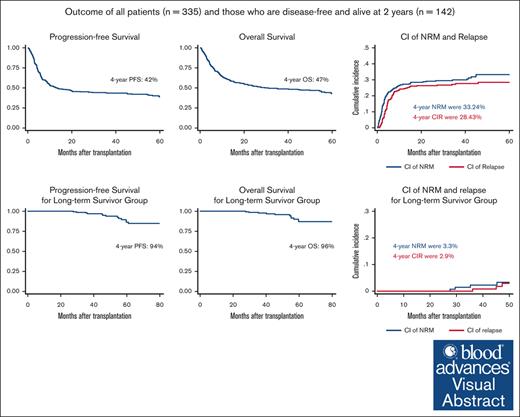
Key Point
Haplo-SCT is associated with excellent long-term outcomes for patients who are relapse-free at 2 years after transplant.
Visual Abstract
The introduction of posttransplant cyclophosphamide (PTCy)–based graft-versus-host disease (GVHD) prophylaxis lead to significant improvements in haploidentical stem cell transplantation (haplo-SCT) outcomes over the past decade. We retrospectively assessed long-term outcomes of patients who had their first haplo-SCT between February 2009 and March 2019. Long-term survivors were defined as patients who were alive and disease-free at 2 years after transplant. Three hundred thirty-five patients with a median age of 48 years (range, 18-72) were identified. Of these, 142 patients were disease-free and alive at 2 years after transplant. The 4-year progression-free survival (PFS) and overall survival (OS) for all study patients were 42% and 47%, respectively. With a median follow-up of 52 months for the long-term survivor group, the 4-year PFS and OS were 94% and 96%, respectively. The 4-year cumulative incidence of relapse and non-relapse mortality (NRM) were 2.9% and 3.3%, respectively. Age ≥55 years was the only predictive factor in multivariate analysis for inferior PFS (hazard ratio [HR], 3.41; 95% confidence interval [CI], 1.21-9.60; P = .020) and OS (HR, 3.31; 95% CI, 1.08-10.18; P = .037). Thirteen patients (9%) died in the long-term survivor group, only 2 of whom died of relapsed disease. Secondary primary malignancy was the most frequent cause of NRM (n = 4), followed by infection (n = 2). For haplo-SCT with PTCy–based GVHD prophylaxis, our findings suggest an excellent long-term survival for patients who were disease-free and alive at 2 years after transplant. Late relapses were rare, and age was the only predictive factor for long-term outcomes.
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-SCT) remains the only curative treatment intervention for a large proportion of patients with high-risk hematologic malignancies. Until late 2000s, successful transplants relied primarily on the availability of fully human leukocyte antigen (HLA)–matched donors, as patients with haploidentical stem cell transplantation (haplo-SCT) from first-degree mismatched related donors had poor outcomes.1 The failure of haplo-SCT was mainly attributed to increased risk of graft-versus-host disease (GVHD), graft failure, and infections. The introduction of posttransplant cyclophosphamide (PTCy) lead to remarkable improvements in unmanipulated haplo-SCT outcomes,2-5 with significant reduction in GVHD, graft failure, and non-relapse mortality (NRM).
With the improved haplo-SCT outcomes, there has been a steady increase in the number of haplo-SCT recipients over the past decade. Hence, a larger proportion of patients now have access to allo-SCT, most of whom had otherwise in the past, limited alternative donor sources and a missed opportunity for cure. Despite the very encouraging early results, there remains limited data on the long-term outcomes for patients with high-risk hematologic malignancies who received haplo-SCT. We aimed from this study to assess the long-term outcomes of patients who underwent unmanipulated haplo-SCTs with PTCy–based GVHD prophylaxis.
Methods
Patients
In this retrospective study, we included all consecutive adult patients with high-risk hematologic malignancies who underwent their first haplo-SCT between February 2009 and March 2019. Disease eligibility included patients with acute myeloid leukemia, myelodysplastic syndrome (MDS), acute lymphoblastic leukemia, myeloproliferative neoplasms (MPNs), and patients with lymphoid malignancies. The study was approved by the institutional review board at The University of Texas MD Anderson Cancer Center.
Clinical endpoints
The primary end points included progression-free survival (PFS) and overall survival (OS) for the long-term survivors. Long-term survivors were defined as patients who were alive and disease-free at 2 years after transplant. Patients who relapsed/progressed or died without progression within 2 years of transplant were grouped under short-term disease-free survivors. OS was defined as the time from transplant to death from any cause. PFS was defined as the time from transplant to disease progression or death from any cause. Surviving patients were censored at the time of the last follow-up. Secondary end points included cumulative incidence of relapse (CIR), NRM and GVHD-free relapse-free survival (GRFS). NRM was defined as death without evidence of relapse or progression of malignancy. Disease relapse/progression was defined as any evidence of recurrence if they were in remission at time of transplant, otherwise defined as progression of malignancy if they were not in remission at time of transplant. GRFS was defined as the time from transplant to the first occurrence of any of the following events: grade 3-4 acute GVHD, extensive chronic GVHD, disease relapse or death from any cause.
Statistical analysis
The baseline patient-, disease-, and treatment-related characteristics were summarized using descriptive statistics. Median and range used for continuous measures and proportions and frequencies for categorical measures. For comparisons, the chi-square or Fisher exact test were used for categorical variables, and the Wilcoxon rank-sum test was used to compare continuous variables. Kaplan-Meier method was used to estimate time-to-event data including PFS and OS. Fine and Gray competing risk regression model was used to compute the cumulative incidence of progression/relapse and for NRM. The primary focus of this analysis was for patients who survived for at least 2 years after transplant without evidence of disease progression, and hence we chose the landmark cutoff point at 2 years for the long-term survivor group. Predictors of long-term survival were evaluated using Cox proportional hazards regression analysis. Variables with significant P value of < .1 in the univariate analysis were included in the final multivariate model. P values were 2-sided and significance level were set at <.05. The 95% confidence intervals (CIs) were provided for survival probabilities and/or cumulative incidences. Statistical analyses were performed using primarily STATA 16.0 (StataCorp, College Station, TX).
