody. Panel B P1 inset plot: effect of control (red - no TFPI antibody or PS antibody), TFPI antibody (green), and TFPI antibody + PS antibody (purple) on plasma thrombin generation.
Effect of PS antibody on plasma thrombin generation. (A) Effect of PS antibody on plasma thrombin generation in 3 FIX-deficient commercial plasmas (George King, Stago, and HTI plasmas) represented as red (1), blue (2), and green (3) bars, respectively. The plasmas were incubated with 90 nM of FIX without (empty bar in each category) and with (filled bar in each category) a 450-nM PS antibody. Thrombin generation was measured in the presence of corn trypsin inhibitor (1 μM) and reagent B containing 0.4-pM tissue factor. The thrombin generation was initiated with a mixture of CaCl2 and the fluorogenic substrate Z-Gly-Gly-Arg-AMC. The bars represent the peak thrombin that formed. Although many patients had recently received prophylactic FIX infusions, peak thrombin still increased in the presence of PS antibody. Student uncoupled t test was performed (P < .0001) to determine the level of significance as described.34 (B-D) Effect of PS antibody on thrombin generation in patient plasmas (patients 1-11). Results are grouped according to severity of hemophilia, with panels B-D representing severe, mild, and moderate hemophilia, respectively. Patient 6, the single severe patient managed with an SHL product, is denoted by highlighting with a purple box. Patient plasmas were incubated with (blue curve) and without (red curve) PS antibody. Panel B P1 inset plot: effect of control (red - no TFPI antibody or PS antibody), TFPI antibody (green), and TFPI antibody + PS antibody (purple) on plasma thrombin generation.
Most importantly, we found that the addition of PS antibody to patient plasma produced an increase in thrombin generation (Figure 3B-D). We obtained the data from at least 3 experiments, and the averages of 3 different data sets are plotted for each patient sample. The patient thrombin generation assay data (Figure 3B-D) are arranged according to severity. Again, all the severe patients (patients 1, 2, 6, 8, 9, and 10) are grouped together (Figure 3B). Patient 6, the single severe patient managed with an SHL product, is denoted by a purple box. Results for mild patients are grouped in a separate panel (Figure 4C), with the moderate patient’s thrombin generation data depicted in a separate panel (Figure 4D). To simulate the true physiologic state, we used 90 nM FIX in the thrombin generation assay. Notably, although many patients had recently received prophylactic FIX infusions, peak thrombin still increased in the presence of PS antibody.
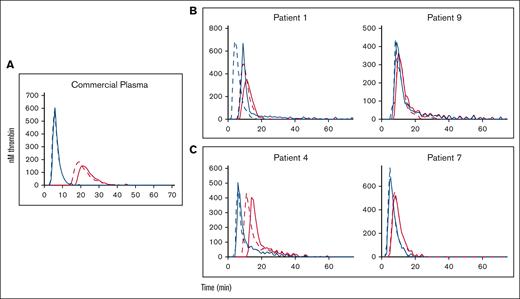
Effect of TFPI antibody on plasma thrombin generation in control and patient plasmas. Results are grouped as follows: commercial plasma (A), severe hemophilia (B), and mild hemophilia (C). Plasmas were incubated without TFPI antibody (solid red lines), with TFPI antibody (dashed red lines), with PS antibody (solid blue lines), and with TFPI + PS antibodies (dashed blue lines).
Effect of TFPI antibody on plasma thrombin generation in control and patient plasmas. Results are grouped as follows: commercial plasma (A), severe hemophilia (B), and mild hemophilia (C). Plasmas were incubated without TFPI antibody (solid red lines), with TFPI antibody (dashed red lines), with PS antibody (solid blue lines), and with TFPI + PS antibodies (dashed blue lines).
As anticipated, patients with severe FIX deficiency had a lower baseline peak thrombin (compared with patients with moderate and mild FIX deficiency). The average baseline peak thrombin in severe patients treated with an EHL vs an SHL product was ∼300 and 226 nM (Table 2), respectively. We observed a considerable increase in thrombin generation with the addition of PS antibody. Overall, we found an average increase of ∼60% (Table 2, group A) in plasma from patients with severe FIX deficiency who had been treated with EHL products. We observed an increase of ∼70% for thrombin generation in the plasma from the severe patient who was managed with an SHL product. (Table 2, group B). Patients with mild FIX deficiency had an average baseline thrombin generation of 400 nM. This baseline thrombin generation increased by ∼20% with the addition of 450 nM PS antibody (Table 2, group D).
Effect of PS antibody on thrombin generation in presence of TFPI antibody
PS acts as a cofactor for TFPI in inhibiting free factor Xa in the extrinsic coagulation pathway.15 We performed the thrombin generation assay with the addition of TFPI antibody (see “Materials, patients, and methods”) in the absence and presence of PS antibody. The addition of TFPI antibody enabled us to measure exclusively the activity of PS antibody in promoting thrombin generation, independent of effects from TFPI. We used plasma from 2 patients with severe FIX deficiency (patients 1 and 9) (Figure 4, B frame) and 2 patients with mild FIX deficiency (patients 4 and 7) (Figure 4, C frame). The average baseline thrombin generation in the severe patient plasma was ∼370 nM. The addition of the TFPI antibody caused an increase in average thrombin generation by only ∼8%. This effect was amplified with the addition of PS antibody, leading to an average increase of ∼54% of thrombin produced. A higher amount of thrombin was formed (average 460 nM) at baseline with the plasmas from the patients with mild deficiency. The average amount of thrombin generated increased only by 2% (average) with the addition of the TFPI antibody. Thrombin generation was intensified with the addition of PS antibody, yielding an average thrombin generation of 632 nM (Figure 4).
Effect of PS antibody on thrombus formation in HB mice
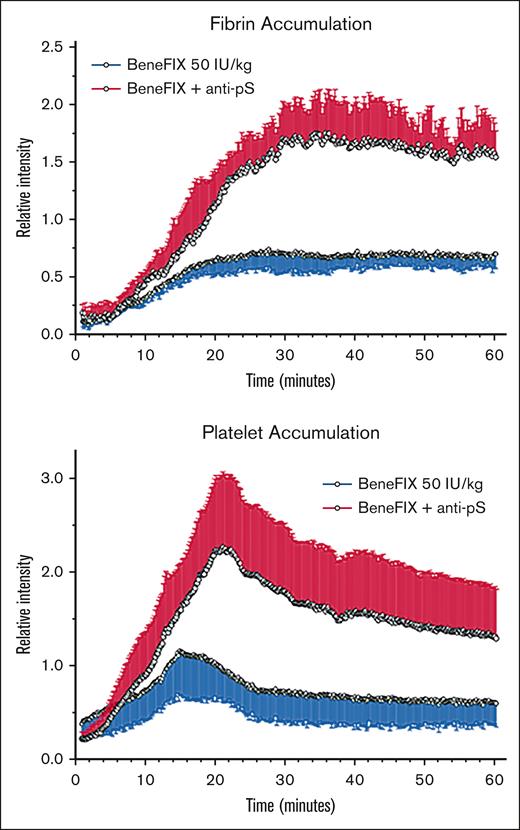
Because ex vivo assay results showed promise for using a PS antibody to boost FIX therapy, we sought similar evidence in vivo. We used a mouse femoral vein injury model to assess thrombus formation in HB mice by monitoring fibrin and platelet accumulation in the presence of the FIXa ± PS antibody (Figure 5). We delivered a 30-second electrolytic injury to the vein surface, resulting in a thrombus that peaked in growth at 20 to 60 minutes (see “Materials, patients, and methods”). To block PS and FIXa interaction, we inhibited PS activity with PS antibody injected prior to thrombus induction and then assessed disruption of that interaction (Figure 5). We undertook statistical analysis of quantitative data for the accumulation of fibrin (Figure 5A) and platelets (Figure 5B) at 15, 30, and 60 minutes of thrombus development. We observed that fibrin accumulation was not significantly elevated at 15 minutes for rFIX alone vs rFIX plus PS antibody (P > .05), but both groups had increased 15-minute fibrin accumulation in comparison to the saline-only controls (P < .05). At the later times of 30 and 60 minutes, rFIX plus PS antibody showed increased fibrin (4.5-fold higher) compared with treatment with rFIX alone and saline-only controls (P < .002); fibrin in the rFIX alone group was also significantly elevated at these times compared with the saline-only controls (P < .05). Platelet accumulation (Figure 5B) also showed significant elevations for the rFIX plus PS antibody group compared with the other groups (P < .01), but only achieved statistical significance at the 30-minute time point for rFIX alone vs the saline controls (P < .05). These findings reconfirmed16 PS as a principal in vivo inhibitor of FIXa, and the results provided compelling support for inhibiting PS to enhance FIX therapy for HB. We also captured these inspiring data in supplemental Videos 1-2.