Results
Patient characteristics
Three hundred and thirty-five patients with a median age at transplantation of 48 years (range, 18-72) were identified during the study period. Of these, 142 patients were disease-free and alive at 2 years after transplant and were grouped under long-term survivors for the sake of this analysis. The median age for patients in the long-term survivor group was 45 years (range, 18-72). Table 1 summarizes patient, disease, and transplant characteristics for all study patients, long-term survivors, and short-term disease-free survivors. Overall, there were no notable differences in baseline characteristics between all study patients and the long-term survivors, but a higher proportion of patients with low/intermediate disease risk index (DRI) were noted in the long-term survivor group. A total of 183 patients were identified in the short-term disease-free survivor group: 88 had disease progression and 95 died within 2 years of transplant.
Baseline patient, disease, and transplant characteristics for all study patients, and for long-term vs short-term survivors
| . | All study patients∗ . | Disease-free and alive at 2 years . | Progression and/or death at 2 years . | P value . |
|---|---|---|---|---|
| (N=335) . | (N=142) . | (N=183) . | ||
| Age at transplant, y | ||||
| Median (range) | 48 (18-72) | 45 (18-72) | 52 (20-70) | .086 |
| Age at transplant, n (%) | ||||
| <55 y | 206 (61.49%) | 95 (66.90%) | 105 (57.38%) | .086 |
| ≥55 y | 129 (38.51%) | 47 (33.10%) | 78 (42.62%) | |
| Gender, n (%) | ||||
| Male | 195 (58.21%) | 82 (57.75%) | 105 (57.38%) | 1.000 |
| Female | 140 (41.79%) | 60 (42.25%) | 78 (42.62%) | |
| Race, n (%) | ||||
| Asian | 22 (6.71%) | 9 (6.47%) | 12 (6.70%) | |
| Black | 69 (21.04%) | 35 (25.18%) | 32 (17.88%) | .374 |
| Hispanic | 72 (21.95%) | 32 (23.02%) | 39 (21.79%) | |
| White | 165 (50.30%) | 63 (45.32%) | 96 (53.63%) | |
| Disease subtype, n (%) | ||||
| AML/MDS | 195 (58.21%) | 79 (55.63%) | 112 (61.20%) | |
| ALL | 55 (16.42%) | 21 (14.79%) | 32 (17.49%) | .115 |
| MPNs | 37 (11.04%) | 22 (15.49%) | 13 (7.10%) | |
| Lymphoid malignancies | 48 (14.33%) | 20 (14.08%) | 26 (14.21%) | |
| Time from diagnosis to transplant, n (%) | ||||
| <6 mo | 90 (26.87%) | 38 (26.76%) | 51 (27.87%) | .749 |
| 6-12 mo | 80 (50.75%) | 37 (26.06%) | 41 (22.40%) | |
| >12 mo | 165 (49.25%) | 67 (47.18%) | 91 (49.73%) | |
| Period of transplant, n (%) | ||||
| Transplant during 2009 to 2012 | 68 (20.30%) | 29 (20.42%) | 38 (20.77%) | |
| Transplant during 2013 to 2016 | 158 (47.16%) | 69 (48.59%) | 87 (47.54%) | .989 |
| Transplant during 2017 to 2019 | 109 (32.54%) | 44 (30.99%) | 58 (31.69%) | |
| KPS at transplant, n (%) | ||||
| KPS 90-100 | 206 (68.21%) | 92 (70.23%) | 107 (66.05%) | .454 |
| KPS < 90 | 96 (31.79%) | 39 (29.77%) | 55 (33.95%) | |
| DRI, n (%) | ||||
| Low/intermediate DRI | 189 (59.06%) | 99 (72.79%) | 82 (47.13%) | <.0001 |
| High/very high DRI | 131 (40.94%) | 37 (27.21%) | 92 (52.87%) | |
| HCT-CI, n (%) | ||||
| HCT-CI ≤ 3 | 217 (64.78%) | 99 (69.72%) | 112 (61.20%) | .128 |
| HCT-CI > 3 | 118 (35.22%) | 43 (30.28%) | 71 (38.80%) | |
| Stem cell source, n (%) | ||||
| Peripheral blood | 53 (15.82%) | 18 (12.68%) | 32 (17.49%) | .279 |
| Bone marrow | 282 (84.18%) | 124 (87.32%) | 151 (82.51%) | |
| Conditioning regimen, n (%) | ||||
| Reduced intensity | 297 (88.66%) | 125 (88.03%) | 164 (89.62%) | .723 |
| Myeloablative | 38 (11.34%) | 17 (11.97%) | 19 (10.38%) | |
| Patient-donor CMV, n (%) | ||||
| Seropositive-seropositive | 185 (64.01%) | 77 (65.81%) | 102 (61.82%) | .531 |
| Seropositive-seronegative | 104 (35.99%) | 40 (34.19%) | 63 (38.18%) | |
| Donor age, n (%) | ||||
| <35 y | 168 (50.15%) | 67 (47.18%) | 96 (52.46%) | .372 |
| ≥35 y | 167 (49.85%) | 75 (52.82%) | 87 (47.54%) | |
| Donor/recipient gender, n (%) | ||||
| Male/male | 125 (37.31%) | 50 (35.21%) | 69 (37.70%) | |
| Female/female | 60 (17.91%) | 29 (20.42%) | 30 (16.39%) | .611 |
| Male/female | 80 (23.88%) | 31 (21.83%) | 48 (26.23%) | |
| Female/male | 70 (20.90%) | 32 (22.54%) | 36 (19.67%) | |
| Donor/recipient relationship, n (%) | ||||
| Parent/child | 36 (10.75%) | 15 (10.56%) | 20 (10.93%) | |
| Sibling/sibling | 132 (39.40%) | 62 (43.66%) | 63 (34.43%) | .192 |
| Child/parent | 164 (48.96%) | 65 (45.78%) | 97 (53.00%) | |
| Other | 3 (0.90%) | 0 | 3 (1.64%) | |
| Anti-HLA antibody, n (%) | ||||
| No DSA | 240 (71.64%) | 105 (73.95%) | 128 (69.95%) | |
| Anti HLA class I Ab | 42 (12.54%) | 16 (11.27%) | 24 (13.11%) | .801 |
| Anti HLA class II Ab | 25 (7.46%) | 9 (6.34%) | 16 (8.74%) | |
| Anti HLA class I and II Ab | 28 (8.36%) | 12 (8.45%) | 15 (8.20%) | |
| Acute GVHD grades 2-4, n (%) | ||||
| No | 191 (57.01%) | 90 (63.38%) | 96 (52.46%) | .055 |
| Yes | 144 (42.99%) | 52 (36.62%) | 87 (47.54%) | |
| Chronic GVHD, n (%) | ||||
| No | 290 (86.57%) | 114 (80.28%) | 167 (91.26%) | .005 |
| Yes | 45 (13.43%) | 28 (19.72%) | 16 (8.74%) |
| . | All study patients∗ . | Disease-free and alive at 2 years . | Progression and/or death at 2 years . | P value . |
|---|---|---|---|---|
| (N=335) . | (N=142) . | (N=183) . | ||
| Age at transplant, y | ||||
| Median (range) | 48 (18-72) | 45 (18-72) | 52 (20-70) | .086 |
| Age at transplant, n (%) | ||||
| <55 y | 206 (61.49%) | 95 (66.90%) | 105 (57.38%) | .086 |
| ≥55 y | 129 (38.51%) | 47 (33.10%) | 78 (42.62%) | |
| Gender, n (%) | ||||
| Male | 195 (58.21%) | 82 (57.75%) | 105 (57.38%) | 1.000 |
| Female | 140 (41.79%) | 60 (42.25%) | 78 (42.62%) | |
| Race, n (%) | ||||
| Asian | 22 (6.71%) | 9 (6.47%) | 12 (6.70%) | |
| Black | 69 (21.04%) | 35 (25.18%) | 32 (17.88%) | .374 |
| Hispanic | 72 (21.95%) | 32 (23.02%) | 39 (21.79%) | |
| White | 165 (50.30%) | 63 (45.32%) | 96 (53.63%) | |
| Disease subtype, n (%) | ||||
| AML/MDS | 195 (58.21%) | 79 (55.63%) | 112 (61.20%) | |
| ALL | 55 (16.42%) | 21 (14.79%) | 32 (17.49%) | .115 |
| MPNs | 37 (11.04%) | 22 (15.49%) | 13 (7.10%) | |
| Lymphoid malignancies | 48 (14.33%) | 20 (14.08%) | 26 (14.21%) | |
| Time from diagnosis to transplant, n (%) | ||||
| <6 mo | 90 (26.87%) | 38 (26.76%) | 51 (27.87%) | .749 |
| 6-12 mo | 80 (50.75%) | 37 (26.06%) | 41 (22.40%) | |
| >12 mo | 165 (49.25%) | 67 (47.18%) | 91 (49.73%) | |
| Period of transplant, n (%) | ||||
| Transplant during 2009 to 2012 | 68 (20.30%) | 29 (20.42%) | 38 (20.77%) | |
| Transplant during 2013 to 2016 | 158 (47.16%) | 69 (48.59%) | 87 (47.54%) | .989 |
| Transplant during 2017 to 2019 | 109 (32.54%) | 44 (30.99%) | 58 (31.69%) | |
| KPS at transplant, n (%) | ||||
| KPS 90-100 | 206 (68.21%) | 92 (70.23%) | 107 (66.05%) | .454 |
| KPS < 90 | 96 (31.79%) | 39 (29.77%) | 55 (33.95%) | |
| DRI, n (%) | ||||
| Low/intermediate DRI | 189 (59.06%) | 99 (72.79%) | 82 (47.13%) | <.0001 |
| High/very high DRI | 131 (40.94%) | 37 (27.21%) | 92 (52.87%) | |
| HCT-CI, n (%) | ||||
| HCT-CI ≤ 3 | 217 (64.78%) | 99 (69.72%) | 112 (61.20%) | .128 |
| HCT-CI > 3 | 118 (35.22%) | 43 (30.28%) | 71 (38.80%) | |
| Stem cell source, n (%) | ||||
| Peripheral blood | 53 (15.82%) | 18 (12.68%) | 32 (17.49%) | .279 |
| Bone marrow | 282 (84.18%) | 124 (87.32%) | 151 (82.51%) | |
| Conditioning regimen, n (%) | ||||
| Reduced intensity | 297 (88.66%) | 125 (88.03%) | 164 (89.62%) | .723 |
| Myeloablative | 38 (11.34%) | 17 (11.97%) | 19 (10.38%) | |
| Patient-donor CMV, n (%) | ||||
| Seropositive-seropositive | 185 (64.01%) | 77 (65.81%) | 102 (61.82%) | .531 |
| Seropositive-seronegative | 104 (35.99%) | 40 (34.19%) | 63 (38.18%) | |
| Donor age, n (%) | ||||
| <35 y | 168 (50.15%) | 67 (47.18%) | 96 (52.46%) | .372 |
| ≥35 y | 167 (49.85%) | 75 (52.82%) | 87 (47.54%) | |
| Donor/recipient gender, n (%) | ||||
| Male/male | 125 (37.31%) | 50 (35.21%) | 69 (37.70%) | |
| Female/female | 60 (17.91%) | 29 (20.42%) | 30 (16.39%) | .611 |
| Male/female | 80 (23.88%) | 31 (21.83%) | 48 (26.23%) | |
| Female/male | 70 (20.90%) | 32 (22.54%) | 36 (19.67%) | |
| Donor/recipient relationship, n (%) | ||||
| Parent/child | 36 (10.75%) | 15 (10.56%) | 20 (10.93%) | |
| Sibling/sibling | 132 (39.40%) | 62 (43.66%) | 63 (34.43%) | .192 |
| Child/parent | 164 (48.96%) | 65 (45.78%) | 97 (53.00%) | |
| Other | 3 (0.90%) | 0 | 3 (1.64%) | |
| Anti-HLA antibody, n (%) | ||||
| No DSA | 240 (71.64%) | 105 (73.95%) | 128 (69.95%) | |
| Anti HLA class I Ab | 42 (12.54%) | 16 (11.27%) | 24 (13.11%) | .801 |
| Anti HLA class II Ab | 25 (7.46%) | 9 (6.34%) | 16 (8.74%) | |
| Anti HLA class I and II Ab | 28 (8.36%) | 12 (8.45%) | 15 (8.20%) | |
| Acute GVHD grades 2-4, n (%) | ||||
| No | 191 (57.01%) | 90 (63.38%) | 96 (52.46%) | .055 |
| Yes | 144 (42.99%) | 52 (36.62%) | 87 (47.54%) | |
| Chronic GVHD, n (%) | ||||
| No | 290 (86.57%) | 114 (80.28%) | 167 (91.26%) | .005 |
| Yes | 45 (13.43%) | 28 (19.72%) | 16 (8.74%) |
Totals may vary because of missing data.
ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; CMV, cytomegalovirus; DSA, donor-specific antibody; HCT-CI, hematopoietic cell transplantation–specific comorbidity index; KPS, Karnofsky performance scale.
Ten patients had a follow-up time of <2 years and were disease-free and alive at time of last follow-up, and hence were excluded from the long-term survivor and the short-term disease-free survivor groups.
Compared with the long-term survivor group, these patients had significantly higher proportion of high/very high risk DRI (53% vs 27%; P < .0001), and were older (median age, 52 vs 45; P = .086) and with higher hematopoietic cell transplantation–specific comorbidity index of >3 (39% vs 30%; P = .128).
PFS and OS
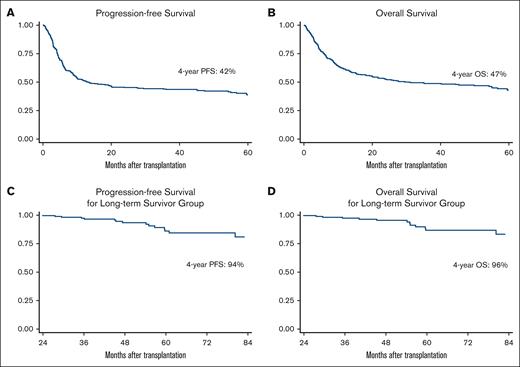
With a median follow-up of 50 months (range, 5-133), the 4-year PFS and OS rates for all study patients were 42% and 47%, respectively (Figure 1A-B). For the long-term survivors, with a median follow-up of 52 months (range, 24-133), the median PFS and OS were not reached and the 4-year PFS and OS rates were 94% and 96%, respectively (Figure 1C-D).
Progression-free survival and overall survival. PFS (A) and OS (B) for all study populations. PFS (C) and OS (D) for patients who are disease-free and alive at 2 years from transplant.
Progression-free survival and overall survival. PFS (A) and OS (B) for all study populations. PFS (C) and OS (D) for patients who are disease-free and alive at 2 years from transplant.
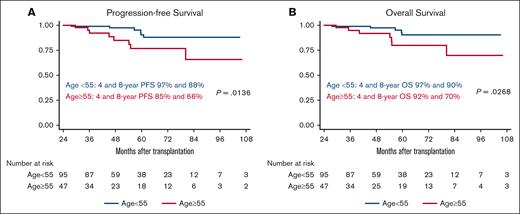
Table 2 summarizes univariable analysis for several of the potential risk factors to predict transplant outcomes in the long-term survivor group (Table 2). In both univariable and multivariate analyses, only age ≥55 years was significantly associated with both inferior PFS (hazard ratio, 3.409; 95% CI, 1.211-9.600; P = .020) and OS (hazard ratio, 3.312; 95% CI, 1.078-10.180; P = .037). The 4-year and 8-year PFS for patients aged ≥55 years were 85% and 66%, respectively, compared with 97% and 88%, respectively, for those who were <55 years (P = .0136). Similarly, younger patients had better 4-year and 8-year OS of 97% and 90%, respectively, compared with 92% and 70%, respectively for patients aged ≥55 years (P = .0268) (Figure 2A-B).