Relative intensities of thrombus-targeting fluorophores in femoral veins of FIX-deficient mice after electrolytic injury, showing fibrin (top graph) and platelet (bottom graph) accumulation during 60 minutes of thrombogenesis. Data points are from image analysis of video frames taken every 10 seconds, showing means and standard deviations (error bars), normalized for mouse weight and amount of preinjected fluorophore. Red lines show accumulation for mice preinjected with rFIX and PS antibody, and blue lines are for mice preinjected with rFIX only.
Relative intensities of thrombus-targeting fluorophores in femoral veins of FIX-deficient mice after electrolytic injury, showing fibrin (top graph) and platelet (bottom graph) accumulation during 60 minutes of thrombogenesis. Data points are from image analysis of video frames taken every 10 seconds, showing means and standard deviations (error bars), normalized for mouse weight and amount of preinjected fluorophore. Red lines show accumulation for mice preinjected with rFIX and PS antibody, and blue lines are for mice preinjected with rFIX only.
Discussion
Our results indicate that PS antibody can function as an adjunct therapy in the management of HB. This conclusion is based on the consistent decrease in clotting time and the increase in thrombin generation (Figures 2 and 3, panels B-D) when we added PS antibody to FIX-deficient plasma. We used different commercially sourced plasma in addition to actual FIX-deficient patient plasma to ensure reproducibility. We found the most significant increases in thrombin generation with plasmas from patients who had severe HB. Patients were affected similarly by PS antibody regardless of having received EHL or SHL, a paramount finding because this patient group experiences the most pronounced bleeding complications. The enhanced thrombin generation produced by PS antibody occurred even though the patients had recently received FIX replacement therapy. Moreover, the type of rFIX product (EHL or SHL) that was used for replacement therapy did not affect the action of PS antibody in upregulating FIX activity (Figure 3). Our findings clearly showed that TFPI antibody contributed to a much lesser extent than PS antibody (Figure 4) to increase thrombin generation in commercially sourced plasma and those from patients with severe and moderate HB. These data agreed with our previous work, in which we showed that PS inhibited thrombin generation (in PS-deficient plasma) by ∼90%.16 In that study, we initiated thrombin generation with a low tissue factor concentration to direct thrombin generation predominantly by the FIXa-FVIIIa pathway.16,17,24,25 We found that thrombin generation was essentially independent of TFPI (only a 10% increase in thrombin generation in the presence of TFPI antibody).16
We extended our in vitro studies to FIX-deficient mice. In a venous thrombosis model,28 we induced the formation of a thrombus that peaked in growth at 20-60 minutes (see “Materials, patients, and methods”). The inclusion of PS antibody caused a significant increase in the rate of thrombus formation in this mouse model (Figure 5; supplemental Videos 1-2). These findings substantiated an important regulatory function of PS as an in vivo inhibitor of FIXa. Our in vivo data clearly indicate that PS antibody prevented PS from inhibiting FIXa, thereby enhancing fibrin formation in HB mice. Previously, we showed that PS inhibition of FIXa in plasma was completely independent of APC.16 Thus, it was expected that PS antibody would block PS from inhibiting FIXa and enhance fibrin formation in HB mice.
PS also acts as a cofactor of TFPI,15,29 but in inhibiting FXa in vivo, PS enhances only platelet and endothelial cell TFPI, not cell surface–associated TFPI.30 Moreover, we found that the addition of TFPI antibody to FIX-deficient plasma produced only a small increase in thrombin generation (Figure 4). A marginal increase in thrombin generation in the presence of TFPI antibody indicated that TFPI had only a slight effect on FXa generation in the initial phase of thrombin generation (lags shown in Figure 3A). Under these conditions, the rate of FXa generation was slow, and TFPI at least partly regulated thrombin generation (≈10% more thrombin generation compared with that in the absence of TFPI antibody).
Clinical and histological findings suggest that HB may be less severe than hemophilia A (HA).31 However, the perceived differences between HA and HB are not evident in children, for whom the severity of bleeding is similar during the early stage of treatment.22 Moreover, treating children with rFIX has a distinct disadvantage with respect to its pharmacokinetics. Children aged <15 years have a 25% to 30% lower in vivo recovery rate after rFIX administration.6,32 Thus, for our novel treatment approach, we were especially interested in testing plasma from young children.