Univariable analysis evaluating predictors of PFS, OS, NRM and relapse rate of the long-term survivor group
| . | N . | PFS . | P value . | OS . | P value . | NRM . | P value . | Relapse rate . | P value . |
|---|---|---|---|---|---|---|---|---|---|
| 144 . | HR (95% CI) . | HR (95% CI) . | HR (95% CI) . | HR (95% CI) . | |||||
| Age | |||||||||
| <55 y | 95 | 1 | 1 | 1 | 1 | ||||
| ≥55 y | 47 | 3.409 (1.211-9.600) | .02 | 3.312 (1.078-10.180) | .037 | 2.475 (0.769-7.966) | .129 | 7.128 (0.693-73.312) | .099 |
| Sex | |||||||||
| Male | 82 | 1 | 1 | 1 | |||||
| Female | 60 | 0.416 (0.129-1.346) | .143 | 0.551 (0.168-1.812) | .327 | 0.679 (0.204-2.257) | .528 | N/A | |
| Race | |||||||||
| White | 63 | 1 | 1 | 1 | |||||
| Asian | 9 | 0.453 (0.049-4.172) | .489 | 0.707 (0.079-6.333) | .757 | 0.952 (0.073-12.353) | .970 | N/A | |
| Black | 35 | 0.536 (0.317-2.093) | .370 | 0.781 (0.194-3.152) | .729 | 1.130 (0.273-4.674) | .866 | N/A | |
| Hispanic | 32 | 0.607 (0.159-2.314) | .465 | 0.871 (0.217-3.488) | .845 | 1.277 (0.284-5.737) | .750 | N/A | |
| Time from diagnosis to transplant | |||||||||
| <6 mo | 38 | 1 | 1 | 1 | |||||
| 6-12 mo | 37 | 0.922 (0.620-1.371) | .689 | 0.927 (0.615-1.396) | .927 | 1.457 (0.239-8.866) | .683 | N/A | |
| >12 mo | 67 | 0.964 (0.693-1.340) | .825 | 0.915 (0.650-1.290) | .915 | 1.439 (0.291-7.122) | .655 | 0.531 (0.079-3.555) | .514 |
| Period of transplant | |||||||||
| Transplant 2009 to 2012 | 29 | 1 | 1 | 1 | 1 | .329 | |||
| Transplant 2013 to 2016 | 69 | 0.653 (0.215-1.982) | .452 | 0.713 (0.212-2.397) | .584 | 0.577 (1.79-1.860) | .357 | 1.001 (0.077-12.933) | |
| Transplant 2017 to 2019 | 44 | 0.587 (0.060-5.767) | .648 | 0.772 (0.073-8.210) | .830 | N/A | 5.163 (0.191-139.261) | ||
| KPS | |||||||||
| 90-100 | 92 | 1 | 1 | 1 | 1 | ||||
| <90 | 39 | 1.749 (0.537-5.696) | .354 | 1.551 (0.409-5.885) | .519 | 1.048 (0.217-5.051) | .953 | 4.021 (0.563-28.718) | .165 |
| DRI | |||||||||
| Low/intermediate | 99 | 1 | 1 | 1 | 1 | ||||
| High/very high | 37 | 2.161 (0.748-6.242) | .155 | 2.097 (0.664-6.626) | .207 | 1.856 (0.547-6.230) | .321 | 2.752 (0.379-19.965) | .317 |
| HCT-CI | |||||||||
| ≤3 | 99 | 1 | 1 | 1 | 1 | ||||
| >3 | 43 | 0.766 (0.240-2.447) | .654 | 1.013 (0.309-3.326) | .983 | 0.794 (0.227-2.780) | .718 | 0.719 (0.073-7.089) | .777 |
| Patient-donor CMV | |||||||||
| Seropositive-seropositive | 77 | 1 | 1 | 1 | 1 | ||||
| Seropositive-seronegative | 40 | 0.952 (0.290-3.128) | .936 | 0.920 (0.253-3.353) | .9 | 0.708 (0.195-2.568) | .6 | 0.914 (0.178-4.692) | .915 |
| Donor Age | |||||||||
| <35 y | 67 | 1 | 1 | 1 | 1 | ||||
| ≥35 y | 75 | 0.795 (0.288-2.199) | .659 | 0.735 (0.246-2.188) | .580 | 0.759 (0.238-2.427) | .129 | 0.868 (0.124-6.065) | .887 |
| Donor/recipient gender | |||||||||
| Male/male | 50 | 1 | 1 | 1 | 1 | ||||
| Female/female | 29 | 0.523 (0.106-2.607) | .431 | 0.863 (0.158-4.715) | .865 | 1.089 (0.180-6.567) | .926 | N/A | N/A |
| Male/female | 31 | 0.409 (0.079-2.108) | .286 | 0.682 (0.123-3.788) | .662 | 0.842 (0.159-4.450) | .840 | N/A | N/A |
| Female/male | 32 | 1.295 (0.392-4.282) | .671 | 2.077 (0.547-7.879) | .283 | 2.097 (0.452-9.723) | .344 | 0.495 (0.049-5.015) | .552 |
| Donor/recipient relationship | |||||||||
| Parent/child | 15 | 1 | 1 | 1 | 1 | ||||
| Sibling/sibling | 62 | 0.919 (0.101-8.294) | .940 | 0.640 (0.066-6.202) | .700 | 0.665 (0.71-6.228) | .721 | N/A | N/A |
| Child/parent | 65 | 2.635 (0.335-20.689) | .357 | 2.310 (0.291-18.351) | .428 | 1.745 (0.222-13.681) | .596 | N/A | N/A |
| Anti HLA antibody | |||||||||
| No DSA | 105 | 1 | 1 | 1 | |||||
| Anti HLA class I Ab | 16 | 1.119 (0.245-5.110) | .885 | 1.391 (0.298-6.484) | .674 | 1.744 (0.325-9.348) | 0.516 | N/A | N/A |
| Anti HLA class II Ab | 9 | 0.900 (0.113-7.156) | .920 | 1.165 (0.145-9.373) | .886 | 1.431 (0.393-5.217) | 0.587 | N/A | N/A |
| Anti HLA class I and II Ab | 12 | 0.598 (0.076-4.674) | .624 | 0.791 (0.100-6.280) | .824 | 0.995 (0.114-8.679) | 0.996 | N/A | N/A |
| Acute GVHD grades 2-4 | |||||||||
| No | 90 | 1 | 1 | 1 | 1 | ||||
| Yes | 52 | 1.031 (0.366-2.899) | .954 | 1.006 (0.328-3.080) | .992 | 0.889 (0.269-2.938) | .847 | 1.525 (0.231-10.047) | .661 |
| Chronic GVHD at 2 years | |||||||||
| No | 114 | 1 | 1 | 1 | 1 | ||||
| Yes | 28 | 1.642 (0.523-5.160) | .396 | 1.200 (0.328-4.392) | .782 | 1.646 (0.416-6.516) | .477 | 1.434 (0.148-13.922) | .756 |
| . | N . | PFS . | P value . | OS . | P value . | NRM . | P value . | Relapse rate . | P value . |
|---|---|---|---|---|---|---|---|---|---|
| 144 . | HR (95% CI) . | HR (95% CI) . | HR (95% CI) . | HR (95% CI) . | |||||
| Age | |||||||||
| <55 y | 95 | 1 | 1 | 1 | 1 | ||||
| ≥55 y | 47 | 3.409 (1.211-9.600) | .02 | 3.312 (1.078-10.180) | .037 | 2.475 (0.769-7.966) | .129 | 7.128 (0.693-73.312) | .099 |
| Sex | |||||||||
| Male | 82 | 1 | 1 | 1 | |||||
| Female | 60 | 0.416 (0.129-1.346) | .143 | 0.551 (0.168-1.812) | .327 | 0.679 (0.204-2.257) | .528 | N/A | |
| Race | |||||||||
| White | 63 | 1 | 1 | 1 | |||||
| Asian | 9 | 0.453 (0.049-4.172) | .489 | 0.707 (0.079-6.333) | .757 | 0.952 (0.073-12.353) | .970 | N/A | |
| Black | 35 | 0.536 (0.317-2.093) | .370 | 0.781 (0.194-3.152) | .729 | 1.130 (0.273-4.674) | .866 | N/A | |
| Hispanic | 32 | 0.607 (0.159-2.314) | .465 | 0.871 (0.217-3.488) | .845 | 1.277 (0.284-5.737) | .750 | N/A | |
| Time from diagnosis to transplant | |||||||||
| <6 mo | 38 | 1 | 1 | 1 | |||||
| 6-12 mo | 37 | 0.922 (0.620-1.371) | .689 | 0.927 (0.615-1.396) | .927 | 1.457 (0.239-8.866) | .683 | N/A | |
| >12 mo | 67 | 0.964 (0.693-1.340) | .825 | 0.915 (0.650-1.290) | .915 | 1.439 (0.291-7.122) | .655 | 0.531 (0.079-3.555) | .514 |
| Period of transplant | |||||||||
| Transplant 2009 to 2012 | 29 | 1 | 1 | 1 | 1 | .329 | |||
| Transplant 2013 to 2016 | 69 | 0.653 (0.215-1.982) | .452 | 0.713 (0.212-2.397) | .584 | 0.577 (1.79-1.860) | .357 | 1.001 (0.077-12.933) | |
| Transplant 2017 to 2019 | 44 | 0.587 (0.060-5.767) | .648 | 0.772 (0.073-8.210) | .830 | N/A | 5.163 (0.191-139.261) | ||
| KPS | |||||||||
| 90-100 | 92 | 1 | 1 | 1 | 1 | ||||
| <90 | 39 | 1.749 (0.537-5.696) | .354 | 1.551 (0.409-5.885) | .519 | 1.048 (0.217-5.051) | .953 | 4.021 (0.563-28.718) | .165 |
| DRI | |||||||||
| Low/intermediate | 99 | 1 | 1 | 1 | 1 | ||||
| High/very high | 37 | 2.161 (0.748-6.242) | .155 | 2.097 (0.664-6.626) | .207 | 1.856 (0.547-6.230) | .321 | 2.752 (0.379-19.965) | .317 |
| HCT-CI | |||||||||
| ≤3 | 99 | 1 | 1 | 1 | 1 | ||||
| >3 | 43 | 0.766 (0.240-2.447) | .654 | 1.013 (0.309-3.326) | .983 | 0.794 (0.227-2.780) | .718 | 0.719 (0.073-7.089) | .777 |
| Patient-donor CMV | |||||||||
| Seropositive-seropositive | 77 | 1 | 1 | 1 | 1 | ||||
| Seropositive-seronegative | 40 | 0.952 (0.290-3.128) | .936 | 0.920 (0.253-3.353) | .9 | 0.708 (0.195-2.568) | .6 | 0.914 (0.178-4.692) | .915 |
| Donor Age | |||||||||
| <35 y | 67 | 1 | 1 | 1 | 1 | ||||
| ≥35 y | 75 | 0.795 (0.288-2.199) | .659 | 0.735 (0.246-2.188) | .580 | 0.759 (0.238-2.427) | .129 | 0.868 (0.124-6.065) | .887 |
| Donor/recipient gender | |||||||||
| Male/male | 50 | 1 | 1 | 1 | 1 | ||||
| Female/female | 29 | 0.523 (0.106-2.607) | .431 | 0.863 (0.158-4.715) | .865 | 1.089 (0.180-6.567) | .926 | N/A | N/A |
| Male/female | 31 | 0.409 (0.079-2.108) | .286 | 0.682 (0.123-3.788) | .662 | 0.842 (0.159-4.450) | .840 | N/A | N/A |
| Female/male | 32 | 1.295 (0.392-4.282) | .671 | 2.077 (0.547-7.879) | .283 | 2.097 (0.452-9.723) | .344 | 0.495 (0.049-5.015) | .552 |
| Donor/recipient relationship | |||||||||
| Parent/child | 15 | 1 | 1 | 1 | 1 | ||||
| Sibling/sibling | 62 | 0.919 (0.101-8.294) | .940 | 0.640 (0.066-6.202) | .700 | 0.665 (0.71-6.228) | .721 | N/A | N/A |
| Child/parent | 65 | 2.635 (0.335-20.689) | .357 | 2.310 (0.291-18.351) | .428 | 1.745 (0.222-13.681) | .596 | N/A | N/A |
| Anti HLA antibody | |||||||||
| No DSA | 105 | 1 | 1 | 1 | |||||
| Anti HLA class I Ab | 16 | 1.119 (0.245-5.110) | .885 | 1.391 (0.298-6.484) | .674 | 1.744 (0.325-9.348) | 0.516 | N/A | N/A |
| Anti HLA class II Ab | 9 | 0.900 (0.113-7.156) | .920 | 1.165 (0.145-9.373) | .886 | 1.431 (0.393-5.217) | 0.587 | N/A | N/A |
| Anti HLA class I and II Ab | 12 | 0.598 (0.076-4.674) | .624 | 0.791 (0.100-6.280) | .824 | 0.995 (0.114-8.679) | 0.996 | N/A | N/A |
| Acute GVHD grades 2-4 | |||||||||
| No | 90 | 1 | 1 | 1 | 1 | ||||
| Yes | 52 | 1.031 (0.366-2.899) | .954 | 1.006 (0.328-3.080) | .992 | 0.889 (0.269-2.938) | .847 | 1.525 (0.231-10.047) | .661 |
| Chronic GVHD at 2 years | |||||||||
| No | 114 | 1 | 1 | 1 | 1 | ||||
| Yes | 28 | 1.642 (0.523-5.160) | .396 | 1.200 (0.328-4.392) | .782 | 1.646 (0.416-6.516) | .477 | 1.434 (0.148-13.922) | .756 |
HCT-CI, hematopoietic cell transplantation–specific comorbidity index; HR, hazard ratio; N/A, not applicable.