Significant progress has been achieved in the development of therapeutic agents to treat HB, from EHL products to gene therapy.33,34 Nevertheless, treatment is still challenging because of the occurrence of alloantibodies that render the standard replacement therapy with FIX concentrates ineffective and expose patients to significant morbidity and mortality.5 Moreover, there are limitations to long-lasting FIX (EHL) fused with albumin because data from EHL evaluations are not derived from large numbers of patients, and there is a lack of information about the location, severity, treatment, outcomes of bleeding events, and patient adherence (for which true and accurate measurements are difficult to obtain, particularly in retrospective studies).35 Furthermore, in the analysis of EHL, investigators considered only patients who had switched to rIX-FP (rFIX fused with human albumin) from a prior replacement product.35,36 Although our study investigated inhibiting the natural anticoagulant mechanism of PS, in a similar study, Polderdijk et al targeted APC, a serine protease, to restore hemostasis. In a hemophilia mouse model, the investigators used an APC-specific serpin (serine protease inhibitor) that blocked the protein C anticoagulant pathway in the absence of the intrinsic Xase complex. In vivo hemostasis was achieved, and although Polderdijk et al have recently created a mammalian-expressed serpin PC that was advantageous in HA mice (unpublished), there has not been any effort toward finding efficacy in HB.37 Gene therapy is also a treatment area with recent advancements. Fitusiran, a small interfering RNA that silences the gene for antithrombin, was thought to show promise for hemostasis improvement in patients with HA/HB. However, in an open-label phase 3 ATLAS-PPX study, Pasi et al observed that alanine transaminase/aspartate transaminase levels increased to more than 3 times normal in 25.4% of patients, and 3% of patients who used fitusiran experienced thromboembolic events.38
To mitigate the thrombotic risk while using fitusiran, antithrombin levels were targeted between 15% and 35%.39
Concizumab, a humanized recombinant monoclonal antibody directed against the TFPI, was used as a subcutaneous prophylactic treatment for patients with HA/HB who did or did not have inhibitors. In phase 2 trials, concizumab was well tolerated, and a favorable safety profile was shown, with no deaths or thromboembolic events and no adverse events.40 However, the phase 3 trial was paused because of the occurrence of 2 arterial and 3 venous thrombotic events in 3 patients who had HA or HB inhibitors. Thus, risk mitigation plans were developed, including updates to the concizumab dosing regimen and guidelines for the concomitant use of hemostatic agents in the management of bleeding episodes during concizumab prophylaxis. To prevent thrombotic events, standardized techniques to monitor hemostasis in patients who received nonfactor products will assist clinical management in these circumstances.
Although our work strongly suggests that PS antibodies would be an effective therapeutic agent, antibodies are not without shortcomings, particularly the risk of antigenicity, thrombogenicity, and hypersensitivity. Strategies to avoid or minimize the appearance of infusion-related reactions depend on the antibody, type of patient, and reaction (premedication, slowing infusion rates, infusion interruption, or desensitization). Moreover, limitations could be circumvented by, for example, the development of inhibitory anti-PS aptamers. Aptamers are small RNA molecules that bind proteins with high affinity and specificity; many such aptamers inhibit the activities of their target proteins. Moreover, any adverse effects of an aptamer can be reversed quickly with an RNA oligomer complementary to the aptamer.41
In sum, the current treatment of HB involves an infusion of purified FIX. Although this replacement therapy provides some benefits, it is expensive. Thus, the PS antibody’s ability to extend FIX’s functional lifetime is a valuable enhancement to current HB therapy. We showed that such a novel adjunct is attainable and effective.
Acknowledgments
The authors acknowledge Paul Riley from Diagnostica Stago in New Jersey for providing plasma, Laurent Mosnier for helping in animal studies, Dougald Monroe for providing critical feedback, and Howard Fried for editing the manuscript.
The work is sponsored by an R01 grant from the National Institutes of Health (5R01HL118557-05; R.M.).
Authorship
Contribution: H.P.W., A.P., A.W.D., N.K., and S.C. collected experimental data; H.P.W. collected the patient samples, analyzed the data, and wrote the initial version of the manuscript; N.K. and B.C.C. performed the mouse experiment; P.R. performed the statistical analysis; P.R. and M.J. collected HB plasma; M.C.V. was involved in critically analyzing the manuscript; D.P.-V. was involved in critically analyzing the manuscript and helped write the final manuscript; A.L.P. analyzed the manuscript and helped in writing the final draft; and R.M. designed the study, analyzed the data, and wrote the final manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Rinku Majumder, Department of Interdisciplinary Oncology, Louisiana State University Health Sciences Center, Clinical Sciences Research Bldg – 549, 533 Bolivar St, New Orleans, LA 70112; email: rmajum@lsuhsc.edu.
References
Author notes
Data are available on request from the corresponding author, Rinku Majumder (rmajum@lsuhsc.edu).
The full-text version of this article contains a data supplement.