Progression-free survival and overall survival by age group. PFS (A) and OS (B) by age group for patients who are disease-free and alive at 2 years from transplant.
Progression-free survival and overall survival by age group. PFS (A) and OS (B) by age group for patients who are disease-free and alive at 2 years from transplant.
Relapse
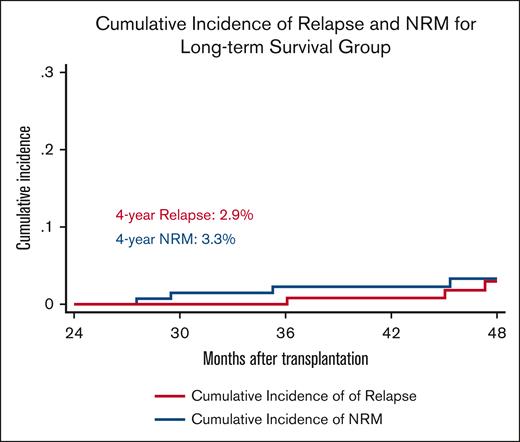
During the study period, a total of 92 (27%) relapsed, with a median time to relapse/progression for all study patients of 6 months (range, 1-61) and a 4-year CIR of 28%. In the long-term survivor group, only 4 (3%) patients progressed. The median time to relapse in this group was 46 months (range, 36-61) and with a 4-year CIR of 2.9% (Figure 3). Among the patients who relapsed within 2 years of transplant, 73 patients died of their disease and 15 patients remain alive at their last follow-up. For the 4 patients who relapsed in the long-term survivor group, 2 patients died and 2 remain alive in remission at the date of last follow-up. None of the factors analyzed in Table 2 were found to predict relapse risk, likely due to the small number of events.
Cumulative incidence of NRM and relapse for patients who are disease-free and alive at 2 years from transplant.
Cumulative incidence of NRM and relapse for patients who are disease-free and alive at 2 years from transplant.
Relapse- and non-relapse mortality
One hundred and eighty-one (54%) patients died during the study period, of which 77 patients died of relapsed disease. The estimated 4-year NRM for all study patients was 30%, and the most frequent causes of NRM in all study patients were infections and GVHD. In the long-term survivor group, 13 (9%) patients died during the study period, of which only 2 (1%) died of relapsed disease. The estimated 4-year NRM in this group was 3.3% (Figure 3), and the most common cause of death was second primary malignancy (4 patients), all of which had non–small cell lung cancers (3 were heavy smokers). Other causes of death in the long-term survivor group included infection (n = 2) and 1 each from GVHD and sudden death. Table 3 summarizes causes of death for all patients and for the long-term survivors. None of the factors analyzed in Table 2 were found to predict NRM risk, likely due to the small number of events after 2 years.
Causes of death
| . | All study patients . | Disease-free and alive at 2 y . |
|---|---|---|
| Cause of death | ||
| Relapsed disease | 77 | 2 |
| Infections | 27 | 2 |
| GVHD | 23 | 1 |
| Organ failure | 15 | 0 |
| Pneumonia | 12 | 0 |
| Hemorrhage | 6 | 0 |
| Graft failure | 5 | 0 |
| Secondary malignancy | 4 | 4 |
| ARDS | 4 | 0 |
| Sudden death | 1 | 1 |
| Unknown | 7 | 3 |
| Total deaths | 181 | 13 |
| . | All study patients . | Disease-free and alive at 2 y . |
|---|---|---|
| Cause of death | ||
| Relapsed disease | 77 | 2 |
| Infections | 27 | 2 |
| GVHD | 23 | 1 |
| Organ failure | 15 | 0 |
| Pneumonia | 12 | 0 |
| Hemorrhage | 6 | 0 |
| Graft failure | 5 | 0 |
| Secondary malignancy | 4 | 4 |
| ARDS | 4 | 0 |
| Sudden death | 1 | 1 |
| Unknown | 7 | 3 |
| Total deaths | 181 | 13 |
ARDS, acute respiratory distress syndrome.
GRFS
Table 1 summarizes the cumulative incidence rates of acute and chronic GVHD for all study patients and for those in the long-term survivor and short-term disease-free survivor groups. For all study patients, 10.75% developed severe acute grade 3-4 GVHD and 9.55% developed extensive chronic GVHD. Patients in the long-term survivor group had significantly lower rates of grade 3-4 acute GVHD compared with short-term survivors (4.22% vs 16.40%; P = .004), but the rates were not statistically significant for extensive chronic GVHD (12.68% vs 7.10% for long-term survivor vs short-term disease-free survivor groups; P = .67). Regarding GRFS, the 1-year and 2-year rates for all study patients were 45% and 39%, respectively (supplemental Figure 1A). For the long-term survivor group, the respective 1-year and 2-year GRFS rates were 90% and 85%, compared with 29% and 8% in the short-term disease-free survivor group (P < .0001) (supplemental Figure 1B).
Discussion
Given the remarkable improvements in the outcomes of haplo-SCT over the past decade, haploidentical donors are increasingly being used as an alternative donor source in patients with high-risk hematologic neoplasms. To our knowledge, this is 1 of the largest single institutional studies to report on the long-term outcomes of patients who underwent unmanipulated haplo-SCT with PTCy–based GVHD prophylaxis. We report excellent long-term survival for patients who were alive and disease-free at 2 years after transplant, with 4-year PFS and OS rates of 94% and 96%, respectively. Furthermore, we noticed very low rates of relapse and relapse-related deaths in the long-term survivor group.
Several studies reported on long-term outcomes after allogeneic SCT, but majority focused on matched donors and only a few addressed conditional survival.6-9 In 1 of the largest studies to report on conditional survival (87% of donors were matched related and the median follow-up was 80 months), the 5-year OS for patients who were alive and disease-free at 2 years was 89%.6 A second large registry study, with a longer median follow-up of 9 years, showed a 10-year OS rate of 85%.7 Both of these studies included predominantly matched related donors with a young patient population (median age <30 years). A more recent single institution study, included 389 patients, reported on conditional survival for patients who were alive and disease-free at 1 year. With a median follow-up of 48.2 months, the 5-year OS was 78%.8 In this study, in contrast to the other aforementioned studies, patients were older (median age, 51 years) and donors were predominantly matched unrelated (39%), followed by matched related (37%), and haploidentical donors (24%). The findings from our report are very encouraging, showing an excellent long-term outcome for patients who underwent transplantation from alternative haploidentical donors. A more recent large registry study using the Center for International Blood and Marrow Transplant Research database compared the outcomes of SCTs for patients with acute leukemia and MDS who received haploidentical (n = 2036) vs matched unrelated donors (284); all patients received PTCy–based GVHD prophylaxis.10 Interestingly, outcomes were favored in this study for matched unrelated SCT only in patients who received reduced intensity but not myeloablative conditioning. However, the median follow-up in this study was relatively short (1 year for matched unrelated vs 2 years for haplo-SCT), and hence long-term outcomes could not be fully assessed.
Age is known to predict transplant outcomes, and until more recently, transplant was generally limited to younger fit patients. Most of published long-term data studies included younger patient populations.6,7,9 In contrast, the median age in our study population was 48 years. In our multivariate analysis, age was the only predictive factor for both PFS and OS. For patients with age <55, the 4-year PFS and OS were 97% and 97%, respectively. Hematopoietic cell transplantation–specific comorbidity index and DRI were not predictive for long-term outcomes in our cohort, likely because most events (NRM and/or relapse) do occur in the first 2 years. Furthermore, GVHD was not predictive to long-term outcomes which can be related to low incidence of severe GVHD with PTCy–based GVHD prophylaxis and the improvements in GVHD treatments. However, longer follow-up might be needed to confirm the impact of cGVHD on long-term outcomes.
Relapse and NRM rates were low in our long-term survivor cohort. Only 3% relapsed after 2 years, and this is encouraging and somewhat favorable when compared with other studies that included predominantly matched donors.6,7,9 Likewise, the 4-year NRM was low at 3.3%, however, longer follow-up is needed because several studies showed continued increased NRM over time with allogeneic SCT.6,9,11 Similar to other studies, second primary malignancies and infections were among the most frequent causes of NRM. In contrast, we report lower rate of deaths related to GVHD which could be again attributed to the use of PTCy, better GVHD treatments, and/or the need for longer follow-up. Likewise, we have not seen an increased risk of death from cardiovascular diseases, which might be related to the type of conditioning used (majority of patients in our cohort received reduced intensity conditioning) and improvements in management of cardiovascular diseases. Despite these encouraging outcomes, we acknowledge the need for longer follow-up and larger studies to better define the impact of using PTCy on the incidence of posttransplant second primary malignancies and cardiovascular diseases.
Our study is limited by its observational retrospective nature. Despite the relatively small sample size compared with registry cancer studies, to our knowledge, this is 1 of the largest single-center studies to report on long-term outcomes of unmanipulated haplo-SCT patients, all of which received PTCy–based GVHD prophylaxis. Given that the use of haplo-SCT with PTCy-based GVHD prophylaxis has only remarkably increased over the past decade, we have limited long-term follow-up beyond 10 years.
In summary, our findings suggest excellent long-term survival outcomes for patients who underwent haplo-SCT and were disease-free at 2 years after transplant. Late relapses were very low. Late NRM was mostly related to second primary malignancies and infections, and hence, the need for increased awareness and primary prevention strategies to further improve long-term outcomes.
Authorship
Contribution: S.A.S. devised the research and designed and performed the statistical analysis; S.S. contributed to statistical analysis and wrote the first draft of the manuscript; S.A.S. and S.S. contributed to data collection, analyzed and interpreted data, and wrote the manuscript; and S.A.S., S.C., U.P., J.R., Q.B., A.A., J.C., G.R., A.O., J.I., C.H., E.S., and R.C. contributed to the treatment of patients and critically reviewed and edited the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Samer A. Srour, Department of Stem Cell Transplantation and Cellular Therapy, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd, Unit 423, Houston, TX 77030; email: ssrour@mdanderson.org.
References
Author notes
Data are available on request from the corresponding author, Samer A. Srour (ssrour@mdanderson.org).
The full-text version of this article contains a data supplement.